ABSTRACT
The effects of supplemental Ca2+ supply and NaCl salinity on the levels of the osmoticants; proline, glycinebetaine and activities of proline metabolism enzymes viz pyrroline-5-carboxylate reductase (P-5-CR), proline oxidase, γ-glutamyl kinase on root and shoot of salt tolerant cv. T-44 were investigated. During germination of seeds of green gram subjected to NaCl salinity stress, proline and glycinebetaine contents in the root and shoot increased continuously. A further increase in proline and glycinebetaine concentration was observed with the addition of 5 mM CaCl2 to 200 mM (maximum salinity stress). Calcium ions play an important role in osmoprotection. The effects of Na+ and Ca2+ are thus additive in causing accumulation of proline and glycinebetaine. Ca appears to confer greater osmoprotection to the seedling exposed to salinity. P-5-CR and γ-glutamyl kinase play an important role in the synthesis of proline, while another enzyme proline oxidase catalyzes the conversion of proline to glutamate, thus reducing the levels of proline. Addition of 5 mM CaCl2 to 200 mM NaCl-stressed plants increased the proline concentration by decreasing the level of proline oxidase and increasing the levels of γ-glutamyl kinase and P-5-CR activities. Salinity stress, in the absence of calcium, increased proline due to decreased proline oxidase activity and increased accumulation of P-5-C reductase and γ-glutamyl kinase activities in both root and shoot of green gram. Thus, calcium ions increase both proline and glycinebetaine production in NaCl stressed green gram seedlings.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajpp.2006.1.12
URL: https://scialert.net/abstract/?doi=ajpp.2006.1.12
INTRODUCTION
Salinity in soil presents a stress condition for crop plants that are predominately sensitive to the presence of high concentration of salt (Dodd and Donvan, 1999; Misra and Dwivedi, 2004). Germination of seeds, one of the most critical phases of plant life, is greatly influenced by salinity. It has been reported that growth as well as metabolism are altered under saline stress (Misra and Dwivedi, 1990, 1995, 2004; Misra and Gupta, 2005). Cellular events such as salt compartmentation and osmotic adjustment in salt-tolerant plants allow continued growth in presence of NaCl (Volkmar and Steppuhn 1998). Salt ions play an important role in osmotic adjustment, in stressed sugar beet plants (Ghoulam et al., 2002) as they were efficiently compartmentalized at the cell level. Osmotic adjustment, at the physiological level, is an adaptive mechanism involved in drought or salinity tolerance (Misra and Gupta, 2005), which permits the maintenance of turgor under conditions of water deficit (Morgan, 1984) commonly proposed mechanism include compartmentation of ions in vacuoles (Yeo, 1998), accumulation of compatible solutes in the cytoplasm (Hare and Van Staden, 1998) and also genetic salt resistance.
Under salt stress, osmotic adjustment is usually achieved by the uptake of inorganic ions from the growth media (Prat and Fathi-Ettai, 1990). Mineral toxicity and nutritional imbalance often accompany the accumulation of ions. Production and accumulation of compatible organic solutes in the cytoplasm obtain osmotic adjustment, in addition to several other cellular and molecular mechanisms. The major compatible solutes include proline and glycinebetaine (Misra and Gupta, 2005, Girija et al., 2002) are thought to function as osmoprotectants for protein (Bohnert and Jenson, 1996). These solutes also provide a protective environment for enzymes and macromolecular structure and function. Glycinebetaine acts as osmotic solutes. It has been reported that glycophytes viz. tomato, peas, beets, oat, beans and carrots showed an increase in glycinebetaine with increasing salinity (Sudhakar et al., 1993). NaCl stress increased the level of glycinebetaine in peanut cotyledons and embryonic axis (Girija et al., 2002). It has also been reported in my previous paper that glycinebetaine accumulation in both root and shoot increased in salt tolerant cultivar in presence of salinity stress (Misra and Gupta, 2005). Different approaches have been used to genetically engineered plants for enhancing stress tolerance in plants by manipulating the level of compatible solute glycinebetaine (Sakamoto and Murata, 2000).
Two enzymes of proline metabolism viz γ-glutamyl kinase and γ-glutamyl phosphate reductase are regarded as an enzyme complex called P5C synthetase because it catalyzes and product glutamine γ-semialdehyde (GSA) is non-enzymatically converted to pyrroline-5-carboxylate. So that, the regulation of proline biosynthesis is mainly controlled by the activity of P-5-C synthase (Boggess et al., 1976). The enzyme proline oxidase also influences the level of proline accumulation as it degrades proline to glutamate. Such reduction in proline oxidase activity concomitant with increase in proline accumulation was also reported in presence of salinity stress in green gram cultivar (Misra and Gupta, 2005).
One of the other ions that play a vital role in mediating plant response to external stimuli of both abiotic and biotic origin is calcium (Snedden and Fromn, 1998). Ca2+ triggers a number of cellular processes that influence growth development and physiology of plants and which in turns allows adaptation of plants to the changing environment. Under salinity stress, an increase in the concentration of Ca2+ can often ameliorate the inhibitory effects on growth (Lauchli, 1990). Addition of Ca2+ in the medium distresses growth inhibition by salt of glycophytic plants (Kinraide, 1998). Ca2+ sustains K+ transport and K+/Na+ selectivity under salt stresses plants (Lauchli, 1990). It has also been known that Ca2+ helps the plants to survive at higher concentration of sodium (Epstein, 1998). The role of Ca2+ in K+ and K+/Na+ selectivity was re-emphasized by the novel discovery of SOS3 gene from Arabidopsis (Liu and Zhu, 1998).
Studies on the combined effects of NaCl and CaCl2 on the accumulation of proline, glycinebetaine and their metabolism are scarce. Therefore, in the present study the hypothesis was that the Ca2+ ions play a key role in control of the accumulation of osmoprotectants, proline and glycinebetaine, as well as in metabolism of proline in germinating salt tolerant green gram in absence and in the presence of NaCl, CaCl2 and their combination.
MATERIALS AND METHODS
Plant Material, Growth, Stress Conditions
Seeds of green gram (Phaseolus aureus, Family Leguminosae) cultivar T-44 (salt tolerant; hybrid between T-1 and T-49) was found to be fairly tolerant to saline stress (Misra and Dwivedi, 2004). The seeds were surfaced sterilized with 1% sodium hypochlorite and germinated as described by Misra and Dwivedi (2004). Seeds were spread over on washed neutral sand covered with filter paper in petri dishes. Seeds were allowed to germinate for 5 days at 30°C ± 1°C in an incubator with a 12 h light/12 h dark photoperiod using uniform quantities of triple- distilled water (control) and respective salt solutions.
The seeds were germinated in four sets (1) distilled water, or (2) 5 mM CaCl2 (3) 200 mM NaCl, or (4) 200 mM NaCl+5 mM CaCl2. Three replicates were maintained for each treatment. Starting with the 4-h soaked seeds (zero h of germination), the germinated seeds were taken out at 24 h intervals up to 5 days, root and shoot (along with cotyledons) were separated from the seeds.
Solute Determination
Proline
Proline accumulation was determined as described by Bates et al., (1973) with slight modification. Powdered frozen (-70°C) tissue (50 mg) both root and shoot were weighed into 1.5 mL centrifuge tubes and then added 3% 1.2 mL of sulphosalicylic acid to precipitate protein. Samples were mixed, centrifuged at 18,000-x g for 10 min and the filtrate transferred to a fresh 1.5 mL tube. Total 500 μL of filtrate and made up 1ml with distilled water. Added 1ml glacial acetic acid and 1 mL ninhydrin reagent [3% (w/v) ninhydrin in 60% (v/v) 6M phosphoric acid] for one hour at 90°C. After cooling of the tubes in ice, the products were extracted with 2 mL of toluene by vortex mixing and the upper (toluene) phase decanted into a glass cuvette and absorbency read at 520 nm. Proline concentrations were calculated using proline standards (0-50 μ mol mL-1) in identical manner.
Glycinebetaine
Glycinebetaine was measured in root and shoot tissue extracts as described by Lever et al., (1992) with some modification. Powered frozen (-700C) tissue (50 mg) both root and shoot were weighed into 1.5 mL centrifuge tubes and then added 60 μL drying reagent [5% (w/v) silver ortho phosphate in anhydrous di sodium hydrogen phosphate] followed by 1 mL extraction solvent [10% (v/v) methanol and 90% (v/v) anhydrous acetonitrile]. Samples were mixed for 20 min., centrifuged at 18,000-x g for 10 min. and then filtrate was transferred to a fresh tube, 400 μL supernatant was taken and added 20 μL 10%(w/v) magnesium oxide. After mixing 50 μL of derivatizing agent (4-isomophenyl triflate) was added. Mixture was centrifuged for 5 min 18,000 x g and aliquots (10 μL) of the supernatant injected directly into a 100 x 48 mm silica column and eluted with a solvent phase of 1.8 mM citric acid in 40 mM triethanolamine (15%) and isopropanol (80%) through HPLC. Glycinebetaine peaks in each extract were identified by authentic glycinebetaine standard curve.
Enzyme Extraction and Enzyme Assays
A crude enzyme extract was prepared by homogenizing 500 mg tissue (root and shoot (along with cotyledons) in 0.1M Tris HCl buffer, pH 7.5, at 4°C. The homogenate was centrifuged at 30,000-x g for 30 min. the supernatant was used as the crude extract enzyme preparation for pyrroline-5-carboxylate reductase activity and the pellet was used as the crude enzyme preparation for proline oxidase and γ-glutamyl kinase activity. For γ-glutamyl kinase enzyme activity the whole extract were kept in freezer at –200C (Misra and Gupta, 2005).
Pyrroline-5-carboxylate reductase was assayed according to Rena and Splittstoesser (1975) with a slight modification. The assay mixture of 1.0 ml contained 100 mole L-1 L-pyrroline-5-carboxylate and 100 mM sodium phosphate buffer, pH 7.5. The decrease in absorbance at 340 nm was measured.
Proline oxidase activity was determined according to Huang and Cavalieri (1979) with a slight modification. The pellet was mixed with 1 mL Tricine, KOH buffer (pH 7.5) containing 6 M sucrose. This extract was used for assay. The assay mixture contained 1.2 mL 50 mM Tris HCl buffer (pH 8.5), 1.2 mL 5 mM MgCl2, 0.1ml 0.5 mM NADP, 0.1 mL 1 mM KCN, 0.1 mL 1 μ phenazine methosulfate (PSM), 0.1 mL 0.06 mM 2,6 dichlorophenol indophenol (DCPIP) and 0.1 mL 0.1M proline in a final volume of 3 mL. The increase in absorbance was recorded at 600 nm at r.t. using proline to initiate the reaction. The rate of reduction of DCPIP was used to determine the enzyme activity expressed as mM DCPIP reduced min-1 mg-1 protein.
γ-glutamyl kinase [ATP: L- glutamate 5- phosphotrasferase [EC 2.7.2.11] activity was assayed by the method of Hayzer and Leisinger (1980) with slight modification. The frozen sample was suspended in 10 mL 0.1 M Tris-HCl buffer containing 1mM 1,4-dithiothreitol (DTT) to rupture the cell and centrifuged at 30,000-x g for 30 min. The crude extract (3.0 mL) was desalted on a Pharmacia PD-10 (sephadex G-24) column equilibrated with 50 mM Tris HCl buffer, pH 7.5 containing 1mM DTT. The assay mixture contained 50 mM L-glutamate, 10 mM ATP, 20 mM MgCl2, 100 mM hydroxylamine HCl and 50 mM Tris HCl, pH 7.0 with 200 μL of desalted extract in a final volume of 500 μL. The reaction was started by the addition of enzyme extract. After 30 min of incubation at 370C, the reaction was stopped by the addition of 1.0 mL FeCl3.3H2O (2.5% w/v) and trichloroacetic acid (6% w/v) in 2.5 M HCl. Protein was get precipitated and removed by centrifugation at 12,000 x g (40C) and absorbance was recorded at 540 nm. One unit of γ-glutamyl kinase activity defined as μg of γ-glutamyl hydroxamate formed per min per mg protein. γ-glutamyl hydroxamate was used as standard.
All the enzyme activities were calculated in terms of katal.
Determination of Soluble Proteins
Soluble protein was measured by the Bio-Red micro assay modification of the Bradford (1976) using bovine serum albumin as standard.
Statistical Analysis
Each treatment was analyzed with at least three replicates and standard deviation (SD) was calculated. Statistical analysis was performed using the students t-test; p<0.05 was considered statistically significant.
RESULTS
The proline content was significantly higher in the presence of 200 mM NaCl in both root (a) and shoot (b) during day 1 to 5 of plant growth in cv. T-44 (Fig. 1), while further in addition of 5 mM CaCl2 to 200 mM NaCl, proline content was found to be increased. In the presence of only 5 mM CaCl2 the similar result was found as in control. However, on day 5 of plant growth, the proline content increased approximately 5.0-fold in shoot (b) (Misra and Gupta 2005) and 2.6-fold in roots (a) in the presence of 200 mM NaCl. A further increase in proline content was observed with the addition of 5 mM CaCl2, 1.28-fold in shoot (b) and 1.37-fold in root (a). Shoot always maintained higher level of proline accumulation than root.
Figure 2 depicted pyrroline-5-carboxylate reductase activity in root (a) and shoot (b) in absence and in the presence of CaCl2, NaCl and their combination. The level of P-5-C reductase activity in both root and shoot increased from day 1 to 5 of plant growth in non-saline control as well as in the presence of 5 mM CaCl2, 200 mM NaCl and their combination.
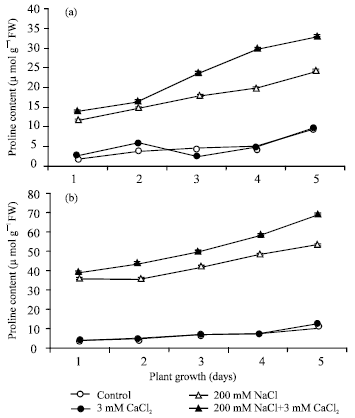 | |
| Fig. 1: | Free proline content (μmol g-1 FW) in root (a) and in shoot (b) during seed germination under conditions of absence and presence of different salts. Each value represents mean of three independent observations and SD determined. Data are statistically significant at p<0.05 |
Among all treated sets, 200 mM NaCl exhibited higher P-5-C reductase activity than the others in both root (a) and shoot (b) of green gram from day 1 to 5 of plant growth. Shoot (b) always maintained higher activity than the root (a). Thus, on day 5, 200 mM NaCl brought about 2.5 fold higher (p<0.05) P-5-C reductase activity in shoot (b) over the respective non-saline control. While in root (a) on day 5 (peak activity) approximately 2.1 fold increase in P-5-C reductase activity was observed as compared to non-saline control. In addition of 5 mM CaCl2 to 200 mM NaCl, the P-5-C reductase activity was further increased, about 1.17-fold in shoot (b) and 1.22-fold in root (a) as compared to 200 mM NaCl on day 5 of plant growth.
Figure 3 depicted proline oxidase activity in both root (a) and shoot (b) in non-saline control as well as in the presence of CaCl2, NaCl and their combination. The level of proline oxidase activity in both root and shoot increased from day 1 to 5 of plant growth in non-saline control and in the presence of 5 mM CaCl2, 200 mM NaCl and their combination. In all three treatments, the proline oxidase activity was found to decline throughout the germination in both root and shoot as compared to non-saline control. However, proline oxidase activity decreased in the presence of 200 mM NaCl by about 3.2- fold (p<0.05) in shoot (b) (Misra and Gupta, 2005) and 1.6-fold in roots (a) on day 5 of plant growth. In addition of 5 mM CaCl2 to 200 mM NaCl the proline oxidase activity was further decreased from day 1 to 5 of plant growth in both root and shoot as compared to 200 mM NaCl. In addition of 3 mM CaCl2 to 200 mM NaCl, proline oxidase activity was decreased about 1.17-fold (p<0.05) in shoot (b) and 1.39-fold decrease in root (a) on day 5 of plant growth as compared to 200 mM NaCl.
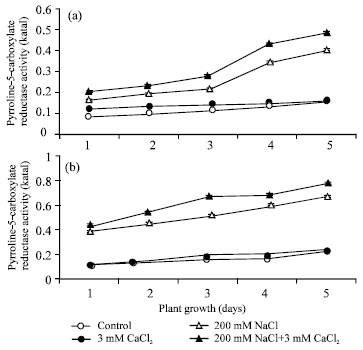 | |
| Fig. 2: | Pyrroline-5-carboxylate reductase activity in root (a) and in shoot (b) during seed germination under condition of absence and presence of different salts. The P-5-CR activity was calculated in terms of katal. Each value represents mean of three independent observations and SD determined. Data are statistically significant at p<0.05 |
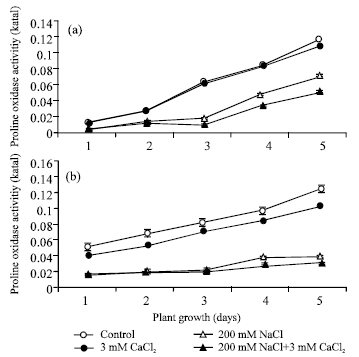 | |
| Fig. 3: | Proline oxidase activity in root (a) and in shoot (b) during seed germination under condition of absence and presence of different salts. The proline oxidase activity was calculated in terms of katal. Each value represents mean of three independent observations and SD determined. Data are statistically significant at p<0.05 |
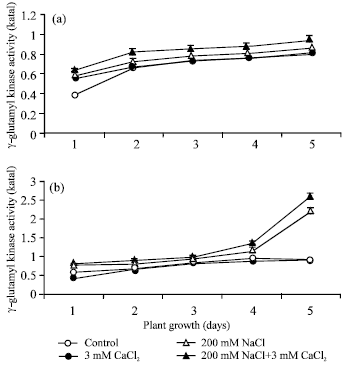 | |
| Fig. 4: | γ-glutamyl kinase activity in root (a) and in shoot (b) during seed germination under condition of absence and presence of different salts. The γ-glutamyl kinase activity was calculated in terms of katal. Each value represents mean of three independent observations and SD determined. Data are statistically significant at p<0.05 |
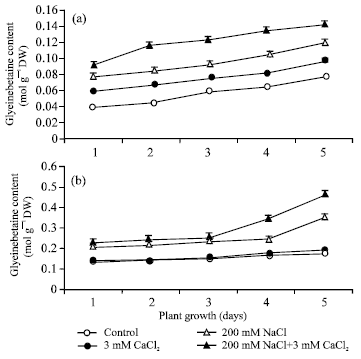 | |
| Fig. 5: | Glycinebetaine content (mol g-1 DW) in root (a) and in shoot (b) during seed germination under conditions of absence and presence of different salts. Each value represents mean of three independent observations and SD determined. Data are statistically significant at p<0.05 |
Shoot always maintained higher activity than the root. Salt inhibition of proline oxidase could contribute to the higher levels of proline as noted in Fig. 1.
γ-glutamyl kinase activity in both root (a) and shoot (b) tissues increased from day 1 to 5 of plant growth in non-saline control as well as in all treated sets (Fig. 4). The higher activity of γ-glutamyl kinase was found in presence of 200 mM NaCl in root (a) about 1.1-fold and in shoot (b) about 2.3-fold (p<0.05) on day 5 of plant growth. Shoot always maintained higher activity than the root. In addition of 5 mM CaCl2 to 200 mM NaCl, the γ-glutamyl kinase activity was further increased in both root as well as in shoot. In addition of 5 mM CaCl2 to 200 mM NaCl, γ-glutamyl kinase activity about 1.2-fold and 1.1-fold increased was observed in shoot (b) and in root (a), respectively, on day 5 of the plant growth.
Figure 5 depicted the level of glycinebetaine in non-saline control and in the presence of CaCl2, NaCl and their combination. The accumulation of glycinebetaine was considerably higher in shoot (b) than in root (a) and increased during plant growth from day 1 to 5. About 2.0-fold and 1.6-fold increased in shoot (b) and in root (a), respectively, on day 5 of plant growth compared to non-saline control. In addition of 5 mM CaCl2 to 200 mM, glycinebetaine accumulation was further increased about 1.3-fold in shoot (b) and 1.67-fold in root (a) on day 5 of plant growth. It has been also observed that glycinebetaine accumulation significantly increased in the presence of 3 mM CaCl2 as compared to non-saline control in shoot.
DISCUSSION
Metabolic stress caused by sodium chloride may result in decreased plant growth (Misra and Dwivedi, 2004). Cellular events such as compartmentation and osmotic adjustment in salt tolerant plants may allow continued growth in presence of NaCl (Misra and Gupta, 2005; Volkmar and Steppuhn, 1998). Proline may also act as an enzyme protectant stabilizing the structure of macromolecules and organelles. Generally proline accumulation appears to be a plant response associated with salt tolerance not a reaction to salt stress damage (Misra and Gupta, 2005; Lacerda et al., 2003). It has been reported that proline accumulation was not related to salt tolerance or to salt stress acclimation, but probably is just on expression of the seedling reaction to the stress damaging (Lacerda et al., 2003).
Increased proline accumulation under NaCl stress may be due to breakdown of proline-rich protein or de novo synthesis of proline (Tiwari and Singh, 1991). It may also be due to prevention of feedback inhibition of the biosynthetic enzyme caused by sequestering proline away from its site of synthesis or by relaxed feedback inhibition of the regulatory step or by decreased activity of proline oxidase involved in degradation of proline. Therefore, accumulation of proline oxidation or diminished incorporation of proline into protein due to impaired protein synthesis and reduced growth. The increased proline content in presence of 200 mM NaCl can be correlated with the increased synthesis of Δ1pyrroline-5-carboxylase reductase activity (Madan et al., 1995) and the low activity of the degrading enzyme, proline oxidase. Proline accumulation under salt stress can be explained by the higher inhibitory rate of proline dehydrogenase and proline oxidase
The level of proline oxidase activity was decreased in root and shoot of green gram seedlings in presence of sodium chloride stress (Misra and Gupta, 2005). Proline oxidase activity is inversely correlated with proline content of both the tissues under salt stress. Proline oxidase and proline dehydrogenases were significantly inhibited in the roots and shoots of salt-stressed mung bean seedlings (Sudhakar et al., 1993). Therefore, this enzyme converts free proline into glutamate. Decreased proline oxidase activity and the simultaneously increase in proline accumulation occurred in drought-stressed barley leaves, or low temperature stress in wheat (Charest and Phan, 1990). Addition of CaCl2 to the salt stressed seedlings caused decreased proline oxidase activity in root and in shoot of green gram. However, the level of proline oxidase activity in the CaCl2 treated seedlings was higher than that of the control. It has also been reported in Sorghum bicolor roots that the Ca2+ supplement enhanced the NaCl (150 mM NaCl) stress-induced proline accumulation (Colmer et al., 1996). In contrast, in Ulva fasciata, there is an inverse relationship between Ca2+ and proline contents (Lee and Liu, 1999). So that, we suggest that Ca2+-Na+ interaction is important for the osmotic adjustment of NaCl stressed plants and of fundamental importance for the maintenance of plant growth.
The increased level of γ-glutamyl kinase activity during plant growth in presence of salinity stress was inversely correlated with the proline oxidase activity of salt stressed green gram tissues.
P-5-C reductase, a key enzyme, has been shown to catalyze the reaction from glutamate to proline. P-5-C reductase activity has been shown to increase under salt stress (Kumar et al., 2003, Ramanjulu and Sudhakar 2001) as proline accumulates. Salt enhanced proline accumulation may, therefore, be due to increased production of P-5-C, which is then converted to proline by normal or enhanced level of P-5-C reductase. Pyrroline-5-carboxylate reductase was reported increasing under salt stress in Chlorella (Laliberte and Hellebust, 1989), Soybean (Delauney and Verma, 1990) and in NaCl adapted cells of Mesembryanthemum nodoflorum (Treichel, 1986). Therefore, proline accumulation in the root and shoot of NaCl stressed green gram seedlings can be attributed in part to the increased levels of P-5-C reductase and γ-glutamyl kinase activity. Addition of CaCl2 increased the P-5-C reductase activity as well as γ-glutamyl kinase activity in salt tolerant green gram. The enzyme activity of the calcium-treated plants was higher than that of untreated plants. Increased levels of both the enzymes activities coincide with the increased level of proline content in root and shoot of CaCl2 treated green gram seedlings.
The importance of glycinebetaine in osmotic adjustment and the activation of its biosynthesis under osmotic stress have been investigated (Rhodes and Hanson, 1993; Russell et al., 1998). Different approaches have been used to genetically engineered plants for enhancing stress tolerance in plants by manipulating the level of glycinebetaine (Sakamoto and Murata, 2000) and genes encoding enzymes for scavenging active oxygen species, late bryogenesis proteins, transcription factors, membrane modifying enzymes and proteins involved in maintaining in homeostasis (Zhang et al., 2000). Sodium chloride stress increased the level of glycinebetaine in shoot and root of green gram cultivar. Addition of 5 mM CaCl2 to the 200 mM NaCl stressed seedlings increased the glycinebetaine content to a greater extent than in untreated seedlings (control). Subcellular compartmentation of glycinebetaine biosynthesis was high (Sakamoto et al., 1998) in rice, is play an important role in salt tolerance. Although the exact mechanism of organic solute accumulation as a result of the Ca2+-Na+ interaction has not been established, it was suggested that an increase in cytosolic free calcium concentration could act at a transcriptional level inducing osmotic regulated gene expression of enzymes involved in the biosynthetic pathway of proline (Zhu et al., 1997). We believe that this mechanism, at least for proline accumulation, could be operating in salt tolerant green gram cultivar T-44.
An overview of the data presented here suggests that salt stressed seedlings in the presence of Ca2+ increased both osmoticants accumulation in both root as well as in shoot of salt tolerant green gram cultivar relative to those grown in 200 mM NaCl, suggesting an interactive effect of Ca2+ and Na+ on organic osmoregulators accumulation in these tissues. Shoot always maintained a higher concentration of both the solutes than the root. Supplementation of calcium to the system leads to an increase in proline and glycinebetaine. Increased proline accumulation in calcium chloride treated seedling is due to decreased level of proline oxidase (degrading enzyme) and increased levels of the activities of P-5-C reductase and γ-glutamyl kinase (synthesizing enzyme). Therefore, it may also suggest that Ca2+ could have a protective effect in tissues, which is of fundamental importance for the maintenance of plant growth in salt stressed seedlings.
ACKNOWLEDGMENTS
The authors are thankful to Dr. Aseem Chauhan, Chairman and Director General K.K. Ohari, Amity University, Lucknow Campus, Lucknow for their support and encouragement.
REFERENCES
- Bates, L.S., R.P. Waldren and I.D. Teare, 1973. Rapid determination of free proline for water-stress studies. Plant Soil, 39: 205-207.
CrossRefDirect Link - Boggess, S.F., C.R. Stewart, D. Aspinatl and L.G. Paleg, 1976. Effect of water stress on proline synthesis from radioactive precursors. Plant Physiol., 58: 398-401.
Direct Link - Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248-254.
CrossRefPubMedDirect Link - Charest, C. and C.T. Phan, 1990. Cold acclimation of wheat (Triticum aestivum): Properties of enzymes involved in proline metabolism. Physiol. Plant., 80: 159-168.
CrossRefDirect Link - Delauney, A.J. and D.P.S. Verma, 1993. Proline biosynthesis and osmoregulation in plants. Plant J., 4: 215-223.
CrossRef - Dodd, G.L. and L.A. Donovan, 1999. Water potential and ionic effects on germination and seedling growth of two cold desert shrubs. Am. J. Bot., 86: 1146-1153.
PubMedDirect Link - Epstein, E., 1998. How calcium enhances plant salt tolerance. Science, 280: 1906-1907.
CrossRefDirect Link - Ghoulam, C., A. Foursy and K. Fares, 2002. Effects of salt stress on growth, inorganic ions and proline accumulation in relation to osmotic adjustment in five sugar beet cultivars. Environ. Exp. Bot., 47: 39-50.
CrossRefDirect Link - Girija, C., B.N. Smith and P.M. Swamy, 2002. Interactive effects of sodium chloride and calcium chloride on the accumulation of proline and glycinebetaine in peanut (Arachis hypogaea L.). Environ. Exp. Bot., 47: 1-10.
CrossRefDirect Link - Hayzer, B.J. and T.H. Leisinger, 1980. The gene enzyme relationships of proline biosynthesis in Escherichia coli. J. Gen. Microbiol., 118: 287-293.
PubMedDirect Link - Huang, A.H.C. and A. Cavalieri, 1979. Proline oxidase and water stress induced proline accumulation in spinach leaves. Plant Physiol., 63: 531-535.
Direct Link - Kinraide, T.B., 1998. Three mechanisms of calcium alleviation of mineral toxicities. Plant Physiol., 118: 513-520.
Direct Link - Kumar, S.G., A.M. Reddy and C. Sudhakar, 2003. NaCl effects on proline metabolism in two high yielding genotypes of mulberry (Morus alba L.) with contrasting salt tolerance. Plant Sci., 165: 1245-1251.
CrossRefDirect Link - De Lacerda, C.F., J. Cambraia, M.A. Oliva, H.A. Ruiz and J.T. Prisco, 2003. Solute accumulation and distribution during shoot and leaf development in two sorghum genotypes under salt stress. Environ. Exp. Bot., 49: 107-120.
CrossRefDirect Link - Lee, T.M. and C.H. Liu, 1999. Correlation of decreased calcium contents with proline accumulation in the marine green macroalga Ulva fasciata exposed to elevated NaCl contents in seawater. J. Exp. Bot., 50: 1855-1862.
Direct Link - Lever, M., L. Bason, C. Leaver, C.M. Hayman and S.T. Chambers, 1992. Same-day batch measurements of glycinebetaine, carnitine and other betaine in biological material. Anal. Biochem., 205: 14-21.
Direct Link - Liu, J. and J.K. Zhu, 1998. A calcium sensor homologue required for plant salt tolerance. Science, 280: 1943-1944.
PubMedDirect Link - Madan, S., H.S. Nainawatte, R.K. Jain and J.B. Choudhury, 1995. Proline and proline metabolizing enzymes in vitro selected NaCl tolerant Brassica juncea under salt stress. Ann. Bot., 76: 51-57.
Direct Link - Misra, N. and U.N. Dwivedi, 1990. Nitrogen assimilation in germinating Phaseolus aureus seeds under saline stress. J. Plant Physiol., 135: 719-724.
CrossRef - Misra, N. and U.N. Dwivedi, 1995. Carbohydrate metabolism during seeds germination and seedling growth in green gram under saline stress. Plant Physiol. Biochem., 33: 33-38.
Direct Link - Misra, N. and U.N. Dwivedi, 2004. Genotypic difference in salinity tolerance of green gram cultivars. Plant Sci., 166: 1135-1142.
CrossRefDirect Link - Moftah, A.E. and B.E. Michel, 1987. The effect of sodium chloride on solute Potential and Proline accumulation in soybean leaves. Plant Physiol., 83: 238-240.
Direct Link - Prat, D. and R.A. Fathi-Ettai, 1990. Variation in organic and mineral components in young Eucalyptus seedlings under saline stress. Physiol. Plant, 79: 479-486.
Direct Link - Ramajulu, S. and C. Sudhakar, 2001. Alleviation of NaCl salinity stress by calcium is partly related to the increased proline accumulation in mulberry (Morus alba L.) callus. J. Plant Biol., 28: 203-206.
Direct Link - Rhodes, D. and A.D. Hanson, 1993. Quaternary ammonium and tertiary sulfonium compounds in higher plants. Ann. Rev. Plant Physiol. Plant Mol. Biol., 44: 357-384.
CrossRef - Russell, B.L., B. Rathinasabapathi and A.D. Hanson, 1998. Osmotic stress induces expression of choline monooxygenase in sugar beet and Amaranth. Plant Physiol., 116: 859-865.
Direct Link - Sakamoto, A. and N. Murata, 2000. Genetic engineering of glycinebetaine synthesis in plants: Current status and implications for enhancement of stress tolerance. J. Exp. Bot., 51: 81-88.
CrossRefDirect Link - Snedden, W.A. and H. Fromn, 1998. Calmodulin, calmodulin-related proteins and plant responses to the environment. Trends Plant Sci., 3: 299-304.
CrossRefDirect Link - Sudhakar, C., P.S. Reddy and K. Veeranjaneyulu, 1993. Effects of salt stress on the enzymes of proline synthesis and oxidation of green gram seedlings. J. Plant Physiol., 141: 621-623.
Direct Link - Tewari, T.N. and B.B. Singh, 1991. Stress studies in lentil (Lens esculenta Moench). II. Sodicity-induced changes in chlorophyll, nitrate and nitrite reductase, nucleic acids, proline, yield and yield components in lentil. Plant Soil, 136: 225-230.
CrossRefDirect Link - Volkmar, K.M., Y. Hu and H. Steppuhn, 1998. Physiological responses of plants to salinity: A review. Can. J. Plant Sci., 78: 19-27.
CrossRef - Yeo, A., 1998. Molecular biology of salt tolerance in the context of whole plant physiology. J. Exp. Bot., 49: 915-929.
Direct Link - Zhang, J., N.Y. Kluaeva, X. Wang, R. Wu, T.H.D. Ho and H.T. Nguyen, 2000. Genetic engineering for abiotic stress resistance in crop plants in vitro. Cell Dev. Biol. Plant, 36: 108-114.
CrossRefDirect Link - Zhu, J.K., P.M. Hasegawa, R.A. Bressan and H.J. Bohnert, 1997. Molecular aspects of osmotic stress in plants. Crit. Rev. Plant Sci., 16: 253-277.
CrossRefDirect Link - Misra, N. and A.K. Gupta, 2005. Effect of salt stress on proline metabolism in two high yielding genotypes green gram. Plant Sci., 169: 331-339.
CrossRefDirect Link - Morgan, J.M., 1984. Osmoregulation and water stress in higher plants. Annu. Rev. Plant Physiol., 35: 299-319.
CrossRefDirect Link - Hare, P.D., W.A. Cress and J. van Staden, 1998. Dissecting the roles of osmolyte accumulation during stress. Plant Cell Environ., 21: 535-553.
CrossRefDirect Link - Bohnert, H.J. and R.G. Jensen, 1996. Strategies for engineering water-stress tolerance in plants. Trends Biotechnol., 14: 89-97.
CrossRefDirect Link








