ABSTRACT
What cause the mitosis (without asters) and cytokinesis for normal higher plant cells is a question that has never been answered quantitatively and theoretically according to our knowledge. In this study, based on published experimental data, Newton`s laws and Coulomb`s law, we develop novel theoretical models of natural and normal mitosis (without asters) and cytokinesis for higher plant cells in M phase, to dynamically explicit several movements of biological objects (chromosomes, the spindle poles and microtubules). We hypothesize for wild types of higher plant cells: 1, No naturally and normally replicated chromosomes can occupy the same nucleus, without growing sizes of the nucleus and the cell (Chromosomes Exclusion); 2, No normally and naturally doubled nuclei can occupy the same cell, if the doubled size of the nuclei is not far smaller than the size of the cell (Nuclei Exclusion); 3, The spontaneous and strong repulsive electromagnetic field forces are the primary cause for the exclusions.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ajpp.2006.21.27
URL: https://scialert.net/abstract/?doi=ajpp.2006.21.27
INTRODUCTION
Biological cell divisions were found about 130 years ago. During the 1870s, A. Schneider observed and described eukaryotic mitosis and cytokinesis (Dowben, 1971). Since then, many experimental data have been published in a process of mitosis and cytokinesis for higher plant cells (Alberts et al., 2002). Recently, more experiments are involved in activations or roles of kinases MAPKs, CDKs or the preprophase band (PPB) in mitosis and cytokinesis (Bögre et al., 1999; Corellou et al., 2001; Chan et al., 2005); formations of the cell plate, differentiation, regulation, degradation and inhibition during cytokinesis (Lee et al., 2001; Boudolf et al., 2004; Patel et al., 2004; Culligan et al., 2004; Weingartner et al., 2004; Weinl et al., 2005). A few models or assumptions have been proposed to explain mechanisms of rearrangements of microtubules during preprophase band and spindle formation (Dhonukshe and Gadella, 2003), mechanisms of chromosome dynamics in mitosis and meiosis (Zoller et al., 2004), or mechanisms of AtE2Fa controls in cell division and plant development (He et al., 2004). However, those models do not provide either a force kind or a force source that separate the two sets of chromosomes and cleave a eukaryote in a view of physics.
Recently, we theoretically proposed and developed several physics models to explain the driving forces during mitosis and cytokinesis for animal cells (Cheng and Zou, 2004) and during amitosis for bacteria cells (Cheng and Zou, 2005). In this study, based on the concepts in our recent models, the published experimental data in literatures, Newton’s laws and Coulomb’s law, we propose and develop our theoretical models for higher plant cells, to depict natural and normal proliferation and movements of discrete chromosome rings and spindle poles during mitosis and cytokinesis.
MATERIALS AND METHODS
To establish our models of mitosis and cytokinesis for higher plant cells, we use concepts of a discrete chromosome ring (DCR) in a division plane and a Meridian Plane (MP) in a three dimensional (3-D) cylindrical coordinate system (CCS). The MP (r, z) passes z axis in a CCS (r, θ, z) and in a laboratory reference because we focus on the objects’ movements in r and z directions. We fix the nuclear center on the z axis for a convenient analysis (Fig. 1 and 2) without loosing general meaning. Variables are functions of r, θ, z and time t and integrals are vectorial, in all equations of this study (Cheng and Zou, 2004, 2005). Based on the concepts, the published experimental data (Alberts et al., 2002), Newton’s laws and Coulomb’s law (Edminister, 1979; Meggs, 1988), we propose and develop our theoretical models of mitosis and cytokinesis without asters in our institute of science research in 2005.
Results (Model Development)
We approximately consider the protoplasm as electrolyte with an uneven charge distribution; the electromagnetic field (EMF) forces are weak in the protoplasm at G1 interphase of a cell cycle. The EMF is actually a quasi-static electric field (EF) because it is mostly electric and slowly variant. According to the experimental data (Alberts et al., 2002), microtubules are roughly evenly distributed in the cell cortex (Fig. 1A) at G1 interphase. We model this experimental result as that the even distribution of the microtubules is because of the same distribution of the electric charge on each of the microtubules. At and after S interphase, chromosomes replication provokes a spontaneous and strong quasi-static EF in protoplasm and cortex. We assume a nucleus or a chromosome has a net negative charge. EF lines directions (the arrows point to) are reversed if a net positive charge is found there. This modification will not affect our electric analysis in principle. The EF through nuclear envelope pores is stronger than that through other locations. The stronger the EF, the more microtubules grow. Microtubules self assemble at the negative growing ends. We also assume the microtubules usually have net positive charges (including some bound ions, e.g., Ca++, or other materials), net positive charges at positive growing ends and negative charges at negative growing ends. The EF lines are plotted in Fig. 1B. The EF forces in z direction make the microtubule band; the EF forces in r direction drive the microtubules into the cytoplasm and make the band disappear. The cell wall is rigid, therefore no contraction occurs as that in an animal cytokinesis.
We consider the nucleus and two complexes of the assembled microtubule ends (-) as wholes (roughly spheres), respectively, to develop our models conveniently (a case with multiple complexes can be derived in the same way).
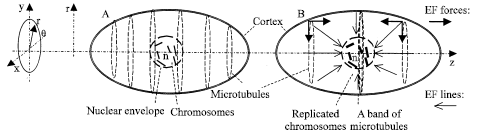 | |
| Fig. 1: | Diagrams in MP. A. EF forces are weak at G1 interface. B. EF. forces are strong at preprophase |
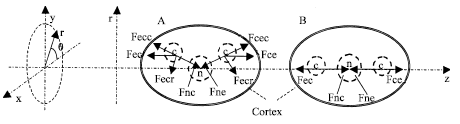 | |
| Fig. 2: | Forces in MP. n is the charge center of a cell nucleus; c and e are two charge centers of two microtubule ends |
In Fig. 2, Fce and Fec are EF forces between the two complexes and they must be repulsive because they have the same polarity of electrical charges. At a moment, the vector forces are estimated with Coulomb’s law:
| (1) |
Where, Qc and Qe are net charges of the two complexes, respectively. d is the distance between charge centers of the two complexes and ace0 is a unit vector, Fcer and Fcec are force components of rotation and centrifugation about the nucleus, respectively. Because, centrifugal and centripetal (between the complexes and the nuclei) forces are balanced from each other, the net forces on the two complexes, respectively are (Cheng and Zou, 2004):
| (2) |
1st items are the rotation forces. fc and fe (vector sums) are resistances including frictions that are always against the movements. mc and me are masses and vc and ve are velocities. The complexes will rotate around the nucleus (Fig. 2A) when |Fecr| > |fc| or |Fcer| > |fe|. The three center points will be on one line (Fig. 2B) when |Fecr| = |Fcer| = 0. We consider this line is on z axis for a convenient illustration of our models. At the temporary equilibrium state, the total energy of the complexes and the nucleus in the protoplasm must fit the principle of minimum energy state. On the other hand, centrifugal and centripetal forces could split the nuclear membrane when they are stronger than the combination forces of the membrane.
At a moment, the vector sum of forces at replicated chromosomes with a net charge Qa is:
| (3) |
Where Fca and Fea are Hooke’s forces toward to points c and e, respectively, pulled by the related kinetochores microtubules; fa (a vector sum) is the resistance including friction; 4th and 5th items are Coulomb’s forces from complexes c and e, respectively. 6th item is a Coulomb’s force from other replicated chromosomes (including + ends of microtubules); 7th item is a Coulomb’s force from microtubules; ma and va are mass and velocity, respectively (Fig. 3A).
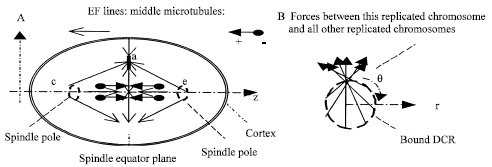 | |
| Fig. 3: | At metaphase: A. EF lines in a MP. Point a is a net charge center of a replicated chromosomes. c and e are net charge centers of the two sets of spindle poles and they are on the z axis. Microtubules generally and roughly grow along the stronger EF lines when they are available. B. Forces acted on a sister chromatid in bound DCR |
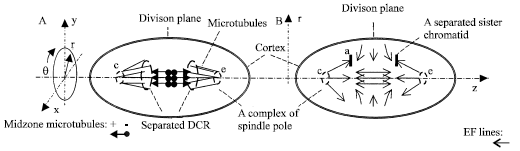 | |
| Fig. 4: | Diagrams of mitosis and cytokinesis at anaphase: A. 3-D plot of the separated DCR, the microtubules and the complexes; B. The EF lines, the forces in a MP |
In the longitude z direction, z components of the EF forces, are ruling forces to determine the position of a spindle equator plane. Ideally, at metaphase, all replicated chromosomes are in the equator plane.
In the transverse plane (Fig. 3B), 6th item in Eq. 3 plays a very interesting role. The vector components at rotation direction are roughly canceled from each other. The radial force component is balanced by the radial components of other items in the equation.
Figure 4A shows a 3-D diagram of charged objects that are roughly symmetric about z axis and the spindle equator plane at anaphase. Net charges of twin sister chromatids, the two sets of complexes and microtubules in the two sides must have the same electric attributes, respectively. Sums of the forces from the two sides of the division plane must be repulsive and can separate the two sides of charged objects.
Figure 4B shows the EF lines about Fig. 4A. The EF distribution is approximately symmetric about z axis and division plane. z components of the EF forces are 0 in the division plane. Also use we Eq. 3 to estimate the vector sum of forces on a separated sister chromatid with charge Qa, but Fea = 0. The forces drive the chromatid to its linked complex. The EF forces mostly guide and transport free negatively charged objects to the middle zone of the phragmoplast and free positively charged objects to nuclei, respectively. The electrical transportation continues at telophase and cytokinesis until a cell wall is formed (Fig. 5).
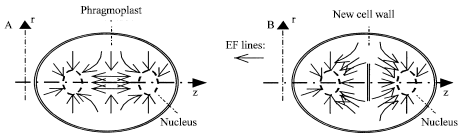 | |
| Fig. 5: | The EF lines at telophase (A) and cytokinesis (B) in MP. |
At telophase, the nuclear envelopes are re-formed around the separated chromosomes because of the electric polarization. After the two nuclei are separated from each other by the new cell wall, the spontaneous and strong repulsive EMF disappears in the protoplasm.
DISCUSSION
In this study, we propose and develop innovational theoretical models using Newton’s laws and Coulomb’s law: equations and diagrams that depict quantitatively EF driving forces and movements of objects during mitosis and cytokinesis for higher plant cells.
Equation 1 and 2 and Fig. 2 depict forces and movements of the two spindle poles. Fce and Fec in Eq. 1 are EF forces between the two charged complexes during mitosis and must be repulsive because they have the same electric attributes. We believe the EF forces are prevailing driving forces to cause the departure of the two spindle poles from each other. The EF forces could be also involved in splitting the nuclear membrane.
Equation 3 and Fig. 3 and 4 describe what forces act at the replicated chromosomes and how the forces drive the chromosomes to move. Because the EF driving forces are repulsive between two sets of chromosomes, we believe that No naturally and normally replicated chromosomes can occupy the same nucleus, without growing sizes of the nucleus and the cell. We assume this phenomenon as Chromosomes Exclusion.
Figure 5 elucidates the EF driving forces are repulsive between two replicated nucleus. We believe that No naturally and normally doubled nuclei can occupy the same cell, if the doubled size of the nuclei is not far smaller than the size of the cell. We assume this phenomenon as nuclei exclusion.
The results in this study and our recent studies (Cheng and Zou, 2004; 2005) are supported from each other in principle. Also support our results the universal Newton’s laws and Coulomb’s law in classical studies.
The real world EF could be modified experimentally and can be alternated environmentally. The weak EF forces can only guide and transport small free charged objects, such as Ca++. We can directly calculate EF and the corresponding forces as long as we know the related data, such as charge distribution and permittivity. Therefore, the theoretical models could be tested in future by measuring the high accurate EMF or EF.
One of the limitations of the models is to obtain the distributions of electric charges and permittivities in real world environments today. Other drawbacks of the methods could be errors of neglecting magnetic field MF effect, because there must be some magnetic fields according Maxwell’s equation or Ampere’s law.
However, we believe our models provide both a kind and a source of the forces that drive the discrete chromosome rings to separate from each other and that divide a higher plant cell into two cells, explicit mechanisms of the movements of the charged objects in a view of physics, are also in principle applied to, meiosis as well as mitosis and cytokinesis of multiple nuclei or mutant chromosomes, for higher plant cells.
In this study, to our knowledge, we are the first to use Newton’s laws and Coulomb’s law to quantitatively describe the proliferation and the several movements of biological objects (chromosomes, the spindle poles and microtubules) during natural and normal mitosis (without asters) and cytokinesis for higher plant cells. We propose and hypothesize: 1, No naturally and normally replicated chromosomes can occupy the same nucleus, without growing sizes of the nucleus and the cell (Chromosomes Exclusion); 2, No normally and naturally doubled nuclei can occupy the same cell, if the doubled size of the nuclei is not far smaller than the size of the cell (Nuclei Exclusion); 3, The spontaneous and strong repulsive electromagnetic field forces are the primary cause for the exclusions.
ACKNOWLEDGMENTS
We greatly appreciate that Dr. Cheng, Yaoting and Mrs. Li, Qiong Qiu; Mr. Zou, Zi Jia and Mrs. Zheng, Chao Ying for financial support for this study; Miss Vivien Cheng for helpful suggestions, comments and english correction for this publication.
REFERENCES
- Bogre, L., O. Calderini, P. Binarova, M. Mattauch and S. Till et al., 1999. A map kinase is activated late in plant mitosis and becomes localized to the plane of cell division. Plant Cell, 11: 101-114.
Direct Link - Boudolf, V., B. Rosa, A.E. Janice de, V. Aurine and B. Tom et al., 2004. B1-Type cyclin-dependent kinases are essential for the formation of stomatal complexes in Arabidopsis thaliana. Plant Cell, 16: 945-955.
Direct Link - Chan, J., G. Calder, S. Fox and C. Lloyd, 2005. Localization of the microtubule end binding protein EB1 reveals alternative pathways of spindle development in arabidopsis suspension cells. Plant Cell, 17: 1737-1748.
CrossRefDirect Link - Cheng, K. and C. Zou, 2004. 3-D physical models of mitosis (with asters) and cytokinesis. Biomed. Sci. Instrum., 39: 413-418.
Direct Link - Cheng, K. and C. Zou, 2005. 3-D physical models of amitosis (cytokinesis). Med. Hypoth., 64: 88-91.
CrossRefDirect Link - Corellou, F., C. Brownlee, L. Detivaud, B. Kloareg and F.Y. Bouget, 2001. Cell cycle in the fucus zygote parallels a somatic cell cycle but displays a unique translational regulation of cyclin-dependent kinases. Plant Cell, 13: 585-598.
Direct Link - Culligan, K., A. Tissier and A. Britt, 2004. ATR regulates a G2-phase cell-cycle checkpoint in Arabidopsis thaliana. Plant Cell, 16: 1091-1104.
CrossRefDirect Link - Dhonukshe, P. and T.W.J. Jr. Gadella, 2003. Alteration of microtubule dynamic instability during preprophase band formation revealed by yellow fluorescent protein-CLIP170 microtubule plus-end labeling. Plant Cell, 15: 597-611.
CrossRefDirect Link - He, S.S., J. Liu, Z. Xie, D. O'Neill and S. Dotson, 2004. Arabidopsis E2Fa plays a bimodal role in regulating cell division and cell growth. Plant Mol. Biol., 56: 171-184.
Direct Link - Lee, Y.R.J., H.M. Giang and B. Liu, 2001. A novel plant kinesin-related protein specifically associates with the phragmoplast organelles. Plant Cell, 13: 2427-2439.
PubMedDirect Link - Meggs, W.J., 1988. Electric fields determine the spatial organization of microtubules and actin filaments. Medical Hypotheses, 26: 165-170.
PubMedDirect Link - Patel, S., A. Rose, T. Meulia, R. Dixit, R.J. Cyr and I. Meier, 2004. Arabidopsis WPP-domain proteins are developmentally associated with the nuclear envelope and promote cell division. Plant Cell, 16: 3260-3273.
CrossRefDirect Link - Weingartner, M., C. Marie-Claire, M. Tamas, B. Pavla and S. Anne-Catherine et al., 2004. Expression of a nondegradable cyclin B1 affects plant development and leads to endomitosis by inhibiting the formation of a phragmoplast. Plant Cell, 16: 643-657.
Direct Link - Weinl, C., A.S. Marquardt, J.H.K. Suzanne, K.N. Moritz and J.J. Marc et al., 2005. Novel functions of plant cyclin-dependent kinase inhibitors, ICK1/KRP1, can act non-cell-autonomously and inhibit entry into mitosis. Plant Cell, 17: 1704-1722.
Direct Link - Zoller, J.F., R.G. Herrmann and G. Wanner, 2004. Chromosome condensation in mitosis and meiosis of rye (Secale cereale L.). Cytogenet Genome Res., 105: 134-144.
CrossRefDirect Link








