ABSTRACT
Biodegradable polymers derived from biomass have recently obtained much attention from the public because they are synthesized from renewable raw materials. Polyhydroxybutyrate (PHB) is one such biopolymer. The objective of study is to identify PHB accumulating Pseudomonas sp. which is obtained from Microbial Type Culture Collection, Chandigarh. All the 5 species showed positive result for PHB accumulation by rapid screening method using Sudan black B stain and Microscopy. Optimization of time as carried out and it showed that 48 h of incubation found to be optimum for maximum accumulation in Pseudomonas aeruginosa. Biochemical analysis of various cheap substrates such as agricultural residues (rice bran and wheat bran), forestry residues (saw dust) and industrial residues (activated sludge) which can be used as a carbon source showed that activated sludge has less nitrogen, lignin and phenolic content which enhanced maximum accumulation than the other residues in P. aeruginosa. Biochemical analysis of PHB by Nuclear Magnetic Resonance (NMR) spectrophotometer was carried out and the peaks correspond to the chemical group of PHB. Agricultural application showed that PHB enhanced germination and overall plant growth in leguminous plants.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajbmb.2013.293.303
URL: https://scialert.net/abstract/?doi=ajbmb.2013.293.303
INTRODUCTION
The superior qualities such as mouldability, durability, impervious to water and economic oblige of the synthetic plastics makes mankind completely depend on them for their daily life (Skrovanek et al., 1986). The usage of synthetic plastics is increasing day by day, but they create a lot of problem to the environment because of their nondegradability. In the soil they impede the growth of the root of the plants and affect the biological balance of the soil. In the urban areas the plastic bags clog the drainages which provide an excellent breeding ground for the disease causing mosquitoes.
The plastic bags disposed in the marine environment kills many marine animals because they mistake them as food and when they are eating it cause a chocking so the animals are killed. They destroy the home environment of many species present in the marine environment. A loss of biodiversity is observed (Brandl et al., 1990).
People are looking forward for a solution to replace the synthetic plastics with plastic which is ecofriendly. One such plastic is Polyhydroxybutyrate (PHB) produced by microorganisms as Polyhydroxyalkanoate (PHA) and gets accumulated as PHB (Madison and Huisman, 1999). It was first discovered by a French Microbiologist (Lemoigne, 1926) in Bacillus megaterium. It is a polymer of betahydroxybutyric acid and produced during the unbalanced growth condition (C: N ratio). Both Gram positive and Gram negative bacteria have been employed for the production of PHB. Among them Bacillus sp. and Pseudomonas sp. accumulate more amount of PHB and it is nearly 80% of dry weight of cell.
It is a storage polymer and present in amorphous state so it can be easily extracted with organic solvents (Otari and Ghosh, 2009). Since they are biodegradable they find application in medical, agricultural and food industries. Even though PHB is the most investigated among the biodegradable polymers in recent years, but their high price compared with the conventional plastics has hampered their use in wide range of application.
A thorough investigation was carried out to find out the potential PHB accumulators, production of PHB by using less expensive substrates and its application in the field of agriculture.
MATERIALS AND METHODS
Bacterial culture collection from MTCC: Bacterial cultures such as Pseudomonas sp. was obtained from Microbial Type Culture Collection (MTCC), in the Institute of Microbial Technology Council of Scientific and Industrial Research, Chandigarh (Table 1).
Revival of cultures from MTCC: The bacterial cultures from MTCC were obtained in a lyophilized form and revived as per the instruction provided in the manual of MTCC. The ampoules were aseptically opened since the contents were in vacuum. A mark was made on the ampoule near the middle of the cotton wool. The surface around the mark was disinfected with alcohol and the ampoule was broken at the marked area.
The upper end of the ampoule was removed gently and cotton plug was drawn to the other end. About 0.3-0.4 mL of the specified medium was added carefully without frothing and aerosol formed to get a clear suspension of the culture supplied by MTCC.
Checking for culture purity: The cultures were inoculated onto nutrient agar by quadrant streak method with intermittent incineration to obtain isolated colonies.
Identification of PHB accumulators by phenotypic methods
Growth on Nutrient agar with 1% Nile blue A (Ostle and Holt, 1982): Nutrient agar was prepared with 1% Nile blue A and dispensed in sterile Petri dishes. Bacterial cultures were streaked onto the plates and incubated at 37°C for 1-2 days. The appearance of the colonies was noted.
| Table 1: | Bacterial cultures from MTCC |
 | |
Microscopy (Burdon, 1946): Bacterial smears were made on a clean slide by using 48 h old culture heat fixed and flooded with Sudan black for 10 min. Then dipped in xylene for 2-3 min and counter stained with safranin for 1 min. The smears were observed under microscope.
Optimization of time course
Disruption of cells by chemical methods and PHB estimation: Nutrient broth was prepared in test tubes, sterilized and inoculated with MTCC cultures. The medium were incubated at 37°C for 24-96 h and PHB was estimated at every 24 h interval. About 5 mL of the culture centrifuged at 10,000 rpm for 10 min at room temperature and the supernatant was discarded. The pellet was suspended in 2.5 mL of sodium hypochlorite and 2.5 mL of chloroform and it was incubated at 30°C for 1 h. The above contents were centrifuged at 1500 rpm for 10 min at room temperature (Law and Slepecky, 1961).
The upper hypochlorite phase, the middle chloroform containing undisturbed cells and the bottom chloroform phase with PHB were separated using a micropipette. The contents were again centrifuged at 1500 rpm for 10 min at room temperature and the phase other than chloroform with PHB was removed carefully using micropipette. Concentrated sulphuric acid was added to the chloroform phase containing PHB, was then boiled at 100°C in a water bath for 10 min. The absorbance of the samples was read at 230 nm using UV Spectrophotometer. The readings obtained were plotted in standard graph of crotonic acid and the concentration of PHB was determined.
Biochemical analysis of waste substances: The biochemical analysis of various waste substances (carbon, nitrogen, lignin and phenolic content) was carried out in Abitech testing Laboratory, Coimbatore, India.
Estimation of PHB using less expensive substrates
Agro industrial wastes (Rice bran and Wheat bran): About 2 g of rice bran and wheat bran were collected from local agro industry of Coimbatore, washed thoroughly with distilled water for 3 times and immersed in 125 mL of distilled water in 250 mL of flasks and sterilized at 121°C for 15 min. inoculated with Aspergillus sp. and incubated at room temperature for 72 h. Following incubation the fungus was separated by filtration by using Whatmann No. 1 filter paper. About 50 mL of the nutrient broth was prepared and sterilized. Fifty milliliter of the filtrate was added aseptically with it. The flasks were inoculated with Pseudomonas spp. and incubated for 48 h at 37°C.
After the incubation, 10 mL of the culture was taken and centrifuged at 8000 rpm for 15 min. The supernatant was discarded and the pellet was treated with 10 mL of sodium hypochlorite and the mixture was incubated at 37°C for 2 h. The mixture was centrifuged at 5000 rpm for 15 min and then washed with distilled water, acetone, methanol respectively for washing and extraction. After washing, the pellet was dissolved in 5 mL of boiling chloroform (Loba) and it was evaporated by pouring the solution on sterile glass tray and kept at 4°C and the powder was collected for further analysis.
Forestry residue (Saw dust): Saw dust collected from local saw mill of Coimbatore was used as a cheap carbon source for the accumulation of PHB. One gram and 2 g of saw dust was added with 5 mL of 2% dilute sulphuric acid and the resulting mixture was stirred for 1 h at 30°C and then incubated at 120°C for 1 h. Then the pH was adjusted to 7 and filtered through Whamann No.1 filter paper and mixed with 100 mL of nutrient broth.
MTCC cultures were inoculated and incubated at 37°C for 24 h. Following incubation, 10 mL of the culture was taken and centrifuged at 8000 rpm for 15 min. The supernatant was discarded and the pellet was treated with 10 mL of sodium hypochlorite (Loba) and the mixture was incubated at 30°C for 2 h. After incubation, the mixture was centrifuged at 5000 rpm for 15 min and then washed with distilled water, acetone, methanol, respectively for washing and extraction. After washing, the pellet was dissolved in 5 mL of boiling chloroform (Loba) and the chloroform was evaporated by pouring the solution on sterile glass tray and kept at 4°C. After evaporation the powder was collected for further analysis.
Industrial waste (Activated sludge): Activated sludge collected from Amaravathi Paper Mills, Coimbatore was used as a cheap nutrient source for PHB accumulation. Activated sludge of 1 and 2 g was incorporated into the nutrient broth of 100 mL as an additional carbon source. MTCC cultures were inoculated and incubated for 48 h at 37°C. Following incubation, 10 mL of the culture was taken and centrifuged at 8000 rpm for 15 min. The supernatant was discarded and the pellet was treated with 10 mL of sodium hypochlorite (Loba) and the mixture was incubated at 30°C for 2 h. After incubation, the mixture was centrifuged at 5000 rpm for 15 min and then washed with distilled water, acetone, methanol respectively for washing and extraction.
After washing, the pellet was dissolved in 5 mL of boiling chloroform (Loba) and it was evaporated by pouring the solution on sterile glass tray and kept at 4°C. After evaporation the powder was collected for further analysis.
Analysis of PHB by Biochemical method
Nuclear Magnetic Resonance (NMR) spectroscopy: PHB was extracted from maximum PHB producer was suspended in spectrochem grade deuterochloroform (CDCl3). The 1H NMR spectra of sample was obtained at 400 MHZ using a model Bruker Advance 400 NMR spectrometer (Bruker BioSpin AG, Switzerland).
Application of PHB in the field of agriculture: About 100 mg of PHB was mixed with 100 g of garden soil taken in a pot (B). Seeds such as Green gram (Vigna radiata), Black gram (Vigna mungo), Horse gram (Dolichos uniflorus) and Green peas (Pisum sativum) were sown where as the other pot was without PHB (A). The pots were kept under the sunlight. The time taken for germination and overall growth of both treated and untreated plant was noted.
RESULTS
Growth with 1% Nile blue (Ostle and Holt, 1982): All the cultures formed grey colored colonies on nutrient agar with 1% Nile blue A indicates the accumulation of PHB.
Microscopy (Burdon, 1946): All strains were showed positive result for the presence of lipophilic PHB granules. PHB granules were observed as dark gray-black in the pink-sole red vegetative cells by using Sudan black B under the microscope and the result was shown in the Fig. 1.
Optimization of time course
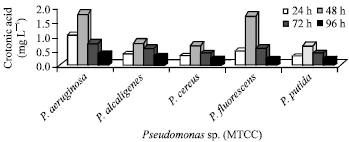
Disruption of cells by chemical methods and PHB estimation: Estimation of PHB by cell disruption method showed that Pseudomonas aeruginosa from MTCC has showed significant accumulation of PHB and the concentration of crotonic acid was found to be 1.0, 1.8, 0.8 and 0.4 mg L-1 in 24, 48, 72 and 96 h, respectively (Fig. 2).
 |
| Fig. 1: | Black sudanophilic granules within the pink cytoplasm |
 |
| Fig. 2: | Optimization of time course (Estimation of PHB by cell disruption method) |
| Table 2: | Composition of waste substances |
 | |
The remaining four species such as P. fluorescens, P. alcaligens, P. putida and P. cereus showed no significant accumulation of PHB and the range of crotonic acid was found to be 0.3-0.5, 0.7-1.0, 0.4-0.6 and 0.2-0.3 mg L-1 in 24, 48, 72 and 96 h, respectively.
Biochemical analysis of waste substances: The biochemical analyses of various waste substances (carbon, nitrogen, lignin and phenolic content) were shown the results presented in Table 2.
From Table 2, it was inferred that the carbon and nitrogen content of activated sludge is slightly higher than rice bran, wheat bran and sawdust whereas the lignin and phenol content of saw dust is higher than the remaining waste substances. The significant lowest value of carbon is 40% and the highest values is 55%. The significant lowest value of nitrogen is 1% and the highest value is 4%. The significant lowest value of lignin is 2% and the highest value is 4%. The significant lowest value of phenol is 0.1% and the highest value is 0.5%.
Agro industrial wastes (Rice bran and wheat bran): P. aeruginosa has accumulated maximum amount of PHB and was 1.0 and 1.4 g L-1, while other four species of Pseudomonas have accumulated minimum amount and showed minimum variation of PHB and the range was 0.4-0.9 and 0.6-1.2 g L-1 in rice bran and wheat bran, respectively (Fig. 3).
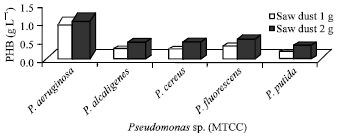
Forestry residue (Saw dust): Pseudomonas aeruginosa has showed maximum amount of PHB and was 1.0 and 1.1 g L-1 in 1 and 2 g of saw dust respectively whereas the other four species such as P. fluorescens, P. alcaligens, P. cereus and P. putida showed minimum amount of PHB and the range of PHB was found to be 0.1-0.3 and 0.3-0.5 g L-1 in 1 and 2 g of saw dust, respectively (Fig. 4).
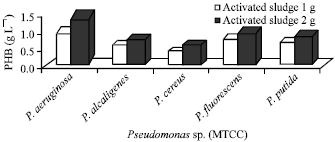
Industrial waste (Activated sludge): P. aeruginosa has accumulated increased amount of PHB and was about 1.0 and 1.5 g L-1 in 1 and 2 g of activated sludge, respectively, the remaining four species such as P. fluorescens, P. putida, P. alcaligens and P. cereus showed decreased accumulation and the range was 0.4-0.8 and 0.6-1.0 g L-1 in 1 and 2 g of activated sludge, respectively (Fig. 5).
 |
| Fig. 3: | Accumulation of PHB in agro industrial wastes |
 |
| Fig. 4: | Accumulation of PHB in forestry residue |
 |
| Fig. 5: | Accumulation of PHB in industrial waste |
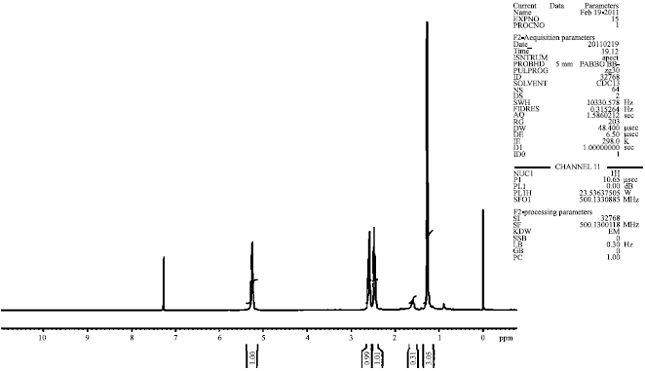
Nuclear Magnetic Resonance analysis (NMR): Analysis of extracted PHB from P. aeruginosa by NMR showed chemical shifts and were reported as values (ppm) relative to internal tetramethylsilane (TMS) in CDCl3. The characteristic peaks were observed at 1.2, 2.4-2.6 and 5.2 ppm. The peaks coincide, corresponding to the different types of carbon atoms present in the PHB structure, (O CH (CH) CH (C O) n 32. The assignments of the 1H NMR signals clearly revealed the extracted product of P. aeruginosa is PHB (Fig. 6).
Agricultural application of PHB: All the seeds sown in the soil treated with PHB germinated after 3 days whereas the seeds sown in the untreated soil have taken 5 days for germination. The overall growth such as height of the seedling, number of leaves and flowers are more in the treated soil than in the untreated soil (Fig. 7-10).
 |
| Fig. 6: | NMR spectrum of PHB |
 |
| Fig. 7: | Green gram (Vigna radiata) Pot A: Without PHB, Pot B: With PHB |
 |
| Fig. 8: | Black gram (Vigna mungo) Pot A: Without PHB, Pot B: With PHB |
 |
| Fig. 9: | Horse gram (Dolichos uniflorus) Pot A: Without PHB, Pot B: With PHB |
 |
| Fig. 10: | Green peas (Pisum sativum) Pot A: Without PHB, Pot B: With PHB |
DISCUSSION
Bioplastics are gaining importance because they are ecofriendly. The present study showed that all the 5 species of Bacillus accumulate PHB in the idiophase of the growth and decline in the late hours indicates that it is used as a carbon and energy source. Even though PHB act as a promising substitute for the synthetic plastics but the cost for upstream process is more. To overcome these difficulty cheap substrates can be used as a carbon source. The present investigation showed that cheap substrates such as agricultural wastes (rice bran and wheat bran), forestry residues (sawdust) and industrial waste (activated sludge) can be used as a carbon source. Among these activated sludge (2 g) encouraged the PHB accumulation may be due more carbon content, less nitrogen, phenol and lignin content because they act as an inhibitory substance for the growth and accumulation of PHB. Effort was made by Yellore and Desai (1998) and Van-Thuoc et al. (2008), to minimize the cost of production of PHB using whey and wheat bran hydrolyzate, respectively. Similar such studies were carried out by Bonatto et al. (2004) using sugar cane liquor, Yilmaz and Beyatli (2005) beetmolasses, Liu et al. (1998) molasses, Ahn et al. (2001) dairy whey, Vijayendra et al. (2007) Corn steep liquor, Chaijamrus and Udpuay (2008) cornsteep, Daneshi et al. (2008) corn syrup, Fukui and Doi (1998) plant oils in Ralstonia pickettii 61A6, Bacillus cereus M5, Recombinant Escherichia coli, Methylobacterium sp. ZP24, Bacillus sp., Bacillus megaterium ATCC 6748, Cupriavidus necator and Alcaligenes eutrophus, respectively, support the investigation. Our study is considerable because Pseudomonas aeruginosa accumulated the maximum amount of PHB compared to the above said studies. The study also showed that the strain is potent enough to accumulate the maximum amount of PHB in activated sludge.
Biochemical analysis by NMR of the extracted PHB showed that the peaks correspond with the chemical structure of PHB. The result is in parallel with Chaijamrus and Udpuay (2008) carried out a similar study in B. megaterium 6748. Similar studies were also reported by Yu and Marchessault (2000), Wendlandt et al. (2001), Sindhu et al. (2011), Deng and Hao (2001), Verlinden et al. (2011), Zhu et al. (2010), Zakaria et al. (2008), Ashby et al. (2011) and Berekaa (2012) supports our results. The use of PHB in the agriculture is very promising because it enhanced the seed germination and overall plant growth and act as carbon and energy source for the plant.
CONCLUSION
Finally, it is concluded that PHB can be used as a substitute for synthetic plastics and the present study also showed that use of PHB in agriculture is very promising. The biopolymer PHB act as a good growth promoting agent in the field of agriculture.
ACKNOWLEDGMENT
We gratefully acknowledge IIT, Chennai, for helping us in NMR work.
REFERENCES
- Ahn, W.S., S.J. Park and S.Y. Lee, 2001. Production of poly(3-hydroxybutyrate) from whey by cell recycle fed-batch culture of recombinant Escherichia coli. Biotechnol. Lett., 23: 235-240.
CrossRefDirect Link - Ashby, R., D. Solaiman and G.D. Strahan, 2011. Efficient utilization of crude glycerol as fermentation substrate in the synthesis of poly(3-hydroxybutyrate) biopolymers. J. Am. Oil Chem. Soc., 88: 949-959.
Direct Link - Berekaa, M.M., 2012. Genotypic detection of polyhydroxyalkanoate-producing bacilli and characterization of phaC synthase of Bacillus sp. SW1-2. Life Sci. J., 9: 518-529.
Direct Link - Bonatto, D., F. Matias, M.P. Lisboa, H.M. Bogdawa and J.A.P. Henriques, 2004. Production of short side chain-poly[hydroxyalkanoate] by a newly isolated Ralstonia pickettii strain. World J. Microbiol. Biotechnol., 20: 395-403.
CrossRefDirect Link - Burdon, K.L., 1946. Fatty material in bacteria and fungi revealed by staining dried, fixed slide preparations. J. Bacteriol., 52: 665-678.
Direct Link - Chaijamrus, S. and N. Udpuay, 2008. Production and characterization of polyhydroxybutyrate from molasses and corn steep liquor produced by Bacillus megaterium ATCC 6748. Agric. Eng. Int., Vol. 10.
Direct Link - Daneshi, A., H. Younesi, M. Ghasempouri and M. Sharifzedeh, 2008. Production of biodegradable biopolymer, poly-3-hydroxybutirate, from corn syrup by Ralstonia eutropha: Effect of nitrogen source. J. Biotechnol., 136: 424-424.
CrossRefDirect Link - Deng, X.M. and J.Y. Hao, 2001. Synthesis and characterization of poly(3-hydroxybutyrate) macromer of bacterial origin. Eur. Polymer J., 37: 211-214.
CrossRefDirect Link - Brandl, H., R.A. Gross, R.W. Lenz and R.C. Fuller, 1990. Plastics from bacteria and for bacteria: Poly(beta-hydroxyalkanoates) as natural, biocompatible and biodegradable polyesters. Adv. Biochem. Eng. Biotechnol., 41: 77-93.
PubMedDirect Link - Fukui, T. and Y. Doi, 1998. Efficient production of from plant oils by polyhydroxyalkanoates from Alcaligenes eutrophus and its recombinant strain. Appl. Microbiol. Biotechnol., 49: 333-336.
PubMedDirect Link - Yu, G. and R.H. Marchessault, 2000. Characterization of low molecular weight poly(β-hydroxybutyrate)s from alkaline and acid hydrolysis. Polymer, 41: 1087-1098.
CrossRefDirect Link - Law, J. and R.A. Slepecky, 1961. Assay of poly-β-hydroxybutyric acid. J. Bacteriol., 82: 33-36.
Direct Link - Liu, F., W. Li, D. Ridgway, T. Gu and Z. Shen, 1998. Production of poly-β-hydroxybutyrate on molasses by recombinant Escherichia coli. Biotechnol. Lett., 20: 345-348.
CrossRefDirect Link - Madison, L.L. and G.W. Huisman, 1999. Metabolic engineering of poly(3-hydroxyalkanoates): From DNA to plastic. Microbiol. Mol. Biol. Rev., 63: 21-53.
CrossRefDirect Link - Ostle, A.G. and J.G. Holt, 1982. Nile blue A as a fluorescent stain for poly-β-hydroxybutyrate. Applied Environ. Microbiol., 44: 238-241.
Direct Link - Otari, S.V. and J.S. Ghosh, 2009. Production and characterization of the polymer polyhydroxy Butyrate-co-polyhydroxy valerate by Bacillus megaterium NCIM 2475. Current Res. J. Biol. Sci., 1: 23-26.
Direct Link - Sindhu, R., B. Ammu, P. Binod, S.K. Deepthi, K.B. Ramachandran, C.R. Soccol and A. Pandey, 2011. Production and characterization of poly-3-hydroxybutyrate from crude glycerol by Bacillus sphaericus NII 0838 and improving its thermal properties by blending with other polymers. Braz. Arch. Biol. Technol., 54: 783-794.
Direct Link - Skrovanek, D.J., P.C. Painter and M.M. Coleman, 1986. Hydrogen bonding in polymers. 2. Infrared temperature studies of nylon 11. Macromolecules, 19: 699-705.
CrossRefDirect Link - Wendlandt, K.D., M. Jechorek, J. Helm and U. Stottmeister, 2001. Producing poly-3-hydroxybutyrate with a high molecular mass from methane. J. Biotechnol., 86: 127-133.
CrossRefDirect Link - Van-Thuoc, D., J. Quillaguaman, G. Mamo and B. Mattiasson, 2008. Utilization of agricultural residues for poly(3-hydroxybutyrate) production by Halomonas boliviensis LC1. J. Appl. Microbiol., 104: 420-428.
CrossRefDirect Link - Verlinden, R.A., D.J. Hill, M.A. Kenward, C.D. Williams, Z. Piotrowska-Seget and I.K. Radecka, 2011. Production of polyhydroxyalkanoates from waste frying oil by Cupriavidus necator. AMB Express, Vol. 1.
CrossRefDirect Link - Vijayendra, S.V.N., N.K. Rastogi, T.R. Shamala, P.K.A. Kumar, L. Kshama and G.J. Joshi, 2007. Optimization of polyhydroxybutyrate production by Bacillus sp. CFR 256 with corn steep liquor as a nitrogen source. Indian J. Microbiol., 47: 170-175.
CrossRefDirect Link - Zakaria, M.R., S. Abd-Aziz, H. Ariffin, N.A. Abdul Rahman, P.L. Yee and M.A. Hassan, 2008. Comamonas sp. EB172 isolated from digester treating palm oil mill effluent as potential polyhydroxyalkanoate (PHA) producer. Afr. J. Biotechnol., 7: 4118-4121.
Direct Link - Zhu, C., C.T. Nomura, J.A. Perrotta, A.J. Stipanovic and J.P. Nakas, 2010. Production and characterization of poly-3-hydroxybutyrate from biodiesel-glycerol by Burkholderia cepacia ATCC 17759. Biotechnol. Prog., 26: 424-430.
CrossRefDirect Link








