ABSTRACT
Abiotic stress conditions such as drought, salt and high temperature reduce yields of crop plants by as much as 70%. Therefore, advancement of salt-tolerance in cotton requires the identification of plant genetic diversity that exists, theoretically, in cultivars, which are adapted to arid growing regions. The exotic race stock accessions should possess salt-tolerant genes since many of these Gossypium hirsutum germlines were collected in the arid regions of Mexico and Central America. The objective of this project was to develop a screening technique to identify the cultivars and exotic germlines that possessed salt-tolerant genes. Since the early stages of a plant`s life cycle are generally believed to be the most sensitive stages, we screened several conventional cultivars as controls and several exotic race stock accessions for their ability to tolerate increased salt levels during seed germination-radical elongation (growth chamber) and seedling maturation (hydroponic conditions in a greenhouse). It was concluded that this screening technique resulted in the identification of plants that survived these conditions and represent likely candidates for use in our plant-breeding program.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajpp.2006.107.112
URL: https://scialert.net/abstract/?doi=ajpp.2006.107.112
INTRODUCTION
Salinity is an on-going problem in many regions of the world where irrigated is necessary to sustain efficient agricultural production. Such situations highlight the need for salt-tolerant crop cultivars (Flowers and Yeo, 1995). Extensive research has been published on the effects and response mechanisms of salinity in plants (Hasegawa et al., 2000; Pessarakli, 2002; Munns and James, 2003) and genetic diversity for salt-tolerance as reported by Munns et al. (2002). Plants that can grow in saline environments are called halophytes; however, most crop species are salt-sensitive or glycophytes (Flowers et al., 1986). The primary effects of high levels of salts cause hyperosmotic stress and ion imbalance in plants; secondary effects involve oxidative damage to enzymatic proteins and membrane integrity (Zhu, 2001; Xiong and Zhu, 2002). In general, halophytic plants adapt to salt stress by avoidance (preventing entry or possessing excretion mechanisms); compartmentalization of salt ions by their movement into the vacuole, salt glands or trichomes and de-toxification of Reactive Oxygen Species (ROS) by osmolytes and anti-oxidant enzymes (Flowers et al., 1986; Zhu, 2001). Most glycophytes lack or possess these mechanisms to a minimal degree (Munns, 2002; Munns et al., 2002). During the last decade, the interactions of these whole-plant, cellular and molecular mechanisms that are involved with the regulation of salt-tolerance has been a primary focal point in many laboratories (Hasegawa et al., 2000; Zhu, 2001; Munns, 2002; Munns et al., 2002; Xiong and Zhu, 2002; Ashraf, 2002; Munns and James, 2003).
In general, the stages of a plant’s life cycle that are most sensitive to salinity stress are seed germination and early seedling development. Theoretically, this sensitivity could be due, in part, to the presence of a high salinity levels found within the germination zone of soils, which is created by the accumulation of soluble salts as a result of evaporation and capillary rise of ground water. Therefore, screening of genotypes or native, heterogeneous lines at these early stages of development for salt-tolerance is advantageous to a plant-breeding program. Generally, cotton is considered to be moderately tolerant to salinity and according to Soltanpour and Follett (1995) is ranked second behind barley for salt-tolerance. However, under salt stress conditions, cotton’s yield production is reduced drastically due to poor seed germination (Khan, 1987; Ahmad et al., 1995). In comparison, plants that were salt stressed during their seedling stage produced less than maturing cotton plants that were salt stressed during their vegetative, flowering and boll formation stages (Khan et al., 1995). Improvement in cotton salt-tolerance has been demonstrated with only a one-cycle selection procedure using 12 day-old seedlings that were subjected to 210 mM NaCl and maintained for five weeks under field conditions (Ashraf and Ahmad, 1999).
In 1999, Nasirci and Smith screened 79 phenotypes that were part of the exotic “race stock” accessions found in the US Cotton Collection. Many of these lines were shown to have diverse genetic characteristics (Nasirci and Smith, 1999). In our companion study (Basal et al., 2006) we have described the affects of salt-stress on several of these unique phenotypes. The selected phenotypes and corresponding controls were subjected to seed germination-radical elongation (growth chamber) and seedling development (greenhouse) under increased NaCl stress levels. In this study, we have described the technical procedure in more detail and made reference to salt interchangeably with Na.
Materials and Methods
General Procedure For Seed Germination and Radical Elongation (SG-RE) Under salt-stress Conditions
Seeds (50 per line/treatment) were treated with a fungicide (Delta- Coat ADTM, Wilbur-Ellis, San Francisco, CA, USA) at 50% (v/v) and placed on germination paper (13 1/2x19 cm) that had been previously soaked in either 0, 75 or 150 mM NaCl. Seeds were oriented in a single line about 7 cm from the bottom edge of the germination paper and a second germination paper, which was also soaked in the same treatment was then placed over the seeds. The germination package was rolled within waxed paper (13 1/2x19 cm) and secured with a rubber band. Each germination package was labeled according to genotype and treatment on tape placed on the outside of the waxed paper for easy tracking. The germination packages treated with 0 mM NaCl were placed upright in 2 L beakers with 3 cm of RO water. The germination packages treated with 75 mM NaCl were placed upright in 2 L beakers with 3 cm of 75 mM NaCl and likewise for the 150 mM NaCl treatment. RO water was added morning and afternoon as needed to maintain the treatment concentration as affected by water evaporation. A large tray containing RO water was placed on one of the shelves inside of a growth chamber (Conviron, Inc., Winnipeg, Manitoba, Canada) to help maintain a high level of humidity (59-66%). Air temperatures were regulated at 29°C for daytime and 19°C for nighttime. Light intensity was regulated at 550 μmm-2sec-1 with a combination of fluorescent and incandescent light bank and a photoperiod of 16 h light and 8 h dark. Pilot studies demonstrated that the 75 mM and 150 mM NaCl levels provided an adequate range for seed germination of DPL-50 (Calhoun et al., 1997), the putative salt-sensitive cotton cultivar used as a control. Preliminary data also indicated that surface sterilization (20% Clorox plus a few drops of Tween -20 for 20 min followed by 3 washing steps with sterile Millipore water) of seeds before seed germination negated the salinity affect of both salt levels. After 5 days, each packet was opened and the percentage of seed germination of each line per treatment was determined (data not shown). A seed was considered to have germinated, if it had a radicle length that was equal to or greater than 0.5 cm.
| Table 1: | Guinn’s nutrient solutions |
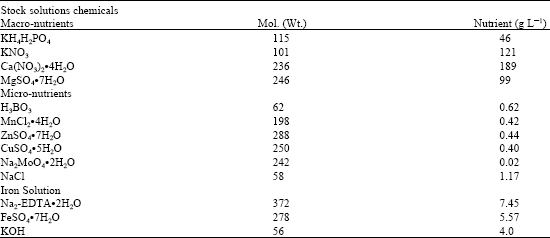 | |
Germinating seedlings (≥18) that possessed a radical length of 2-3 cm or greater were selected for the hydroponic phase using increased levels of salt (0, 150 and 200 mM NaCl) and modified Guinn’s Nutrient Solutions (Table 1) (Guinn, 1974). These two levels of NaCl were selected because previous studies indicated that 150 mM NaCl was the minimum salt concentration that produced significant differences in growth reduction between the more salt tolerant and less salt tolerant cotton cultivars (Gossett et al., 1992).
General Procedures For Seedling Development Under Increased Salt-stressed Hydroponic Conditions
Air stones, tubes and tanks were rinsed (2X) with RO water. Pumps were attached to valve adjusters by tubing; then connected by tubing to two air stones (13 and 25 cm) per tank. RO water (8 L) was placed in each tank (45x33x12 cm) and then the pumps and the aeration system were checked (water discarded). Next, 16 L of RO water were added to each tank, followed by a modification (Table 1) of Guinn’s nutrient solutions (Guinn, 1974). The pH of each tank was adjusted to approximately 6.3 with 0.14 N KOH and/or 0.1 N H2SO4 (checked with pH strips). After the initial set-up, the acid or base aliquot was added in 100 mL RO water to the tank. The level of nutrients/RO water was marked on each tray and adjusted each day.
Next, seedlings that were germinated under increased levels of salt were placed in one of the corresponding salt-stressed hydroponic plastic seedling trays (60x40x2/3 cm with 18 holes/line with 5 lines/tray). Cut, disposable pipet tips (1000 μL) were used to hold the seedlings in place within the plastic seedling trays. Care was taken to prevent light penetration into the nutrient solutions to limit algal growth in the hydroponic tanks. Air temperatures were regulated at 24 to 27°C for day and 21 to 24°C for night. Light intensity was regulated with a combination of GE High-Pressure Sodium LightsTM (1000 watts) and natural daylight at a range of 480 to 1422 μmm-2sec-1. The photoperiod of 16 h light and 8 h dark condition was constant; however, for the 2nd experiment (May-June, 2003), the GE High-Pressure Sodium LightsTM (1000 watts) were turned off. The hydroponic experiments were performed in a greenhouse at the Borlaug Biotechnology Center at Texas A and M University, College Station (Texas). Two experiments are reported here: January-February (2003) for experiment 1 and May-June (2003) for experiment 2.
The selected seedlings, which were arranged in the hydroponic set-up were allowed to acclimate for 72 h in the modified Guinn’s medium only. During this growth period, the seed coats were removed from the cotyledons of the developing seedling. Next, the saline treatments were initiated by adding NaCl at 50 mM aliquots at 12 h intervals starting in the early evening, approximately 132 h post set-up. After the addition of the last NaCl allotment, the next day represented the first day of the hydroponic salt-stressed experiment. Starting with the hydroponic set-up day, data recordings of each tank were taken daily as follows: pH via pH strips, Electrical Conductivity (ECe) and water temperature via ECe Meter (Myron L Company, Carlsbad, CA, USA). ECe is a measure of salinity in units of decisiemens per meter, or dSm-1; high levels of ECe indicate a high level of dissolved nutrients in the water.
For each experiment, the nutrient solution of each salt-stressed hydroponic tank was changed on the 7th day (early morning) of the salt-stress period. The pH was adjusted to approximately 6.3 by adding the appropriate amount of KOH (0.14 N) to the 16 L of RO water in tanks as described earlier. The total amount of NaCl for the respective treatments was added at 1800 h on the same day of nutrient exchange.
In experiment 1, random plants were selected for data analyses and others were transferred to pots in Scott’s Metro-Mix 200 (Scott’s Sierra Horticulture Products, Marysville, OH, USA) (30 cm pots) and then taken to the greenhouse. For experiment 2, survivors were potted in Scott’s Metro-Mix 200 during the early morning and, subsequently, allowed to sit inside the potting room until late afternoon. Those plants that showed turgid characteristics were then taken to the greenhouse. The other potted plants that were smaller and/or still visually stressed were put into a growth chamber. Potted plants were placed in evaporation trays. RO water was added to the trays (two potted plants per tray) to insure that adequate moisture was available to the stressed plants in both the growth chamber and the greenhouse. These plants were checked morning and evening and watered as needed. The greenhouse plants were fertilized every 3-days with Peter’s 20-20-20 (196 mg) (Grace Sierra Hart Products Company, Milpitas, CA, USA). Every 2-weeks, plants were fertilized with a complex mixture (196 mg of Peter’s 20-20-20; 28 mg of Peter’s Professional Trace Minerals (M77) (Grace Sierra Hart Products Company, Milpitas, CA, USA) and 1 cap of rooting hormones (IBA and NAA) (Dip ‘N Grow) (Dip ‘N Grow, Inc., Clackamas, OR, USA)] that was placed into 4 of RO water and applied using an aspirator water-line attachment.
Results
Before the nutrient exchange, the plants for the most part looked good; however, the salt-treated plants grew slower than the plants under control conditions. No signs of physiological stress were evident during this time period. However, after two days following the nutrient exchange, the physiological affects of the increased NaCl levels in our experiments became more evident. For experiment 1 (February, 2003), the plants showed only a growth-stressed response, while in experiment 2 (May-June, 2003) the secondary, vegetative growth portion of the plants were severely stressed. In general, plants were wilted at both 150 and 200 mM NaCl, but only a few plants survived the salinity stress conditions at the 200 mM NaCl level in experiment 2.
DISCUSSION
The technical procedure as described in this report was repeated two times and the phenotypic results are discussed in our companion paper (Basal et al., 2006). A total of 46 plants were transferred to pots from two experiments. Seeds from each plant were harvested and advanced to a plant-breeding program.
This salt screening procedure utilizes dry seeds for seed germination-radical elongation phase (growth chamber) and, then, selected seedlings were used for the seedling development phase (hydroponic cultures in a greenhouse). Using both of these selection techniques, it is evident that different genotypes possessed genetic diversity for salt-tolerance when subjected to this procedure. Differences were evident based on plant productivity, fresh and dry weights and number of survivors. This genetic variation was as expected due to the heterogeneity of the population tested (Nasirci and Smith, 1999). Also, the control genotypes responded as expected based on their phenotype to increased salinity. As described in this study, genetic variation for salt-tolerance of the tested heterogeneous populations was similar as found in hydroponic salt-stressed cultures of different commercial tomato F1 hybrids (Yeo and Flowers, 2002). To strengthen this screening procedure, we anticipate that future screening procedures will be coupled with an additional trait-selection step. This analytical step will be performed on the survivors at the end of this selection process to further identify the mechanisms associated with either osmotic pressure, de-toxification of ROS via anti-oxidants or compartmentalization of Na ions (Munns and James, 2003).
As indicated in our experiments, the number of survivors was also affected by the calendar year. The plants tested in experiment 1 received less ambient temperature fluctuations (21-32°C) than the plants tested in experiment 2 (15.5-48°C). Therefore, the number of plants that survived was greater for the 1st experiment than the 2nd experiment. This light-intensity affect was also demonstrated in salt-stressed wild-type and mutagenized Arabidopsis (EMS) seedlings (Tsugane et al., 1999). The EMS mutant seedlings had a higher survival rate than the wild-type when grown for 14-days under salt-stressed conditions and at two moderate levels (26 and 39 μ mm-2 sec-1) of light intensity. This contrast was not due to proline accumulation in seedling leaves, nor ion exclusion or ion compartmentalization, since there was not any significant difference in the K, Na, Ca and Mg content or the K /Na ratio between the wild-type and mutant seedlings. However, under these moderate light intensity/salt-stressed conditions, the EMS mutant seedlings accumulated higher levels of oxygen-scavenging enzymes (superoxide dismutase and ascorbate peroxidase) than the wild-type seedlings (Tsugane et al., 1999). These results indicate that both salt-stressed conditions and light intensity, in part, influenced the rate of photosynthesis resulting in decreased growth rate. Also, excessive light stress has been shown to cause changes in the efficiency of the photosynthetic electron transport system and the increased production of ROS in plant cells, which, in turn, induces the formation of anti-oxidant enzymes as a plant-response defense mechanism (Karpinski et al., 1997; Fryer et al., 2003).
ACKNOWLEDGMENTS
The authors wish to express their appreciation for the financial supported provided by The Cotton Foundation (2000 – 2004) and The Texas Department of Agriculture (2002-2004).
REFERENCES
- Ashraf, M. and S. Ahmad, 1999. Exploitation of intra-specific genetic variation for improvement of salt (NaCl) tolerance in upland cotton (Gossypium hirsutum L.). Hereditas, 131: 253-256.
Direct Link - Ashraf, M., 2002. Salt tolerance of cotton: Some new advances. Crit. Rev. Plant Sci., 21: 1-30.
CrossRefDirect Link - Basal, H., J.K. Hemphill and C.W. Smith, 2006. Shoot and root characteristics of converted race stocks accessions of upland cotton (Gossypium hirsutum L.) grown under salt stress conditions. Am. J. Plant Physiol., 1: 99-106.
CrossRefDirect Link - Flowers, T.J. and A.R. Yeo, 1995. Breeding for salinity resistance in crop plants: Where next? Aust. J. Plant Physiol., 22: 875-884.
CrossRefDirect Link - Fryer, M.J., L. Ball, K. Oxborough, S. Karpinski, P.H. Mullineaux and N.R. Baker, 2003. Control of Ascorbate Peroxidase 2 expression by hydrogen peroxide and leaf water status during excess light stress reveals a functional organization of Arabidopsis leaves. Plant J., 33: 691-705.
PubMedDirect Link - Guinn, G., 1974. Abscission of cotton floral buds and bolls as influenced by factors affecting photosynthesis and respiration. Crop Sci., 14: 291-293.
Direct Link - Karpinski, S., C. Escobar, B. Karpinska, G. Creissen and P.M. Mullineaux, 1997. Photosynthetic electron transport regulates the expression of cytosolic ascorbate peroxidase genes in Arabidopsis during excess light stress. Plant Cell, 9: 627-640.
PubMedDirect Link - Munns, R., 2002. Comparative physiology of salt and water stress. Plant Cell Environ., 25: 239-250.
CrossRefDirect Link - Munns, R. and R.A. James, 2003. Screening methods for salinity tolerance: A case study with tetraploid wheat. Plant Soil, 253: 201-218.
CrossRef - Munns, R., S. Husain, A.R. Rivelli, T. Condon and A.G. Lindsay et al., 2002. Avenues for increasing salt tolerance of crops and the role of physiologically based selection traits. Plant Soil, 247: 93-105.
CrossRefDirect Link - Tsugane, K., K. Kobayashi, Y. Niwa, Y. Ohba, K. Wada and H. Kobayashi, 1999. A recessive Arabidopsis mutant that grows photoautotrophically under salt stress shows enhanced active oxygen detoxification. Plant Cell, 11: 1195-1206.
Direct Link - Xiong, L. and J.K. Zhu, 2002. Molecular and genetic aspects of plant responses to osmotic stress. Plant Cell Environ., 25: 131-139.
PubMedDirect Link








