Review Article
Role of Boron in Plant Nutrition and Human Health
National Research Centre, Department of Fertilization Technology, Dokki, Cairo, Egypt
Boron is the 51st most common element found in the earth’s crust and is found at an average concentration of 8 mg kg-1. It is known as an essential element for plants as early as the beginning of the 20th of the 19th century. Since, that time, numerous papers on deficiency and toxicity reactions in many plant species were appeared. Essentiality of boron was evident to vascular plants, diatoms and some species of marine algae, while bacteria, fungi; green algae apparently do not require boron (Goldbach, 1997). However, some studies proved that Cyanobacteria require boron to be able to form heterocysts capable of nitrogen fixation (Bonilla et al., 1990).
Until now, the physiological role of boron in plants is a matter of controversy. This because of the lake of information about boron ion nature (Goldbach, 1997) and difficulty of boron determination as there are lot of contamination sources to samples.
Boron deficiency found to affect plant growth and reduced yields (Rerkasem, 1995; Anantawiroon et al., 1997; Chunjian et al., 1997; Dell and Haung, 1997; Carpena et al., 2000). Better growth and good yields were obtained when crops were supplied with boron (Blamey et al., 1997; Dunn et al., 2005; Li and Liang, 1997; Oyinlola, 2005; Shaaban et al., 2004; Zude et al., 1997).
Boron excess concentrations were also found to be harmful to some plant species (Chapman et al., 1997). However, some plant species proved to tolerate boron excessive concentrations (Blamey et al., 1997; Yau et al., 1997).
The main objective of this paper is to review different hypotheses and experimental results on boron postulated functions, effects of boron deficiency and toxicity and its correlation with other plant nutrients. Because of boron essentiality for human beings, some effects of boron on physiological functions are shortly reviewed.
BORON MINERALS AND DISTRIBUTION IN NATURE
The primary minerals of boron are ulexite NaCa[B5O6(OH)6]-5H2O, borax Na2[B4O5(OH)4]-8H2O and natural boric acid (sassolite) which may formed near the hot springs. The less soluble colemanite Ca[B3O4(OH)3]-2H2O is released as a result of leaching and weathering, while high-pressure produces kernite Na2[B4O5(OH)4]-2H2O which a less hydrated form of borax (Erd, 1980).
Boron concentration in rocks ranged between 10-20 mg kg-1 and 1-10 mg L-1 in seawater, while its concentration in river water is about 1/350 of that of seawater (Power and Woods, 1997). The same authors stated that soils divided into low boron soil content (<10 mg B kg-1) or high B-content (10-100 mg B kg-1) and the soluble B which is available to plants is usually about 10% of the total B. Plant tissues found to contain 10-100 mg kg-1 on dry weight basis.
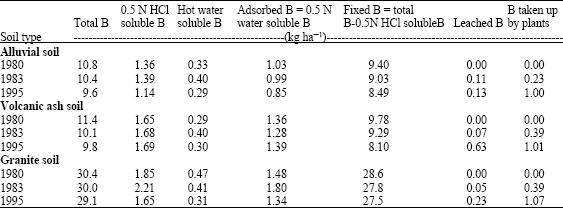
From a long term field experiment (26-29 successive crops), Eguchi and Yamanda (1997) concluded that only 10% of B applied to soil was absorbed by plants, 30-40% left in the soil, while 40-60% was leached out from the top of the soil. In the same experiment, they could determine total and extractable amounts of boron in different soil types (Table 1). They also observed that, the fixed form of B was the greatest portion of the B left in the soil followed by the adsorbed form. The hot water soluble form (considered as available to the plant roots) showed a tendency to slightly increase with the time but far from the toxic level.
BORON UPTAKE AND REQUIREMENTS BY PLANTS
Boron was proposed to taken up by the plant roots as boric acid (H3BO3)0 and/or borate [B(OH)4]-1 (Jiao et al., 2005). Being B taken up passively by the plant roots is well supported. The evidence that B tissue concentrations can be higher than those in the soil solution (Bowen, 1972; Raven, 1980), suggested that an active mechanism might also play a part in B uptake. This opinion is supported also by the findings that not all plants take up the same B when supplied with the same concentration in the growing media (Nable, 1991). Another theory suggested that both passive and active B-uptake by plants is there. Studies showed that when B is sufficient in the root medium, it is taken up in a linear trend and in case of low B-supply, the concentration in the plant tissue exceeds that of the external concentration (Dannel et al., 1997, 2000). This suggests that at sufficient levels, B is taken up passively and in deficient levels it is taken up actively against the concentration gradient. However, Miwa and Fujiwara (2010) could identify the first boron carrier and concluded that plants sense internal and external B conditions and regulate B transport by modulating the expression and/or accumulation of these transporters.Plant species varies greatly for boron demand and the differences are reflected from B-optimum concentrations in leaf tissues (Marschner, 1995). The mechanisms underlying species differences concerning boron uptake are unknown, but may result from differences in membrane permeability (Nable and Paull, 1991), species ability to translocate and mobilize boron (Bellaloui and Brown, 1998; Brown and Shelp, 1997). Accordingly, three groups concerning boron requirements were identified by Goldbach (1997) as follows: (1) Graminaceous species with very low boron requirements and rarely develop boron deficiency symptoms during the vegetative growth. Optimum leaf concentration for this group ranged between 2.0 and 5.0 μg g-1 DM. (b) Plants severely affected by high B-concentrations in the soil which are non-grass monocotyledons and dicotyledons with optimum leaf concentrations between 20 and 80 μg g-1 DM. (3) Latex containing plants: this group has high boron demand with optimum leaf concentration above 80 μg g-1 DM.
| Table 1: | Forms of boron in the soils plot along the years (Eguchi and Yamada, 1997) |
 | |
| Table 2: | Boron requirements determined for 14 plant species (Hu et al., 1996) |
 | |
In the contrary of the previous findings (Hu et al., 1996) could determine boron requirement for 14 plant species (Table 2) and found that Graminaceous species such as barley and wheat need boron for their growth with optimum concentration in the leaves ranged from 4.0 to 10 μg g-1 DW. He also suggested that the higher pectin content in the cell wall the higher required plant to boron. Using boric acid-coupled dyes, Gluesenkamp et al. (1997) found that boron was concentrated in the extension zone and the tips of root hairs.
FACTORS AFFECTING BORON AVAILABILITY FOR PLANT ROOTS
Soil pH
With increasing pH of the soil solution, B became less available to plants as a function to the increase of it's adsorbing on soil particles (Lehto, 1995). Boron adsorption by soils increased as a function of solution pH in the range of pH 3 to 9 (Barrow, 1989; Bingham et al., 1971; Keren et al., 1985; Lehto, 1995; Mezuman and Keren, 1981; Schalscha et al., 1973) and decreased in the range of pH 10 to 11.5 (Goldberg and Glaubig, 1986).
Soil Texture
Boron deficiency often occurs in plants grown in sandy soils (Osman et al., 2005; Shaaban et al., 2004, 2006). Soil adsorbed B found to be soil texture dependent and increased with increasing clay content (Mezuman and Keren, 1981; Wild and Mazaheri, 1979).
Soil Temperature and/or Soil Moisture
Boron availability decreased as a function of soil temperature in the range between 5 and 40° (Goldberg et al., 1993; Ye et al., 2003). Effect of temperature my be also an interactive effect between temperature and soil moisture, since B-deficiency found to associate with dry summer conditions (Goldberg, 1997).
Soil Calcium Carbonate Content
It is well known that addition of calcium carbonate to soil increases pH value of the soil. Thus, calcium carbonate is likely to decrease B-availability (Shaaban et al., 2004, 2006). Besides, calcium carbonate was found to act as a sink for boron in the soil, where it adsorbs a great portion of the soluble boron on the surface of their particles (Goldberg and Forster, 1991).
Soil Organic Matter
Boron adsorption and consequently availability to plant roots found to correlate with soil organic matter content. Yermiyaho et al. (1995) stated that boron adsorption increased with increasing addition of organic matter. However, an equilibrium between soluble boron and boron adsorbed by organic matter was occurred (Lehto, 1995) and hence, addition of a reasonable organic matter quantities to the soil can improve boron availability to plant roots.
POSTULATED FUNCTIONS OF BORON IN PLANTS
Because there is no radioactive isotope is available, most of the known information about boron physiological and biochemical roles in plants were speculations. However, many papers have discussed the nutrition and physiology of boron in plants.
Boron Relation to Cell Wall and Structure
Boron was reported to complex with the cell wall and many of its constituents (Ahmad et al., 2009; Goldbach, 1997; Marschner, 1995; Matoh, 1997; Taiz and Zeiger, 2002). Hemicellulose, pectin, callouse and lignin found to decrease in plants suffering from boron deficiency (Dell and Huang, 1997; Lovatt and Dugger, 1984; O’Neill et al., 2004; Roemheld and Marschner, 1991). Matoh et al. (1996) indicated that, under B-limiting conditions, cell wall contains 95-98% of the cellular B and the majority of the cell wall B (>70 %) was associated with pectin. However, plants grown under adequate B-levels contain less percentage of boron (Table 3). On the other hand, it was observed that cell wall of the Dicots. contains much higher boron than that of Monocots.
Hu and Brown (1994) determined boron levels in the cell wall of tobacco plants and proved that most of boron localize in the cell wall, especially in case of boron deficiency (Table 4).
Cellulose synthesis found also to be affected by boron deficiency. Uridine diphosphate glucose (UDPG)-pyrophosphorylase is inhibited when boron is deficient, causing a decrease in UDPG responsible about cellulose synthesis (Shelp, 1993).Boron Relation to Membrane Function
Ferrol et al. (1993) reported that boron deficiency inhibit ATP-dependant H+ transport and ATP-ase activity in sunflower roots, suggesting that inhibition of H+ gradient formation in microsomes in the absence of boron could be due to the combined effects of reduced ATP-ase activity and increased passive conductance across the membrane. Decrease of ATPase activity suggested also to alter the electrical potential gradients across membranes and decreased the transport of ions, metabolites and enzymes (Agarwala and Chatterjee, 1996; Marschner, 1995; Schon et al., 1990; Shelp, 1993).
| Table 3: | levels of boron in different plant species grown on adequate boron concentrations (Matoh et al., 1996) |
 | |
| Table 4: | Boron content in the cell wall of tobacco leaves as affected with level of boron in the growth medium (Hu and Brown, 1994) |
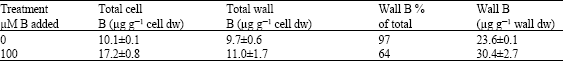 | |
| Table 5: | Effect of boron on ATP hydrolysis and H+ transport in lilium pollen grains (from Obermeyer et al., 1996) |
 | |
Obermeyer et al. (1996) suggested that boron stimulates ATP hydrolysis and H+ transport activity (Table 5) and control membrane voltage charging. A recent study stated that at least three B-binding membrane glycoproteins were detected in the B-deficient plant tissues indicating that B and certain membrane glycoproteins are involved in membrane processes associated with cell growth (Redondo-Nieto et al., 2007). Another remark that surface proteins attached to the membrane via a glycosyl-phosphatidylinositol anchor such as arabidogalactan proteins (AGP) was suggested to be B-binding structures (Goldbach and Wimmer, 2007). Moreover, a recent study showed that B deficiency caused a rapid decrease in the expression of several AGP genes in Arabidopsis roots (Camacho-Cristóbal et al., 2008), which suggests that B might act in membranes by two ways: (1) by stabilizing of membrane-molecules with cis-diol groups, (2) by regulating the expression of genes involved in membrane function.
Metabolism of Nucleic Acids and Protein Synthesis
Krueger et al. (1987) found an inhibition of DNA as short as 6 h after transferring squash plants to boron deficient media. Loomis and Durst (1991, 1992) declared that low intracellular boron concentration in plants should keep optimal protein synthesis and that boron toxicity is partially leads to the inhibition of protein synthesis through the formation of borate ester with ribose. Goldbach (1997) argued that, any control of nucleic acids or protein metabolism can hardly be responsible for the different requirements of plant species. He added that the experimental evidence for a direct and specific involvement of boron in nucleic acids metabolism or protein synthesis is scanty.
Carbohydrate Metabolism and Transport
Lewis (1980) assumed that boron controls the metabolic reactions of carbohydrate transport. Boron was also reported to control different reactions in carbohydrate metabolism such as α-amylase, Glucose 6-phosphate dehydrogenase, β-amylase and reduction of UDPG-synthesis (Goldbach, 1997). The specific B role in carbohydrate metabolism reported to be species dependent (Brown and Hu, 1998). Boron is now known to be mobile in the phloem of all species that utilize polyols (complex sugars) as primary photosynthetic metabolites. In these species a polyol-B-polyol complex is formed in the photosynthetic tissues and is transported in the phloem to currently active sink regions such as vegetative or reproductive meristems. In species that do not produce significant quantities of polyols, B once delivered to the leaf in the transpiration stream cannot reenter the phloem, resulting in essentially complete phloem immobility. Thus, B may cause an accumulation of sugars and starches or a reduction of sucrose (Agarwala and Chatterjee, 1996).
Plant Enzymes and Growth Regulators
There is no agreement on the role of boron in enzymatic activity. While Clarkson and Hanson (1980) and Marschner (1995) suggested that B has no enzymatic function in plants, many others reported that B can affect the activity of numerous enzymes within the plant cells. Accordingly, B deficiency was reported to inhibit the activity of dehydrogenases, oxidases and phosphatases (Dave, 1996; Lovatt and Dugger, 1984; Shelp, 1993). B deficiency found also to increase the activity of polyphenoloxidase (PPO) enzyme that catalyses the oxidation of phenolic compounds into quinines (Pfeffer et al., 1998; Camacho-Cristobal et al., 2002). More recent reports stated that there is an induction in the expression of glutathione S-transferase and glucosyltransferase in tobacco BY-2 cells, under B deficiency conditions (Kobayashi et al., 2004), which suggest that they might be a part of the defense system against the oxidative damage under B deficiency. These findings may be supported by the results of Ruiz et al. (2006) and Corrales et al. (2008) who reported that B induced an enhancement of glutathione levels in sunflower and maize plants subjected to aluminium stress. Boron hypothesized also to suppress IAA oxidase activity, therefore when B is in adequate supply, IAA is in ample supply (Agarwala and Chatterjee, 1996; Lovatt and Dugger, 1984; Marschner, 1995; Roemheld and Marschner, 1991). On the other hand, Jarvis and Booth (1981) found that boron and IAA were act synergistically in bean (Vigna radiata) roots and boron was not needed for the initiation of the root growth. It was also hypothesized that boron stimulates IAA-oxidase and would thus reduced the auxin level to the limit allows the subsequent growth of the roots (Jarvis et al., 1984). Goldbach et al. (1990) concluded that IAA starved tomato cells not react with differences in boron supply and suggested that there is no direct relationship between boron and metabolism or transport of IAA.
Synthesis and Metabolism of Phenolic Compounds
One of boron deficiency symptoms is the accumulation of polyphenolics causing browning of tissues, black necroses and rusting on the fruits (Camacho-Cristobal et al., 2002; Marschner, 1995). Cakmak et al. (1995) found that higher leaching from the young leaves of sunflower was occurred in case of boron deficiency (Fig. 1) and attributed this to the oxidation of phenolics and production of free radicals.
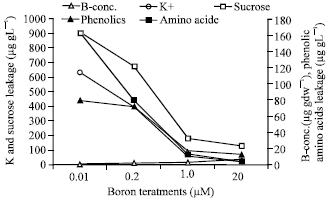 | |
| Fig. 1: | Effect of boron supply on B-concentration, leakage of K, sucrose, Phenolic and ambo acid from leaves of 10 days old sunflower (Helianthus annuus) (sucrose was determined as glucose equic., phenolics) as caffeic acid equiv. And amino acid as glycine equiv.) (Cakmak et al., 1995) |
B deficiency caused also the accumulation of two polyamine-phenolic conjugates which were not present in the B-sufficient plants (Camacho-Cristobal et al., 2002, 2005; Karioti et al., 2006). Thus, B deficiency could also induced qualitative changes in the phenolic pool of plants.
EFFECTS OF BORON DEFICIENCY
Boron Deficiency Symptoms
Boron is known to be mobile in the phloem of all species that utilize polyols (complex sugars) as a primary photosynthetic metabolite (Brown and Hu, 1998). In these species a polyol-B-polyol complex is formed in the photosynthetic tissues and is transported in the phloem to currently active sink regions such as vegetative or reproductive meristems. In species that do not produce significant quantities of polyols, B once delivered to the leaf in the transpiration stream cannot reenter the phloem, resulting in complete phloem immobility (Brown and Shelp, 1997).
Typical symptoms of B deficiency on shoots are crinkled and stunted leaves, upward-cupping leaves, chlorosis of upper leaves, decreased leaf expansion, aborted growing tips and fast-growing auxiliary shoots, yellowing and red veins on the terminal shoots, dead terminal shoots and dieback, death of small areas in the trees bark and tip of the shoots followed by progressive death of the inner bark and cambium (Brown and Hu, 1996; Jiao et al., 2005). The B deficiency symptoms on the roots include: reduced growth with brown discoloration of the root tips (Blevins and Lukaszewski, 1998; Pilbeam and Morely, 2007; Sanzonowicz et al., 1998). Symptoms can be seen on the fruits include: mall flatten or misshapen fruits, drought spot, internal cork, cracking and russet in apple, premature ripening, increased fruit drop and low seed count (Brown and Hu, 1996).
Effects on Plant Nutrients
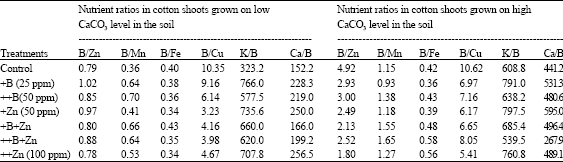
Boron deficiency found to affect most of nutrient concentrations, uptake and balance in the plant tissues. Upon its proposed effects on plasma membrane, Cakmak et al. (1995) suggested that the primary effects of boron deficiency would be an increase of membrane permeability that causes nutrient leakage from the cells and thus, lowering their concentrations in the plant organs. Marschner (1995) reported that boron application increased phosphate uptake by faba bean roots. Muehling et al. (1998) observed that bound cellular calcium was low in the boron deficient faba bean plants than sufficient ones and assumed that the decrease of membrane associated Ca2+ in the boron deficient plants is primary caused by a reduction in the number of negative charges and of Ca2+-borate complex. Camacho-Cristobal and Gonzalez-Fontes (1999) observed that nitrate content in the leaves of the deficient tobacco was dramatically decreased. They observed also that leaf contents of magnesium, calcium and specially potassium were declined under boron deficiency conditions. Zude et al. (1997) found that boron foliar application increases the concentrations of calcium, potassium and magnesium in the leaves of apple. Shaaban et al. (2004) found that boron foliar application leads to significant increases in both concentrations and uptake of calcium, potassium, iron, manganese, zinc and copper in cotton shoots especially plants grown under high calcium carbonate levels in the soil. They found also that a special nutrient balance between boron and other nutrients in the shoot tissues led to a good plant growth (Table 6). Ahmed et al. (2008) reported that boron foliar application increased uptake and concentration of nutrients in wheat leaves.
Boron Effects on Growth and Yield
Boron deficiency was found to severely affect root growth. Dell and Haung (1997) concluded that boron deficiency inhibits root elongation through limiting of cell enlargement and cell division in the growing zone of root tips and that in severe boron deficiency cases, root growth is ceases leading to the death of root tips. They added that the early inhibition in root growth by boron deficiency cause the higher shoot/root ratio. Lukaszewski and Belvins (1996) concluded that root elongation in squash seedlings depend on boron concentration in the medium. They found that the maximum elongation rate was obtained with a boron range of 5-40 μM boron and a 45% inhibition was observed when boron concentration was 1.0 μM. In the absence of boron, root growth was limited to less than 10%.
Camacho-Cristobal and Gonzalez-Fontes (1999) concluded that boron deficiency caused a substantial reduction in root and shoot weight of tobacco plants, which resulted in notably high shoot/root ratio. A reduction of both root and shoot dry weight of pea plants as a result of boron deficiency was observed by Carpena et al. (2000).
Srivastava et al. (1997) stated that boron deficiency is established as a dominant nutritional problem causing flower and pod abortion in chickpea plants. They observed that no pods or grains were formed in the absence of boron and application of 0.5 kg B ha-1 was found to optimally correct the status. On the other hand, Chunjian et al. (1997) observed that lateral buds were significantly increased in pea plants grown under boron deficiency and the case is as typical as in decapitated plants.
| Table 6: | Boron/Nutrient ratios in cotton shoot tissues (31 days age) grown on two calcium carbonate levels in the soil as affected by foliar fertilization of B, Zn and their combinations (Shaaban et al., 2004) |
 | |
Boron deficiency was reported to cause male sterility in wheat (Rerkasem, 1995). Anantawiroon et al. (1997) concluded that boron deficiency causes grain set failure in wheat and led to a great loss in the yield. Halder et al. (2007) found that the highest mean of wheat grain yield (4.8 t ha-1 was recorded with 2.0 kg B ha-1, which was significantly 45% higher over boron control (0.0 B). Ahmed et al. (2008) concluded that both silicon and boron foliar applications could correct to some extent the negative effects of salinity either on growth and yield of wheat plants grown under salinity stress conditions. Field research in Missouri showed that rice yields were greatest when soil B levels were 0.25 to 0.35 ppm by the hot water extraction method and rice receiving soil-applied B produced significantly greater yields than rice with foliar-applied B and rice with no B applied (Dunn et al., 2005).
Blamey et al. (1997) reported that boron deficiency drastically reduced the seed yield of sunflower. Li and Liang (1997) concluded that adding boron to boron deficient soils increased the yield of maize, soybean, rice and sugar beet by 8.5, 4.0, 6.6 and 10.2%, respectively. Oyinlola (2005) concluded that B soil application could increase the yield of tomato plants by 198-233% for the varieties Dandino and Roma VF, respectively.
Shaaban et al. (2004) found that boron foliar application with 25 ppm boron or 25 ppm boron+50 ppm zinc in the spray solution has significantly increased both fresh and dry weight of cotton plants grown under high calcium carbonate level in the soil (Fig. 2a, b). Ziaeyan and Rajaie (2009) stated that Zn and B fertilization significantly increased plant biological yield, grain yield, thousand grain weight, number of grains per stalk, grain protein content and the concentration of B and Zn in corn tissues grown under high CaCO3 conditions.
Zude et al. (1997) stated that boron foliar fertilization 4 weeks after petal fall enhanced boron concentration in apple fruit and reduced both the bitter pit and fruit skin cracking appearance in the cold storage. Brown (2001) stated that boron foliar application could correct transient nutrient deficiency in of pistachio and olive that is not responsive to B soil application and improve fruit site and yield.
Boron Excess
It is generally accepted that there is a narrow range of supply between boron deficiency and boron toxicity in plants (Marschner, 1995). However, Chapman et al. (1997) confirmed that fact, but argued that many crop species are tolerant to high B supply and no generalization should be made in this regard.
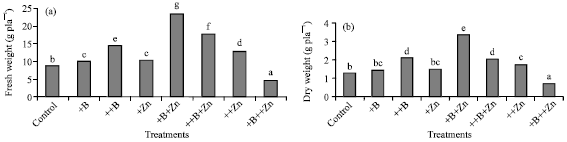 | |
| Fig. 2: | Shoot fresh and dry weight of cotton plants grown on soil contain high CaCo3 level as responded to boron and zinc treatments (bars with same letters are not significantly different, p-0.05) (Shaaban et al., 2004) |
When occurs in excess in the soil due to low rainfall, irrigation and pollution (Roessner et al., 2006), B becomes toxic to sensitive and moderately sensitive plants. A number of physiological processes was reported to be affected by B toxicity include inhibition of cell division and elongation, disruption of cell wall and metabolic disturbance (Reid et al., 2004; Stangoulis and Reid, 2002). Thus, one of the main symptoms of B toxicity is rapid inhibition of root elongation (Reid et al., 2004; Choi et al., 2007). On the other hand, plants exposed to B toxicity found to exhibit increases of malondialdehyde (MDA) and hydrogen peroxide (H2O2) content, resulting in oxidative stress and membrane peroxidation (Ardic et al., 2009; Cervilla et al., 2007, 2009).
Blamey et al. (1997) concluded that sunflower plants are tolerant to high boron concentrations, but a yield reduction by 25% was recorded with a concentration of 1870 mg kg-1 in the young mature leaf blades (YMB).
Surveying 125 durum wheat (Triticum durum) species, Yau et al. (1997) concluded that durum wheat plants absorb high quantities of boron and have high concentrations in shoots that develop toxicity symptoms but most of the absorbed boron was localized in the blade tips and the yield did not significantly affected.
Shaaban et al. (2004) observed that high boron supplementation to cotton in the presence of higher zinc concentrations dramatically reduced the growth of plant shoots (Fig. 2).
BORON EFFECTS ON HUMAN HEALTH
Recent studies confirmed boron essentiality for animal and human beings. In a study with rats, Kenney and McCoy (2000) concluded that boron works with magnesium and suggested that is may be the reason that boron can be beneficial to persons suffering from hyperthyroidism or persons with thyroid disease who have low magnesium symptoms like rapid heart rate and muscle cramping. They added that, boron lessen the effects of low magnesium in diet on body growth, serum cholesterol and ash concentration in bone. Boron interaction with calcium metabolism or utilization found also to affect bone calcification and maintenance (Nielsen, 1998).
In a study with older men and women, Penland (1994) indicated that boron deficiency affect perception and short term memory and suggested that boron playing a role in human brain function.
Upon their results from an experiment on males, Naghii and Samman (1997) found that boron supplementation increased estrogen and testosterone levels and suggested that the effect of boron was on thyroid, since the hypers often have high progesterone levels and low testosterone levels.
There is emerging evidence that boron is one of several dietary factors important in insulin and glucose metabolism (Hunt, 2004). He found that boron deprivation perturbs energy substrate metabolism in humans and animal models, particularly when other nutrients were provided in suboptimal amounts.
Good intakes of born for human consumption were found with diets abundant in B content such as fruits, vegetables, legumes and nuts and the safe boron intake dose for adults was reported in the range of 1.0 to 3.0 mg day-1 (Nielsen, 1998). However, (Trumbo et al., 2001) of the USA stated that no recommended dietary allowance (RDA) has yet been established for boron and the average dietary intakes are thought to range between 0.87 and 1.35 mg per day.
Boron can become toxic when overdosed (Ziegler and Filer, 1996). Signs of acute boron toxicity in humans include nausea, vomiting, diarrhea, dermatitis and lethargy (Linden et al., 1986). High boron intake can also cause increased urinary excretion of riboflavin (Pinto et al., 1978).
Boron is one of the essential micronutrients for plants. Its essentiality to vascular plants, diatoms and some marine algae and nitrogen fixing cyanobacteria is evident. However, boron physiological role in pants is unclear since still there is no marker shows its behavior in plant tissues. Boron minerals in nature are ulexite (hydrated sodium-magnesium borate), Colemanite (hydrated calcium borate), borax (hydrated sodium borate) or boric acid. Boron concentration ranged between 10-20 mg kg-1 in rocks, 1-10 mg L-1 in seawater and 0.003-0.03 mg L-1 in river water, while ranged in soil between 5-100 mg B kg-1. From the total B in soil, only about 10% is available to plants, however, plant tissues contain 10-100 mg B kg-1 D wt. or more (in case of excess) Boron is taken up by the plant roots in the form of boric acid or borate. Most of the absorbed boron found to localize in the cell wall and the extension zone and tips of the root hairs. Soil pH, texture, temperature, moisture, calcium carbonate and organic matter content are the major factors affecting boron availability. Plant species differ markedly in their boron requirements. Graminaceous species found to be of low B-requirements, non-grass monocots. are mild, while the latex containing species are of high boron demand.
Postulated boron functions in the plants are its incorporation in: (1) Cell wall synthesis and structure, (2) Cell membranes, (3) Metabolism of nucleic acids and protein synthesis, (4) Carbohydrate metabolism and transport, (5) Metabolism of plant growth regulators and (6) Phenol metabolism.
Due to boron deficiency, visual symptoms are developed. Vegetative symptoms are: death of terminal shoots, yellowing and red veins on the terminal shoot leaves, short internodes, small stiff thick brittle leaves with smooth margins, death of small areas in the bark and tip of the shoots followed by progressive death of the inner bark and cambium of the trees. Boron deficiency symptoms on the fruits are: small flatten or misshapen fruits, drought spot, internal cork, cracking and russet in apple, premature ripening, increased fruit drip and low seed count.
Studies showed that boron deficiency inhibits root growth, reduce shoot growth and decrease nutrient uptake and concentrations in the plant tissues. Thus, boron deficiency found (for example) to cause flower and pod abortion in chickpea, male sterility in wheat, seed yield reduction in sunflower. Adding of boron fertilizers to boron deficient plants could significantly increase crop yields.
Excess boron caused what is called boron toxicity. Some plant species found to be severely affected with high boron concentration, while others show a moderate sensitivity and some of plant species are tolerant.
Boron deficiency also found to affect physiology of human beings. Affecting thyroid function, boron deficiency is suggested to cause hyperthyroidism and lowering estrogen and testosterone blood levels in males. Because of its role in insulin and glucose metabolism. Boron deficiency found to affect perception and short term memory, especially with alder people. To avoid boron deficiency, fruits, vegetables, legumes and nuts are recommended diet. 1.0-3.0 mg day-1 is a safe intake dose for adults. However, B can be toxic when overdosed.