Research Article
Seed Germination Protocols for Ex situ Conservation of Some Hypericum species from Turkey
Department of Agronomy, Faculty of Agriculture, University of Ondokuz Mayis, Samsun, Turkey
The conservation of genetic resources of medicinal plants has been of increasing interest and seed storage is the simplest and most economical method for ex situ conservation of plant genetic resources (Perez-Garcia et al., 2006). However, each species has particular requirements for seed germination as a result of adaptive radiation into patchy and changing environments (Cerabolini et al., 2004). Germination is a critical stage in the life cycle of weeds and crop plants and often controls population dynamics, with major practical implications (Keller and Kollmann, 1999). Generally, seed germination is restricted by different kind of dormancy in many plant species. Plant growth regulators such as GA (gibberellic acid) and IAA (indoleacetic acid) (Hilhorst and Karssen, 1992; Iglesias and Babiano, 1997); chemicals such as sulphuric acid (H2SO4) and hot water treatments (Tomer and Maguire, 1989; Baes et al., 2002) have been recommended to break dormancy and enhance germination.
Hypericum is a large genus of herbs or shrubs which grow in temperate regions of the world (Patocka, 2003). Species belonging to this genus have been used as traditional medicinal plants due to their wound-healing, bactericide, anti-inflammatory, diuretic and sedative properties for the last two hundred years (Dias et al., 2000). There are about 400 species of Hypericum in relatively dry temperate zones of the world. In Turkey, the genus is represented by 89 species of which 43 are endemic (Davis, 1988).
Today, some Hypericum species such as H. perforatum (Kirakosyan et al., 2004), H. brasiliense (Abreu et al., 2003), H. androsaemum (Valentao et al., 2003), H. perfoliatum (Couladis et al., 2001) and H. scabrum (Matsuhisa et al., 2002) have become a valuable commodity to wild-crafters who supply these species for the herbal industry due to their phytomedicinal properties and the market for only H. perforatum has exceeded $570 million worldwide annually (Sirvent et al., 2002). The increased market demand for Hypericum derived products has led to uncontrolled harvesting of plant materials from wild populations. In Turkey, wild-crafters and some companies have collected Hypericum plants excessively for export to herbal industry. As a result, the availability of wild plants from different Hypericum species in Turkish flora is strongly limited. Thus, the present study focuses on the determination of ex situ requirements for seed germination in the four Hypericum species, namely H. orientale L., H. perfoliatum L., H. pruinatum Boiss. and Bal. and H. origanifolium Willd. by performing some pre-soaking treatments to promote germination as an initial step in their conservation.
Brief Description of Plant Material
Hypericum orientale: Stems 7-45 cm, erect or decumbent and sometimes rooting. Leaves 10-40 mm, oblong or elliptic-oblong to oblanceolate or linear. Sepals unequal, narrowly oblong and ovate or broadly elliptic to obovate. Petals 10-18 mm, entire, without black dotes. Capsule 7-14 mm, ovoid to ovoid-cyclindric. Hypericum pruinatum: Stems 15-35 cm, erect or ascending from a rooting and branching base, pruinose. Leaves on main stems 10-35 mm, oblong to elliptic, pruinaose. Inflorescence pyramidal to cyclindric. Sepals broadly oblong to broadly elliptic, rounded, entire or minutely black-glandular-denticulate. Petals 9-14 mm. Capsule 7-10 mm, ovoid. Hypericum perfoliatum: Stems 15-80 cm, erect or decumbent. Leaves 13-60 mm, ovate to triangular-lanceolate or rarely linear, the upper most sometimes black-glandular-ciliate, usually densely pellucid-dotted, with obscure reticulate venation. Sepals oblong, subacute to rounded, densely and irregularly dark-glandular-denticulate. Petals 9-14 mm, with or without black dotes. Capsule 5-6 mm, broadly ovoid, with dorsal vittae and lateral vesicles. Hypericum origanifolium: Stems 5-30 cm, suberect to ascending, branching but not usually rooting at the base. Leaves 5-30 mm, ovate or obovate with intramarginal and sometimes superficial black dots. Sepals narrowly oblong to oblong-spathulate, usually with some superficial black dots. Petals 9-15 mm, with black dots. Capsule 7-12 mm, with dorsal vittae and lateral vesicles (Fig. 1) (Davis, 1988). The four Turkish species of Hypericum grow wild in Black Sea region of Turkey at high altitudes. The plant materials were identified by Dr. Hasan Korkmaz, Department of Biology, University of 19 Mayis, Samsun, Turkey. Voucher specimens were deposited in the herbarium of Ondokuz Mayis University Agricultural Faculty (OMUZF # 55 for H. orientale, OMUZF # 107 for H. pruinatum, OMUZF # 109 for H. origanifolium and OMUZF # 101 for H. perfoliatum).
Seed Collection
The seeds were handpicked from at least ten randomly selected individuals during October of 2005 and stored at 4±2°C in sealed plastic bags until used for germination tests.
Experimental Procedures
The present study was conducted at laboratory of Ondokuz Mayis University, Agricultural Faculty. In preliminary testing, seeds placed in Petri dishes did not germinate effectively under normal laboratory conditions. The pre-soaking treatments used in the study were different GA and H2SO4 doses, hot water and tap water. Before placing the seeds in Petri dishes, they were soaked in 50, 100 or 150 mg L-1 GA; 0.5, 1 or 1.5% H2SO4 solutions, tap water, 40, 50 or 60°C hot water for 30 min.
 | |
| Fig. 1: | H. perfoliatum (a), H. pruinatum (b), H. origanifolium (c) and H. orientale (d) plants at full flowering |
Seeds also were soaked in 150 mg L-1 GA and then 0.5% H2SO4 solutions for 30 min to evaluate double effect of these chemicals (Cirak et al., 2004a). The treated seeds were placed in individual, sterilised Petri dishes containing moisture-retaining paper liners. Paper liners in the Petri dishes were kept moist throughout the germination period. The study was performed under a photoperiod of 18 h light/8 h darkness in growth chambers. Temperature was set at 20°C, recommended temperature for germination in H. brasiliense and H. perforatum seeds (Bertelle et al., 2004). Germination was measured as a percentage, 20 days after the experiment was initiated. The seeds showing radicle emergence were recorded as germinated (Come, 1970).
Data Analysis
The experimental design was a factorial randomized block arrangement with three replications with 100 seeds in each. Germination percentages from the original data were transformed for statistical analysis (arcsine of square root of percent germination x 0.01). The transformed data were analyzed using ANOVA and differences among treatments were tested using the Duncan Multiple Range Test (level of significance p<0.01) (Yurtsever, 1984).
According to the results of variance analysis (Fig. 2), the pre-soaking treatments tested had a significant (p<0.01) effect on germination rates depending on species.
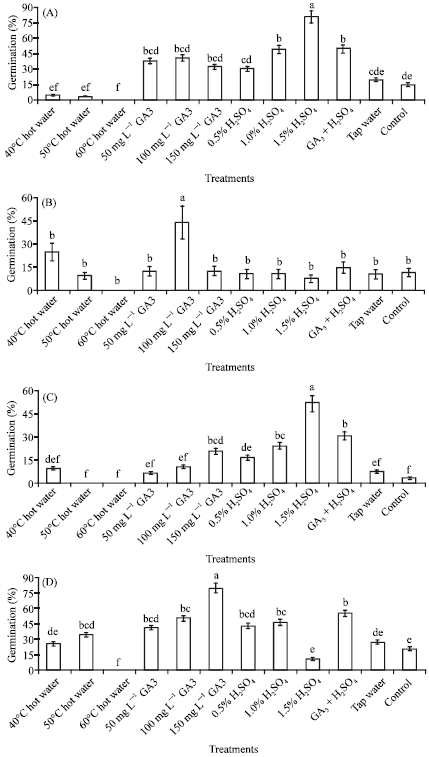 | |
| Fig. 2: | The germination rates of H. orientale (A) H. perfoliatum (B) H. origanifolium (C) and H. pruinatum (D) seeds exposed to different pre-soaking treatments. (Values with different letters within columns for each species differ significantly at the level of p<0.01; bars are±SE) |
In H. orientale and H. origanifolium, H2SO4 treatment was found to be most effective to improve seed germination depending on doses while other treatments were efficient to a lesser degree. The highest germination was induced by H2SO4 1.5% in both species (81 and 52%, respectively), followed by GA + H2SO4 and H2SO4 1% (50 and 49% for H. orientale, 30 and 24% for H. origanifolium, respectively). It is interesting to note that hot water treatments in increasing levels deteriorated germination when compared to control in both species.
GA treatment was the only effective one in promoting germination for H. perfoliatum seeds and GA 100 mg L-1 resulted in the highest germination (43%). The other treatments were all generally ineffective in improving germination and were found in the same statistical group as control.
Germination rates elevated linearly with GA treatment in increasing doses in H. pruinatum seeds. The highest germination was induced by GA 150 mg L-1 (80%), followed by GA + H2SO4 (55%) and GA 100 mg L-1 (50%). On the contrary, germination was suppressed by hot water 60°C and H2SO4 10% treatments.
Light has been recognized since the mid-nineteenth century as a germination-controlling factor and it is frequently found to be a requirement in plant species native to arid lands (Baskin, 2004). In general, absence of light has a negative effect on germination in several Hypericum species such as H. perforatum (Campbell, 1985), H. gramineum (Ash et al., 1998), H. brasiliense (Bertelle et al., 2004) and H. aviculariifolium (Cirak et al., 2007). In a previous study, it was found that 18/6 h light/dark cycle was the most effective to meet light requirement for germination in H. perforatum seeds (Cirak et al., 2004b). Thus, the present study was performed under this photoperiod to supply the probable light requirement.
Seeds of many wild members of different genus have hard seed coats which restrict water absorption by the embryo. Failure to imbibe limits O2 to the embryo and leaching of inhibitors, therein effectively enforcing dormancy of the embryo. For applied uses, dormancy-breaking treatments are required to provide more uniform and rapid seed germination responses. Permeability may be improved by scarifying the seed coat by mechanical means (e.g., clipping, abrasion or immersion in hot water) or chemically with strong oxidative agents (e.g., sulphuric acid or sodium hypochlorite) (Abdallah et al., 1989). In the present study, H2SO4 treatment was found to be most effective to induce seed germination in H. orientale and H. origanifolium. The results indicate the presence of physical dormancy related to hard seed coat and overcame by only acid scarification. The high germination rates obtained with H2SO4 treatments agree with those obtained from other species of Hypericum such as H. lydum and H. tetrapterum (Cirak et al., 2006) as well as some legumes (Teketay, 1996; Grouzis and Danthu, 2001), Prosopis ferox (Baes et al., 2002), Acacia origena, Acacia pilispina and Pterolobium stellatum (Teketay, 1998), Erythrina brucei and Erythrina burana (Teketay, 1994).
Studies of genetics and physiology have shown the important roles of the plant hormones abscisic acid and gibberellin in the regulation of dormancy and germination (Koornneef et al., 2002). Gibberellins comprise the class of hormones most directly implicated in the control and promotion of seed germination. Endogenously applied gibberellins can relieve certain types of dormancy, including physiological dormancy, photodormancy and thermodormancy acting as a substitute for low temperatures, long days, or red light (Seiller, 1998). In this study, GA increased germination rate significantly, depending on concentration in H. perfoliatum and H. pruinatum indicating the presence of physiological dormancy related to partially dormant embryo. Similar results were obtained from studies carried out on other species, such as Sesamum indicum (Kyauk et al., 1995), Rumex dentatus (Ali and Helal, 1996), Zea mays and Glycine max (Wang et al., 1996), Opuntia tomentosa (Carrillo et al., 2003) and Physoplexis comosa (Cerabolini et al., 2004).
Hot water treatments have been reported to enhance germination of hard coated seeds by elevating water and O2 permeability of the testa (Teketay, 1998; Aydin and Uzun, 2001). In present case, hot water treatment in the lowest level induced germination in H. perfoliatum, H. origanifolium and H. pruinatum while it was not effective in promoting germination in H. orientale. The negative effect of the thermal pretreatments on H. orientale seeds was probably due to the combination of high temperature and time, which may cause a damage to the embryo tissue as observed in several species (Khosh-Khui and Bassiri, 1976; Masamba, 1994).
Chemicals that accumulate in the fruit and seed-coat during development and remain in the seed after harvest can act as germination inhibitors. Some of the substances associated with inhibition are various phenols, coumarin and abscisic acid which can be leached out by soaking in water (Booth and Sowa, 2001). In case of H. perforatum and H. aviculariifolium, soaking the seeds in tap water resulted in a significant increase in germination (Cirak et al., 2004c and 2007). In the present study, similarly, tap water treatment slightly increased germination rates in H. orientale and H. pruinatum seeds, but this increase was found to be insignificant.
Seed germination behavior is an integral part of ex situ conservation, especially for developing standard viability monitoring protocols and to ensure sufficient populations for germplasm regeneration. However, germination requirements for native species are often unknown. Here, it is the first time we have described the seed germination requirements of H. orientale, H. perfoliatum, H. origanifolium and H. pruinatum, which are under threat due to uncontrolled harvesting together with other species of Hypericum from Turkish flora. According to the results, propagation from seed is a viable method for the ex situ conservation of these species. However, it should be noted that seed storage and germination are only the first steps in the reinforcement of populations of these species. Further studies based on field experiments to attain baseline data on ex situ plant development and establishment are currently underway.
Author is grateful to Dr. Dursun Ali Tokel, Ondouz Mayis University, Faculty of Science and Art, for his valuable comments and technical assistance.