Research Article
Chemical Characterization of Enzymatically Treated Morinda Juice
Department of Botany, Shri Pancham Khemraj, Mahavidyalaya, Sawantwadi, MS, India
LiveDNA: 91.3600
D.K. Gaikwad
Department of Botany, Shivaji University, Kolhapur
All the fruits have some therapeutic properties as they are rich source of secondary metabolites including xanthones, flavonoids, benzophenones, lactones and phenolic acid. Growing evidence of the role of free radicals and antioxidants in health and ageing has focused more interest on phyto compounds (Lau et al., 2005). During fermentation the biological process results in series of some biochemical transformation due to the action of several enzyme from different microorganisms which are responsible for alcoholic fermentation (Rai et al., 2010). Differences in the antioxidant activity among grape juice, wine and vinegar were attributed to their different phenolic content and composition and to the other non-phenolic antioxidant present in the samples (Davalos et al., 2005). Genus Morinda belongs to family Rubiaceae and about 80 species belongs to this genus. Out of these species 7 species occurs in India (Hooker, 1982). Morinda citrifolia has attained significant economic importance worldwide in recent years through a variety of health and cosmetic products made from its leaves and fruits. These include fruit juices as well as powders made from the fruit or leaves (Nelson, 2006). It is also used in local medicines and as a famine food. Unripe fruits are cooked in curries and ripe fruits are consumed raw with salt. Fruit is cooked and mixed with coconut and eaten as stimulant on long sea voyages (Nelson, 2006). Morinda pubescens J.E. Smith (synonyms: Morinda tinctoria Roxb. and Morinda coreia Buch.) predominantly grows as a weed tree in vacant agricultural land and especially on uncultivated lands and along the boundaries of the cultivated fields. Ancient writings reveal that Morinda has long been cultivated in different parts of the Tamil Nadu state of India. Although the South Indian ancestors realized the therapeutic value of M. pubescens and used it in the traditional Indian medicinal systems like Siddha, lack of proper documentation resulted in loss of that knowledge (Prabhu et al., 2012). Further, its medicinal potential has yet to be studied scientifically.
In the present investigation an attempt have been made to clarify the Morinda juice using enzymes and its further characterization with GC-MS and FT-Raman spectroscopy.
Noni juice extraction: Fully matured and ripening Morinda fruits were collected from the tree and brought in laboratory. Ripened fruit of Morinda citrifolia were yellowish-white in color, while those of Morinda pubescens were brownish black in colour. The fruits were washed well without bruising the skin during cleaning and the juice was separated from the pulp and seeds using a fruit press. The extracted juice samples with pulp then treated with the enzymes pectinase and amylase at 50°C for 2 h. the time, temperature and concentration of enzymes were standardized according to the method of Sharma (2007). The enzymes were used at the concentration of 0.5% for the clarification of juice. The clarified juice ameliorated by additional sucrose and adjusted to 22°C soluble solids. The clarified juice was inoculated with 5% (v/v) active pure yeast culture (Saccharomyces cerevisiae) and fermented at 22±1°C for 45 days. Then the fermented juice was pasteurized (62±1°C) and analyzed for its composition. The clarified, unclarified and fermented juice was analyzed for different physicochemical parameters using A.O.A.C. methods (AOAC., 1980).
FT-raman spectroscopic analysis: The Raman spectra of extracted noni fruits were recorded in the spectral range of 35-4000 cm–1 using FT-Raman spectrometer (Bruker MultiRAM, Germany Make) Nd:YAG laser source with excitation wavelength of 1064 nm and resolution 4 cm–1. The infrared (IR) spectroscopy was used to confirm the composition phase. For this, the powder collected from the extracted samples were characterized by IR spectrometer (PerkinElmer, model 783, USA) in the spectral range of 450-4000 cm–1 with a resolution of 1 cm–1. To record IR spectrograms, a pellet was prepared by mixing KBr with the collected extract powder in 300:1 proportion and then pressing the pellet between the two pieces of polished steel.
GC-MS/MS analysis of the fermented noni juice: The fermented juice obtained after enzymatic clarification were analysed with a Hewlett-Packard (HP) 6890 gas chromatograph fitted with a Gerstel MPS2 auto sampler and coupled to a HP 5973 N mass spectrometer. The Gerstel MPS2 was operated in fast liquid injection mode with a 10 μL syringe (SGE, Australia) fitted. The gas chromatograph was fitted with an approx. 30 m×0.25 mm Phenomenex fused silica capillary column ZB-Wax, 0.25 μm film thickness. The carrier gas was helium (BOC gases, Ultra High Purity), flow rate 1.2 mL min–1. The oven temperature was started at 50°C, held at this temperature for 1 min, then increased to 220°C at 10° min–1 and held at this temperature for 10 min. The injector was held at 200°C and the transfer line at 250°C. For liquid injections, the sample volume injected was 2 μL and the splitter, at 30:1, was opened after 36 sec. Fast injection was done in purge split less mode with an inlet pressure of 25 psi g maintained until splitting. The glass liner (Agilent Technologies) for liquid injections was borosilicate glass with a plug of resilanised glass wool (2-4 mm) at the tapered end to the column. For SPME, a direct borosilicate glass liner with an id. of 1.5 mm (Agilent Technologies) was used and the Gerstel MPS2 was operated in SPME mode with either a 100 μm polydimethylsiloxane (PDMS) or 85 μm polyacrylate/ divinylbenzene (DVB) fibre fitted (fibres sourced from Supelco). Positive ion electron-impact spectra at 70 eV were recorded in the range m/z 35-350 for scan runs. For quantification of the compounds, mass spectra were recorded in the Selective Ion Monitoring (SIM) mode using NIST library.
Statistical analysis: The data on physico-chemical characteristics were analysed stastically by following Completely Randomized Design (CRD) (O'Mahony, 1986). For that triplicates were made and from each again three samples were used for further analysis. Experimental results were subjected to analysis of variance (ANOVA) and differences between means were assessed by Duncan’s new multiple range test as described by Steel and Torrie (1960).
The physico-chemical characteristics of non-clarified, enzymatically clarified and fermented Morinda juice is presented in Table 1. It is clear from the table that the enzymatic treatment (pectinase and diastage) enhances recovery of juice in both species of Morinda. It is also evident that the total polyphenols, flavonoids and acidity decreases in clarified and fermented juice of M. citrifolia while, the content of sugars elevated in clarified juice which further decreases in fermented juice. The fruit juice of M. pubescens also shows decrease intotal polyphenols, flavonoids and acidity on the other hand sugars are increases significantly in clarified as well as fermented juice. The pH is not significantly altered while, the TSS slightly decreases in clarified juice and substantially reduces in fermented juice of M. citrifolia and M. pubescens. The relative viscosity of the juice also decreases in clarified and fermented juice of both fruits. Physico-chemical properties of enzymatically clarified, unclarified and fermented Morinda juice.
FT-raman spectroscopy measurements: The following three signals are of main interest for the identification of different protein backbone confirmations compound: amide I to be detected between 1680 and 1600 cm–1 (stretching vibration of C-O, amide II observed in the range between 1580 and 1480 cm–1 and amide III to be found between 1300 and 1230 cm–1 (both associated with coupled C-N stretching and N-H bending vibrations of the peptide groups (Fig. 1). The peak originated at 1360 cm–1 is due to tryptophan. The FT-Raman spectra obtained from yellow coloured noni fruit, which have treated different treatment show significant key signals of piperine. Apart from the intense-C-H stretching vibrations between 2800 and 3100 cm–1, the main FT-Raman signals occur in the fingerprint range between 1100 and 1630 cm–1.
| Table 1: | Physico-chemical characterization of unclarified, clarified and fermented Morinda juice |
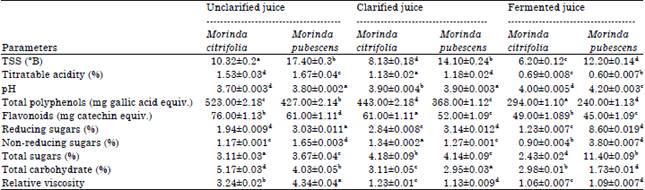 | |
| Data are expressed as Mean±SD (n = 3). Different letters for each column correspond to statistically different values (p<0.05) | |
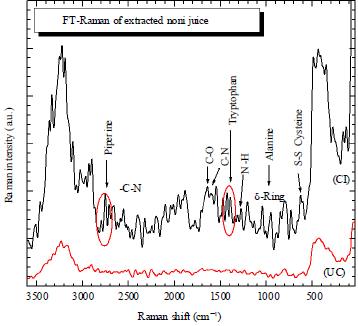 | |
| Fig. 1: | Raman spectrum measured on a fruit juice sample ripened Morinda fruits. The investigated wave number shift range varies between 100 and 3500 cm–1 |
From this is concluded that the extracted material contains the amide material. Ii is noted that this compound was used is the bio medicine applications. The phonon lifetime (τ) can be derived from the Raman spectra via the energy time uncertainty relation:
| (1) |
where, ΔE is the uncertainty in the energy of the phonon mode, ħ is the planck’s constant and Γ is the full width at half maximum (FWHM) of the Raman peak in units of cm–1. Phonon lifetime is mainly limited by two mechanisms: (i) an harmonic decay of the phonon into two or more phonons so that energy and momentum are conserved, with a characteristic decay time τA and (ii) perturbation of the translational symmetry of the crystal by the presence of impurities, defects and isotopic fluctuations, with a characteristic decay time τI. The phonon lifetime deduced from the Raman measurements is therefore written as:
| (2) |
The pectin can be associated with other plant polymers and the cell debris. One of the major problems encountered in the preparation of fruit juices and wine is cloudiness due to the presence of pectins. Commercial sources of fungal pectic enzymes have been used in fruit juice processing since the 1930's for clarifying fruit juices and disintegrating plant pulps to increase juice yields (MacMillan and Sheiman, 1974). Commercial enzymes are similar to the naturally occurring pectinases, cellulases and hemicellulases found in fruit during ripening. Most enzymes are marketed on the basis that they are Generally Recognized As Safe (GRAS) for their intended use in the juice process. Pectinase is a balanced mix of pectolytic enzymes designed to provide controlled maceration and depectinization of fruit. It contains high levels of Pectin Lyase (PL), which depolymerizes pectin chains and polygalacturonase (PG), which randomly cuts the polygalacturonic acid backbone of the insoluble pectin.
Rai et al. (2006) studied the efficacy of various pretreatment processes in comparison to pectinase treatment and explored the possibility of substitution of pectinase for juice pretreatment and reported that pretreatment mainly affects the color, clarity, viscosity and AIS (indicate pectin content), protein and suspended solid contents and has insignificant effect on TSS, acidity, pH and density of the juice. Wang et al. (2009 )studied effects of different maceration enzymes on yield, clarity and anthocyanin and other polyphenol contents in blackberry juice. In our study also the enzymatic treatments of pectinase and diastase shows significant effect on juice parameters such as pH, total acidity and soluble solids. The increase in total sugar percentage in treated juice and decrease in total carbohydrate may be due to the enzyme diastase which converts carbohydrates into simple polysaccharides and sugars. Thus, use of enzymatic treatments for the noni juice suggests its suitability for high yield and more clarified juice along with increase in sugar content. The astringent taste of non-clarified juice may be due to the higher levels of polyphenols and flavanoids. Here, decrease in total phenols and flavanoid content during enzymatic clarification may contribute in the reduction of astringency of juice. It was observed that there was a significant variation (p<0.05) of the TSS, pH and titratable acidity between unclarified, clarified and fermented juice of two species. Significant differences in the phenol and flavonoid content were found among enzymatically clarified (pectinase and diastase) and fermented juice (p<0.05).
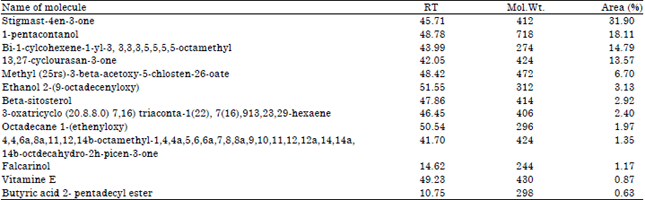
Mass spectral analysis of Bioactive compounds from fermented juice: Mass spectral analysis of fermented juice of Morinda citrifolia and Morinda pubecsens is shown in Table 2 and 3, While the total ion chromatogram and libraray standards are presented in Fig. 2 and 3. The mass spectra of fermented juice of Morinda shows different peaks and identification was made by retantion time, Molecular Weight and % area by compairing National Institute of Standards and Technology (NIST) library. Search results from NIST library gives probable compounds present in the spectra.
The mass spectra of fermented juice of M. citrifolia exhibits 14 important peaks out of which 4 peaks are having larger % area. They are Stigmast-4en-3-one, 1-pentacontanol, Bi-1-cylcohexene-1-yl-3, 3,3,3,5,5,5,5-octamethyl and 13,27-3-one showing their % area 31.90, 18.11, 14.79 and 13.57%, respectively.
| Table 2: | Compounds detected from GC-MS/MS of analysis of Fermented juice of Morinda citrifolia |
 | |
 | |
| Fig. 2: | GC-MS Spectra of fermented juice of M. pubescens fruits |
| Table 3: | Compounds detected from GC-MS/MS of analysis of fermented juice of Morinda pubescens |
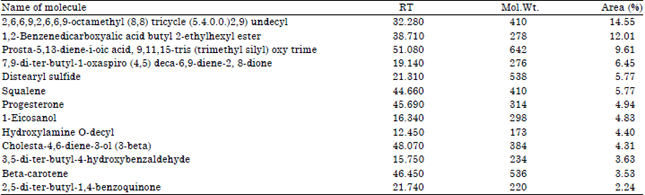 | |
Bioactive compounds and their medicobiological applications detected from fermented juice of M. citrifolia and M. pubescens: Several workers reported the applications of phytosterols like hypocholesterolaemic effects (Peterson et al., 1953), cardiotonic activity (Loynes and Gowdey, 1952) growth promoting and sexual reproduction inducing activity (Nes et al., 1982) in animals and humans.
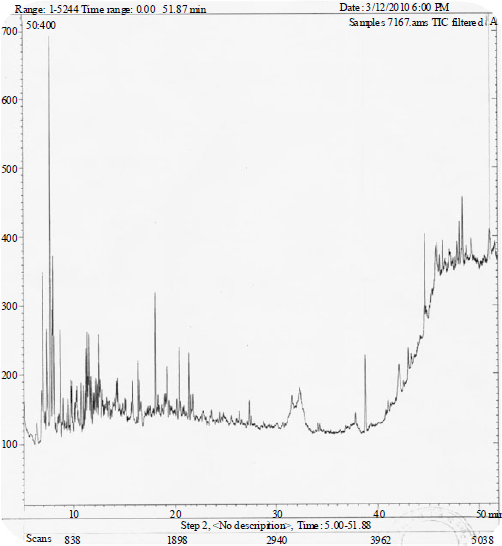 | |
| Fig. 3: | GC-MS Spectra of fermented juice of M. citrifolia fruits |
In the present investigation we also noticed the presence of stigmast-4-en-3-one in fermented juice of M. citrifolia as a major bioactive compound. It is clear from review of literature on Morinda citrifolia that the noni products are recommended as a cardiotonic agent (Wang and Su, 2001).
The isolated plant compounds falcarinol and falcarindiol have proven to be very bioactive (Christensen and Brandt, 2006) and in addition these compounds may interfere with the intracellular ROS production, quenching of these or ROS signaling functions (Zidorn et al., 2005) also found falcarinol to be an active compound with a pronounced toxicity against acute lymphoblastic leukemia cell line CEM-C7H2, with an IC50 of 3.5 μmol L–1. Beta-sitosterol decreases free radical generation in vitro (Moreno, 2003) and modulates antioxidant enzyme levels during pathogenesis (Vivancos and Moreno, 2005). Vitamin E is a fat soluble antioxidant vitamin that helps to neutralize potentially damaging free radicals in human body. American Cancer Society reported that vitamin E, especially when coupled with selenium, may reduce the risk of prostate cancer. Vitamin E supplementation was shown to increase the risk of heart failure (Lonn et al., 2005). Falcarinol, Beta sitosterol, Vit. E play a significant role as an antioxidants as reported by several workers (Christensen and Brandt, 2006). The presence of these phytochemicals in M. citrifolia juice also certainly improves the antioxidant potential. Beta sitosterol may play a role in preventing the development of certain cancers, namely colon, prostate and breast cancer, phosphorylation of Extracellular-signal Regulating Kinase (ERK) and p38 mitogen-activated protein kinase (MAPK) (Moon et al., 2007).
The Morinda fruit juice or products under the trade name Noni are recommended in various pathological activities (anti-viral, anti-fungal, Anti-tubercular and anti-bacterial) and metabolic disorders (arthritis, diabetes, high blood pressure, muscle aches and pains, menstrual difficulties, headaches, heart disease, AIDS, cancers, gastric ulcers, sprains, mental depression, senility, poor digestion, atherosclerosis and blood vessel problems) by several researchers (Hornick et al., 2003; Kamiya et al., 2004). With these above applications the occurrence of 13 different bioactive compounds from fermented juice of M. citrifolia may proves beneficial against the various disorders which will be found beneficial for further designing of drugs from M. citrifolia fruits.
The compounds reported from fermented juice of M. pubescens are about 13 showing their appreciable concentrations in fermented juice. Among these bioactive compounds Squalene, Progesteron, β-Carotene, hydroxylamine and benzoquinones are reported to have various applications by several researchers as follows β-Carotene is an organic compound and classified as a terpenoid. Van Arnum (1998) reported that naturally β-carotene is a precursor to vitamin A via the action of beta-carotene 15,15'-monooxygenase and biosynthesized from geranylgeranyl pyrophosphate. Antioxidant nutrients such as ascorbic acid, α-tocopherol and the carotenoids (e.g., beta carotene) that are present in plant-derived foods may have a major preventive role against carcinogenesis (Ames, 1983).
The 2,5-di(tert-butyl)-1,4-benzohydroquinone (tBHQ) belongs to a large family of phenolic antioxidants commonly used as food preservatives. which also play a significant role in oxidative stress (Yu et al., 1997). Benzoquinone and hydroquinone are the aromatic organic compounds which are the types of phenols. Squalene is used in cosmetics as a natural moisturizer. Squalene is also found in a variety of foods (for example: eggs, olive oil (0.7%), cookies, yeast, meat), cosmetics (for example: eye makeup, lipstick, baby powder), over-the-counter medications and health supplements (Atkinson, 2009). Christian (1982) reported that both squalene and squalane have low acute toxicity and are not significant human skin irritants or sensitizers. Squalene is an isoprenoid compound having six isoprene units that possesses antioxidant activities and it is widely produced in plants. Squalene protects cells against radicals, strengthens the immune system and decreases the risk of various cancers (Das et al., 2003). Squalene has been proposed to be an important part of the Mediterranean diet as it may be a chemopreventative substance that protects people from cancer (Owen et al., 2004).
CONCLUSION
In the present investigation we have reported different bioactive compounds from M. citrifolia and M. pubescens fruits. The fruits of M. pubescens exhibits a derivatives of quinones as well as Squalene as isoprenoid playing significant role in oxidative stress. Thus, the presence of these compounds in Morinda fruits may supports the antioxidant potential of phenolic compounds in M. pubescens fruits. while the Squalene reported from M. pubescens fruits may found beneficial in the development of various safety cosmetic skin moisturizers in safety assessment report of Squalene as well as the application of Squalene as a vaccine against antrax, diphtheria toxoid and influenzas like H1N1 as indicated. Thus, the Squalene from M. pubescens fruits may proves beneficial in designing various types of vaccines against these emerging common society diseases posing a great problem in densely populated country like India. Further it is essential to isolate, purified and characterize these bioactive compounds from Noni juice which may found beneficial in designing of various drugs in future.
The authors are highly thankful to Prof. G. B. Dixit, Head Dept. of Botany, Shivaji University, Kolhapur for providing Laboratory facility. Principal, Shri pancham Khemraj Mahavidyalaya, Sawantwadi and all respected members of SRDSP Mandal, Sawantwadi.