Research Article
Blood Metabolic Profile and Certain Hormones Concentrations in Egyptian Buffalo During Different Physiological States
Department of Animal Production, Faculty of Agriculture, Cairo University, 11431, Giza, Egypt
Blood Metabolic Profile (BMP) is a set of diagnostic procedures that are based on determining the various indicators in the blood of animals (Van Saun, 2000). The most common indicators in the blood of animals used in the preparation of the BMP are biochemical. Blood Metabolic Profile is used in assessing nutritional status and animal health (Herdt et al., 2000; Antunovic et al., 2009). Significant variations in the blood metabolic profile depend on many, genetic and non-genetic factors. One of the important factors is physiological status which affects on concentration of indicators in blood that are involved in the development of the blood metabolic profile (Antunovic et al., 2002; Roubies et al., 2006). The metabolic profile test that was by Payne et al. (1970), has been used for assessing metabolic status and diagnosis of metabolic disorders in dairy herds (Kida, 2002). The changes in blood constituents can reflect the physiological condition as well as nutritional and health status of cows. The thyroid hormones maintain the homeostasis of energy and protein metabolism, thermo-regulation, growth and productivity parameters (Huszenicza et al., 2002). High pregnancy and lactation, especially in the early stages, are very demanding physiological state of the organism when nutritional requirements are increased (Chilliard et al., 2005). During lactation the mammary gland secretary cells utilize 80% of the circulating metabolism in the blood for milk synthesis (Karapehlivan et al., 2007).
The aim of this study is to determine the influence of physiological status on blood metabolic profile, enzymes and concentrations of leptin, IGF-1 and thyroid hormones in the blood of buffalo cow.
This study was carried out at Mehlet Moussa Experimental Station which belongs to Animal Production Research Institute, north part of the Nile Delta, Egypt. Investigations were carried out on 24 buffalo (12 pregnant buffalo cows on the 60th day prior to parturition and 12 lactating buffalo cow from 10th day of lactation. Blood samples (10 mL) were collected from the jugular vein into heparinzed tubes. Blood samples were centrifuged at 3000 rpm for 20 min to separate plasma which was used for measuring minerals including calcium (Ca), inorganic phosphorus (p), potassium (K), sodium (Na) and chloride (Cl) concentrations determined by using Turbidimetric method using special kits delivered from Bio-Diagnostic®, Dokki, Giza, Egypt. Insulin like growth factor-1 (IGF-1, nmol L–1) was assessed by radioimmunoassay technique (RIA) using ready-made kits (Immunotech SAS-130 av. kit, France). The standard curve was between 0 and 1200 ng mL–1. The analytical sensitivity was reported to be 2 ng mL–1. The cross reaction of the antibody with other hormones was found to be extremely low. The samples were determined in one run and the intra-assay variation coefficient was 6.3%. While leptin concentration was determined by ELISA reader (BIO TEK ELX808), using Leptin ELISA kit sandwich (DRG Instruments GmbH, Germany) according to the manufacturer's guidelines. The standard curve was between 0 and 100 ng mL–1. The sensitivity of the curve was reported to be 1.0 ng mL–1. Direct radioimmunoassay technique was performed for assessment of plasma triiodothyronine (ng dL–1) and thyroxine, T4 (nmol L–1) concentrations by using special kits delivered from Immunotech radiova, Czech Republic. Aspartic aminotransferase (AST) and alanine transaminase (ALT) activities were measured by using colorimetric method according to Reitman and Frankel (1957). Alkaline phosphatase (ALP) activity was measured by using colorimetric method according to Belfield and Goldberg (1971). Glucose (mg dL–1) was determined by using enzymatic colorimetric method using Bio-Diagnostic® kit delivered from Dokki, Giza, Egypt according to Trinder (1996). Total protein (g dL–1) was determined colorimetrically by using Bio-Diagnostic® kit delivered from Dokki, Giza, Egypt according to Gornall et al. (1949). Cholesterol concentration (mg dL–1) in plasma was determined quantitatively (Colorimetric Method Liquzyme) by using (STAT LAB SZSL60-SPECTRUM) according to (Finley et al., 1978). Estimation of Triglycerides (mg dL–1) content in plasma was quantity (Colorimetric Method Liquzyme) determined by using (STAT LAB SZSL60-SPECTRUM). According to (Fossati and Prencipe, 1982). Urea and Creatinine contents were determined with commercial kits by using Bio-Diagnostic® delivered from Dokki, Giza, Egypt and finally measured using stat lab szl60-spectrum.
Statistical analysis of data was carried out applying SAS, package (SAS., 2000). Differences among means were checked according to Duncan (1955).
Blood electrolytes: Results in Table 1 indicated that there was a drop in calcium level during early stage of lactation (4.55‾10.21) than pregnant period buffaloes (10.35‾10.52 mg dL–1). The concentrations of plasma phosphorous were lower during lactation period than in pregnant period. The concentrations of potassium, sodium were higher during pregnant period than in lactation period. Chloride concentrations varied non-significantly during pregnancy and lactation. The concentrations of plasma chloride were higher during lactation than pregnant period.
Blood biochemical parameters: Results in Table 2 indicated that, the concentrations of plasma urea, cholesterol, glucose, triglyceride and total protein were lower during lactation period than in pregnant period. The concentrations of plasma creatinine were higher during lactation period than in pregnant period.
| Table 1: | Blood electrolytes concentration in pregnant and lactating Egyptian buffaloes |
 | |
| a,bMeans within the same row with different superscript differ significantly (p<0.05) | |
| Table 2: | Blood biochemical parameters in pregnant and lactating Egyptian buffalo cows |
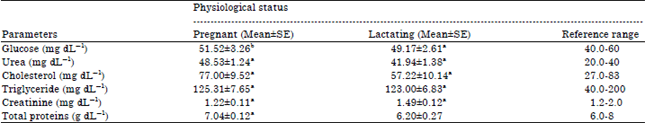 | |
| a,bMeans within column with group with different superscript differ significantly (p<0.05) | |
| Table 3: | Enzymatic activity of liver function in Egyptian buffaloes during pregnancy and lactation |
 | |
| Means within each column do not differ significantly from each other | |
| Table 4: | Influence of physiological status of Egyptian buffalo on blood hormones |
 | |
| Means within each column do not differ significantly from each other | |
The opposite trend recorded by Abd Ellah et al. (2013) measured blood constituents during the period before and after 6 months of pregnancy in buffaloes. Results revealed significantly higher (p<0.05) in serum creatinine after 6 months of pregnancy.
Blood enzyme activity: Data in Table 3 indicated that, the blood enzymes (AST, ALT and AlP) activities were insignificantly higher during pregnant period than in lactation period.
Blood hormones: Data in Table 4 indicated that, the concentrations of plasma IGF-1, leptin and thyroid hormones were lower during lactation period than in pregnant period. However, the difference was not significant.
Blood electrolytes: The lowered trend in Ca++ levels could be a result of the impaired absorption of food metabolites from the gastrointestinal precursor, excessive losses through urine, colostrums as it was much more drained in the colostrums during excessive milking and due to insufficient mobilization from the skeleton. As the stage of lactation progresses the serum calcium level increased which corroborates with the findings of Nale (2003). Serdaru et al. (2011) found that the calcium, phosphorus and magnesium concentrations in lactating group of buffaloes were lower in comparison with pregnant group. Hagawane et al. (2009) found that there was a drop in calcium level during early stage of lactation (8.19‾10.83) than normal healthy buffaloes (11.21‾10.19 mg dL–1). Hussain et al. (2001) indicated that the highest mean concentration of calcium was observed in pre-partum stage at 8th month of pregnancy which was 9.30‾10.12 mg dL–1. On the other hand, the lowest mean value of calcium was observed at parturition which was 6.90‾10.20 mg dL–1. Some researchers (Moghaddam and Hassanpour, 2008; Yokus and Cakir, 2006; Krajnicakova et al., 2003; Abdelrahman et al., 2002) said that, Ca levels were lower at postpartum than gestation period.
Moderate reduction in the levels of phosphorus might be due to the necessity of it for the colostrum synthesis (Serdaru et al., 2011) and enhanced carbohydrate metabolism. Hussain et al. (2001) indicated that the highest mean concentration of phosphorous was observed in pre-partum stage at 8th month of pregnancy which was 6.98‾10.12 mg dL–1. On the other hand, the lowest mean value of phosphorous was observed at parturition which was 3.77‾10.08 mg dL–1. Hagawane et al. (2009) found that the serum phosphorus level in early stage of lactation (4.64‾10.53 mg dL–1) was significantly (p<0.05) lower than the normal healthy control buffaloes.
Abd Ellah et al. (2013) measured blood constituents during the period before and after 6 months of pregnancy in buffaloes. They found that the values of serum potassium and phosphorus were significantly higher after 6 months of pregnancy compared with before Hussain et al. (2001) indicated that the highest mean concentration of potassium was observed in pre-partum stage at 8th month of pregnancy which was 4.90‾10.21 mol L–1. On the other hand, the lowest mean value of potassium was observed at parturition which was 4.31‾10.21 mmol L–1. Kuhn et al. (2006) found that sodium and potassium plasma levels showed highest values (p<0.05) at 7th day respect to 30th day both in lactation and dry period. Hussain et al. (2001) indicated that the highest mean concentration of sodium was observed in pre-partum stage at 8th month of pregnancy which was 147.14‾12.62 mmol L–1 and the lowest mean value of sodium was observed at two month after parturition (137.69‾12.441 mmol L–1). Akhtar et al. (2010) reported that the mean serum sodium, potassium and chloride concentrations varied non-significantly during pregnancy and lactation. Abd Ellah et al. (2013) measured blood constituents during the period before and after 6 months of pregnancy in buffaloes. They revealed that serum chloride, iron and zinc levels were significantly higher before 6 months of pregnancy compared with those after.
Blood biochemical parameters: The lowered means of blood glucose concentrations in early stage of lactation were due to large amount of blood glucose withdrawal by the mammary gland for the synthesis of milk lactose. The hypoglycemia after parturition was attributed to heavy drain of glucose for lactose synthesis (Nale, 2003). Abdulkareem (2013) found that plasma glucose concentration remained steady at calving and postpartum periods. On the other hand, non-significant differences in glucose concentrations during calving and postpartum periods may explain the decline in maternal circulating insulin concentrations. The insulin level in the blood of cows is low during the initiation period of lactation (Accorsi et al., 2005). Mir et al. (2008) found that, the level of plasma glucose was significantly (p<0.05) lower during late and mid pregnancy when compared with early pregnancy. Glucose levels found in crossbred cattle ranged between 48.23‾12.08-54.00‾11.70 mg dL–1 which are close to the values observed by Singh et al. (2002) in two regimes of feeding. Elitok et al. (2006) reported exponent decrease in serum glucose concentration as the parturition approached in dairy cattle and the significant decrease in blood glucose level during late pregnancy signifies rapid utilization of glucose towards the end of the pregnancy. Cows generally go ketosis during third trimester (Mandali et al., 2002) and insufficient feed intake during the winter months may also result in the lower glucose levels in pregnant cows. Several workers have reported blood glucose level in cattle averaging 43.24‾10.56 mg dL–1 (Prudhvi Reddy et al., 2003), 49.05‾13.97 and 41.96‾12.37 mg dL–1 (Nath et al., 2004), respectively. Hagawane et al. (2009) found that the blood glucose level is regarded as one of the indicators of energy status in ruminants. The blood glucose was significantly higher in dry buffaloes (52.72‾14.22 mg dL–1) than the early and late lactating buffaloes (48.23‾13.44 mg dL–1).
The lowered trend in urea levels probably associated with the use of urea for protein synthesis on rumen-hepatic pathway due to compensation of the low protein uptake during the dry period (Yokus et al., 2006). Hagawane et al. (2009) found that the urea nitrogen values differ significantly (p<0.05) amongst different groups of lactating buffaloes. The mean blood urea value in early lactating stage (17.93‾10.79 mg dL–1) was significantly lowered as compared to the normal healthy control (26.76‾10.35 mg dL–1). They indicated that the increase in blood urea nitrogen levels in dry pregnant buffaloes in late pregnancy could be either due to increased deamination or increased protein intake (Hagawane et al., 2009). Several workers have reported that, serum urea in buffaloes is influenced by the days in milk, (Serdaru et al., 2011; Grasso et al., 2004) and by the diet. The values of serum blood urea are considered to be an indicator of total protein intake. These reports support the hypothesis that changes in blood urea content during lactation could depend on milk synthesis (El-Sherif and Assad, 2001).
Consistently high level of total protein has been seen by Yadav et al. (2006) in buffaloes as also observed in their study hence reinforcing the fact that high levels of plasma protein in the late trimester of pregnancy are needed for the optimum secretion of gonadotropin release factors and number of other hormones needed in the culmination of the pregnancy. Consistent with our finding, increasing pattern of serum total protein was also found in non-lactating yaks by Pouroucholtamane et al. (2005) and this phenomenon could be attributed to the non-lactating state of the cows in late trimester of the pregnancy. Changes in blood concentrations of glucose and proteins may all reflect alterations in liver function associated with fat accumulation during pregnancy (Reid and Robert, 1982). Hagawane et al. (2009) found that during early stage of lactation the serum total protein values (8.36‾10.47 g dL–1) was slightly elevated than normal healthy control (8.00‾10.57 g dL–1). This increase in total protein concentration following parturition might be attributed to the haemo-concentration and water losses occurred postpartum.
Serdaru et al. (2011) found that, the total protein concentration was 7.30‾10.95 and 5.53‾12.53 g dL–1, respectively in pregnant and lactating buffaloes. Mir et al. (2008) found that, significant (p<0.05) increase in total plasma protein content was observed during mid and late pregnancy when compared to the values observed in early pregnancy.
The lowered trend in total cholesterol and triglyceride levels were due to the buffaloes increased requirement for energy during lactation. Karapehlivan et al. (2007) found that triglycerides are used by mammary gland to form the milky fats and their request increase until the peak of lactation. Probably the high starting values found here are linked to a not yet high milk production; indeed at 30th day. When lactation comes to their, triglycerides show a remarkable decrease. The higher level of cholesterol with advancement of lactation was a physiological adjustment to meet the lactation requirements. The hormonal level of estrogen along with thyroxin played a vital role in reducing the cholesterol levels during pregnancy. Meliani et al. (2011) indicated that the mean values of plasma triglycerides were significantly higher in late pregnancy than in early pregnancy in mares. Serdaru et al. (2011) found that, the lactating group of buffaloes showed lower levels for total cholesterol (59.25‾110.14 mg dL–1) than the pregnant group (79.0‾19.56 mg dL–1). The descending trend in the serum cholesterol concentration in dry pregnant buffaloes compared to lactating buffaloes was observed (Nath et al., 2004, 2005).
Blood enzyme activity: Increase in ALT activity and AST in the blood of buffaloes in lactation indicated an increase in hepatic metabolism. Changes in activities of these enzymes may be related to reduced dry matter intake around parturition, may lead to hepatic lipidosis to alter the normal function of the liver (Greenfield et al., 2000). Serdaru et al. (2011) found that, serum enzymes (AST, ALT and AlP) were significantly lower in the lactating buffaloes groups. Abdulkareem (2013) found that in buffaloes, AST activity was numerically higher (96.06‾18.75-102.61‾111.62 Unit L–1) during postpartum periods in comparison with its value at calving (90.06‾18.04 Unit L–1) indicating that hepatic metabolism might be more stressed and tissue catabolism was more normal range pronounced during this period (Antunovic et al., 2004). However, because enzyme activities were within the normal range, integrity and functionality of liver tissue was obviously maintained during these periods. A similar trend was observed by Reist et al. (2003) for Holstein dairy cows, where AST activity was lower pre-partum and reached its peak during the first week post-partum. Plasma ALP activity exhibited distinct and gradual decreasing from calving to day 60 postpartum. This may be attributed to decreasing placental ALP activity in postpartum period, resulting in decrease plasma ALP activity. ALP normally produced by syncytiotrophoblast cells of placenta and may be involved in migration of primordial germ cell in developing fetus (Heller and Joshi, 2006). In pregnant cows, increase in ALP may be up 4 times normal during mid and late pregnancy, playing an important role in fetus musculature via transfer of phosphate (Fernandez and Kidney, 2007). Similar profiles were also demonstrated by Pizzuti and Salvatori (1993) in Italian buffaloes.
Blood hormones: In ruminants, the action of Growth Hormone (GH) on the mammary gland is through to be mediated mainly by the Insulin-like Growth Factor (IGF) signaling axis (Etherton, 2004). Treatment with exogenous GH increases the concentration of IGF-1 in the circulation (Lucy et al., 2001) which acts directly on the mammary gland (Taylor et al., 2004; Ehrhardt et al., 2000). When administered during mid-lactation, however, GH was associated with an increase in mammary cell proliferation in cows (Capuco et al., 2001). Because local production of IGF, as well as expression of IGF receptors are also physiologically regulated (Sinowatz et al., 2000; Plath-Gabler et al., 2001), this explains the differences in the response to GH across physiological states.
In animals, plasma leptin levels are closely correlated with nutritional status and body condition score (Chilliard et al., 2005). Serum leptin is sensitive to energy balance and is reduced during periods of negative energy balance in ewes (Tokuda et al., 2002). Lactation in many species is associated with marked loss energy through the milk which cannot be fully compensated by food intake (Macajova et al., 2004). During pregnancy, particularly in later pregnancy, plasma leptin levels were increased (Ingvartsen and Boisclair, 2001; Meikle et al., 2004; Block et al., 2003) and decreased sharply after delivery. This is in accordance with some trials with lactating cows (Block et al., 2001) when plasma leptin concentration were reduced by 50% after parturition. Chelikani et al. (2004) indicated that, postpartum IGF-I was a stronger predictor of circulating leptin thyroid hormones (T3 and T4) levels in ewes were determined by Karapehlivan et al. (2007). Lower blood T3 concentration could reduce the rate of oxidation and the rate of continuous breakdown and formation of protein and fat in the most, if not all mammary tissue (Riis and Madsen, 1985). Todini et al. (2007) showed that during gravity in all mammalian species thyroid activity and blood hormone levels were increased.
I express my gratitude to all employees of the Mehlet Moussa Experimental Station which belongs to Animal Production Research Institute, north part of the Nile Delta, Egypt. Also like to thank Research Park (CURP) the faculty of Agriculture, Cairo University for providing facilities and kind help during the experimental work.