ABSTRACT
The objective of this study was to study in detail about the endocrine cells of the camel duodenum. The duodenum is not only an important part of the gastrointestinal tract, but also works as endocrine portion by secreting some hormones that play key roles in the regulation of certain organs. Specimens from eleven dromedarian camels (Camelus dromedarius) of both sexes with age ranging from 2-12 years were examined. The immunohistochemistry was performed using five types of hormones. Gastrin showed high immunoreactivity at the endocrine of tunica mucosa and duodenal glands. Somatostatin showed very high immunoreactivity at endocrine of tunica mucosa and duodenal gland. The performance of insulin, which was done for the first time on the duodenum of camel, showed low immunoreactivity at endocrine cells of tunica mucosa and duodenal glands. Serotonin showed high immunoreactivity at the enteroendocrine cells of the tunica mucosa and few cells that produced serotonin at duodenal glands. Glucagon showed moderate to low immunoreactivity at the endocrine of tunica mucosa and duodenal glands. In conclusion, the study results found that the duodenum of the camel has distinctive characters immunohistochemically. Therefore, further physiological and experimental studies are required.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajava.2012.790.801
URL: https://scialert.net/abstract/?doi=ajava.2012.790.801
INTRODUCTION
The dromedary camel (Camelus dromedarius) is mostly present in the tropical area (Africa and Asia) and known as the ship of the desert in arabian countries. The need of animal proteins directs the attention to the meat and milk production as a better food provider in desert and semi desert area (Sweet, 1965; Azwai et al., 1996). The camel is also used in carrying goods during transportation and travel. The camel is ruminant, but its stomach differs morphologically from that of other ruminants (Eerdunchaolu et al., 1999). The duodenum is not only an important part of the gastrointestinal tract but also works as endocrine portion by secreting some hormones that play roles in the regulation of some organs. For example, somatostatin was involved in the regulation of pancreatic secretion (Young and Heath, 2000). The duodenum is responsible for further processing of the material from the stomach by secreting enzymes which is vital for digestion; it also mixes the digesta with these enzymes within its lumen (Weisbradt, 1987; Guyton, 1991; Dellmann and Eurell, 1998).
Morphological studies at light and electron microscopic level have demonstrated the presence and distribution of hormone-producing cells in the gastrointestinal tract of man and domestic mammals (Facer et al., 1985; Rindi et al., 1986; Kawakita et al., 1990). Gastrointestinal endocrine cells dispersed in the mucosa of digestive tract synthesize various types of gastrointestinal hormones which played an important role in the physiological function of alimentary tract such as nutrient absorption, the secretion of intestinal and associated glands, gut motility and increase intestinal blood flow (Bell, 1979; Deveney and Way, 1983; Huang and Wu, 2005). The endocrine cells of gastrointestinal tract are generally divided into two types, the open type in which the apex of the cell presents microvilli and contact the lumen "found in the epithelial lining of the lumen" and the close type in which the cellular apex is covered by other epithelial cells "found in the intestinal gland region" (Junqueira et al., 1995; Ham, 2002). Moreover the regional distributions and relative frequencies of these endocrine cells vary according to the animal species and feeding habits (Solcia et al., 1975). Many investigators reported that the gastrointestinal tract is the largest endocrine gland in the body in term of both number of hormones and number of endocrine cells (Walsh, 1987; Rehfeld, 1998; Ahlman and Nilsson, 2001). The involvement of these peptide hormones in many of the gastrointestinal function are well established, e.g. secretin and cholecytokinin were found to be involved in regulation of pancreatic secretion, acid secretion, gut motility and gastro-protection (Chey and Chang, 2003; Schmidt et al., 2004; West and Mercer, 2004). The neural and hormonal systems regulating the gastrointestinal function are anatomically distinct but they cannot be considered functionally distinct, in that they interact closely. For example, gastric acid secretion is stimulated by both gastrin and acetylcholine (Soll, 1989); where as it is inhibited by another gastrointestinal hormone which also inhibits release of acetylcholine from enteric neurons (Yamada and Chiba, 1989). The neuroendocrine cells dispersed among the epithelial cells of gastrointestinal tract, together with enteric nervous system play a vital role in the function of the digestive system (Ali et al., 2007).
Previously many immunohistochemical studies were carried on the distribution and frequency of endocrine cells in the gastrointestinal tract of humans, cattle, pigs, lesser mouse deer, sheep, horse, water buffalo, babirusa and camel (Cristina et al., 1978; Calingasan et al., 1984; Kitamura et al., 1984; Ito et al., 1987; Agungpriyono et al., 1994; Carla et al., 1999; Agungpriyono et al., 2000; Eerdunchaolu et al., 2001; Ham, 2002; Ali et al., 2007). Gastrointestinal hormones as regulatory peptides appear to be the major component of body integration and have an important regulatory action on physiological function of the gastrointestinal tract (Solcia et al., 2000; Larsson, 2000).
Gastrin is an important gastrointestinal hormone secreted from neuroendocrine cells located in the antrum of the stomach and proximal duodenum "G cells" (Schubert, 2007). Only G-cells in gastric antrum are regulated by changes in gastric pH, fasting and re-feeding (Brand and Stone, 1988; Wu et al., 1991). Gastrin normally regulates gastric acid secretion by stimulating the proliferation of enterochromaffin-like cells and release of histamine (Junqueira et al., 1995; Mensah-Osman et al., 2008). It has been reported that, an increase in gastrin expression was presumably due to reduce somatostatin expression (Sumii et al., 1994; Park et al., 1999). Previous study showed that modulation of tissue somatostatin level is the most effective regulator of gastrin secretion (Zavros et al., 2003). The level of gastrin secreted by G cells in human duodenum is constitutively low (Brand and Fuller, 1988). The gastrin is highly expressed by pyloric region and duodenum in calves than in adult cow (Kitamura et al., 1985). In the two-humped camel, the gastrin is expressed by pyloric gland and in the duodenum whiles no such expression in other region of gastrointestinal tract except very low expression in the jejunum (Eerdunchaolu et al., 2001). Gastrin was found highly expressed by pyloric region and moderately expressed by duodenum of babirusa (Agungpriyono et al., 2000).
Somatostatin is synthesized in D-cells of pancreas and in endocrine of duodenum (Junqueira et al., 1995; Dellmann and Eurell, 1998; Ali et al., 2007). It was isolated from the hypothalamus of sheep for first time and it has an inhibitory action on secretion of pituitary growth hormones (Brazeau et al., 1973), gastrin (Junqueira et al., 1995), insulin and glucagon (Dellmann and Eurell, 1998). Somatostatin thought to play a role in controlling the secretion of hormones from endocrine cells (Kusumoto et al., 1979). Also it has been shown to inhibit fluid secretion from the gut (Dharmsathaphorn et al., 1980). Somatostatin was found in all parts of gastrointestinal tract of calf and adult cow (Kitamura et al., 1985), sheep (Vergara-Esteras et al., 1990), babirusa (Agungpriyono et al., 2000) and in the two-humped camel, except the rectum (Eerdunchaolu et al., 2001).
Insulin promotes the uptake of glucose by most cells, particularly those of liver, skeletal muscle and adipose tissue, thus lowering plasma glucose concentration (Young and Heath, 2000). The presence of insulin receptors has been shown on the epithelial cells of the gastrointestinal tact in rat (Bergeron et al., 1980) and on the mucosal surface of cells in gastrointestinal tract in rabbit (Pillion et al., 1985). Recent study showed that among the various products of enteroendocrine cells, the human duodenum has the incretin glucose-like peptide-1 (GLP-1 secreted by L-cells) and glucose-dependent insulinotropic peptide (GLP secreted by K-cells). These are key modulators of insulin secretion (Theodorakis et al., 2006). There are many studies on insulin expression in pancreas of different domestic animal but there are little studies on insulin in gastrointestinal tract. A recent study showed no expression of insulin in gastrointestinal tract of reptiles (Huang and Wu, 2005).
Serotonin (5-hydroytryptamine) consists of monoamines and is widely distributed in the nervous system and gastro-enteropancreatic endocrine cells (El-Salhy et al., 1985). It is secreted from the fundus of stomach (Junqueira et al., 1995). It has been also reported that the serotonin is secreted from the enterochromaffin which is a type of enteroendocrine cells (Young and Heath, 2000). Serotonin is known to stimulate strongly the smooth musculature of the gut and secretion of exocrine glands (Furness and Costa, 1982). It is implicated in controlling, inhibition and facilitation of motor function of the esophagus, stomach, small intestine, ileocolonic sphincter and colon, as well as modulating small intestine and colonic secretion (Bulbring and Gershon, 1967; Siriwardena et al., 1991). It is also implicated in the transport of fluid from the intestinal epithelium to the lumen (Munck et al., 1994). It has been reported that the serotonin inhibits gastrin secretion (Guyton, 1988). Serotonin has been found in all parts of gastrointestinal tract of the rat, mice (Sandstrom and El-Salhy, 2000), two-humped camel with high level in pyloric region and small intestine (Eerdunchaolu et al., 2001), babirusa with low level in large intestine (Agungpriyono et al., 2000) and one-humped camel with high level in duodenum, jejunum and colon (Ali et al., 2007).
Glucagon is secreted from pancreatic A-cells (Dellmann and Eurell, 1998). It plays an important role in carbohydrate metabolisms that oppose the action of insulin (Young and Heath, 2000) by breaking the glycogen in liver then increase the blood glucose level (Dellmann and Eurell, 1998). The glucagon was absent in most parts of gastrointestinal tract in the two-humped camel except very low in duodenum (Eerdunchaolu et al., 2001). It has low expression in gastrointestinal tract of babirusa (Agungpriyono et al., 2000) and calf while it is moderate in intestine and absent in other parts of gastrointestinal tract of adult cow (Kitamura et al., 1985).
Studies on the endocrine cells of the duodenum of the one-humped camel (Camelus dromedaries) are virtually lacking. Therefore, the present study was undertaken to study the endocrine cells type of the duodenum of the camel and the pattern of their distribution in the different parts of the duodenum submucosa using immunohistochemistry techniques. as well as discus the information that will be obtained in the light of what is known about the duodenum of other domestic animals.
MATERIAL AND METHODS
Selection of animals: Eleven dromedary camels (Camelus dromedarius) of both sexes were selected from Al-Ahsa slaughterhouse and Camel Research Center, King Faisal University, College of Veterinary Medicine and Animal Resources for study. The animals and specimens were apparently healthy and free from gross pathological changes. The animals age ranged between 2-12 years (Ramadan, 1994). The study was carried during 2009-2010 period.
Small segments of duodenum were collected from eleven adult camels, immediately after slaughtering at Al-Ahsa slaughterhouse or in the Department of Anatomy, College of Veterinary Medicine and Animal Resources, King Faisal University. The samples were collected from the proximal and distal parts of the ampulla of duodenum. While the samples from the thin part were collected from five regions i.e., proximal, proxo-middle, middle, mid-distal and distal. The samples were fixed in 4% paraformaldehyde in PBS for 30-40 h at room temperature for immunohistochemical technique.
The samples were trimmed to small pieces about 1.0 cm in length and processed for immunohistochemical techniques. After fixation, the specimens were dehydrated in ascending grades of alcohol, cleared in xylene and embedded in paraffin wax as blocks.
Serial sections were taken from each block at 5-7 μm with leica rotary microtome, made in Germany (RM 2135). The sections were floated on warm water bath (41°C). The sections were mounted onto gelatin coated glass slides for immunohistochemical techniques.
Sections for immunohistochemical techniques were cleared in xylene, rehydrated in descending grades of alcohol and then washed in Phosphate Buffered Saline (PBS). The sections were then processed according to the procedure included with the kit provided by Dako Cytomation EnVision+Dual Link System-HRP (DAB+). Stained slides were examined under light microscope (Olympus BX51), made in Japan.
RESULTS AND DISCUSSION
The endocrine cells were dispersed in the tunica mucosa and tunica sub-mucosa of the camel duodenum which synthesize various types of hormones. The main endocrine cells were enteroendocrine cells, intestinal glands and duodenal glands.
The immunohistochemistry was carried on the different parts of camel duodenum using 5 types of hormones which were gastrin, somatostatin, insulin, serotonin and glucagon (Table 1).
Gastrin immunoreactivity cells (G cells) were found at endocrine cells of the tunica mucosa and duodenal glands of the tunica submucosa (Fig. 1). High intensity of immunoreactivity of duodenal glands of tunica submucosa was shown in all parts of the duodenum. However, the immunoreactivity was not shown at the endocrine cells of the tunica mucosa in some parts of the duodenum (Fig. 2).
Somatostatin immunoreactivity cells were found in endocrine cells of the tunica mucosa and duodenal glands of the tunica sub-mucosa (Fig. 3). Avery high intensity of immunoreactivity was shown in all parts of the duodenum. However, the immunoreactivity was not shown at the endocrine cell of the tunica mucosa in some parts of the duodenum.
| Table 1: | Results of endocrine immunoreactivity |
 | |
| +: Low positive, +++: High positive, ++++: Very high positive | |
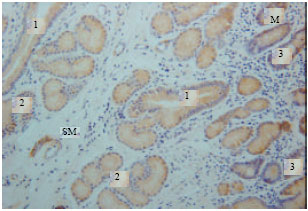 | |
| Fig. 1: | Gastrin immunoreactivity in the tunica mucosa and submucosa. M: Tunica mucosa, SM: Tunica submucosa, 1: Duct connecting the duodenal gland to the lumen, 2: Duodenal gland, 3: Endocrine cells |
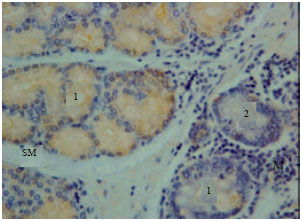 | |
| Fig. 2: | Gastrin immunoreactivity in the tunica mucosa and sub mucosa. M: Tunica mucosa, SM: Tunica submucosa, 1: Duodenal gland, 2: Endocrine cells |
Insulin immunoreactivity cells were found in some endocrine cells of the tunica mucosa and some duodenal glands of the tunica sub-mucosa (Fig. 4). Low immunoreactivity was shown in some duodenal glands and endocrine cells of the tunica mucosa. The immunoreactivity was not shown in all parts of the duodenum (Fig. 5).
Serotonin immunoreactivity cells were found in endocrine cells of the tunica mucosa (enteroendocrine cells) and in a very few endocrine cells of the duodenal glands.
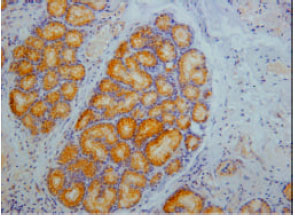 | |
| Fig. 3: | Somatostatin immunoreactivity in the duodenal gland of the tunica submucosa |
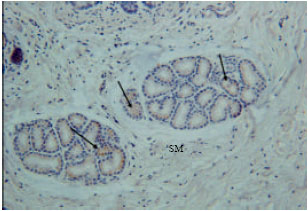 | |
| Fig. 4: | Insulin immunoreactivity at tunica submucosa (SM). Arrows are showing the insulin immunoreactivity at duodenal glands |
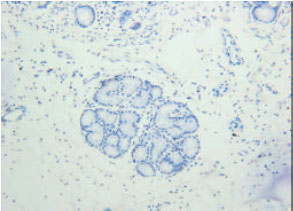 | |
| Fig. 5: | No insulin immunoreactivity at the duodenal glands of tunica submucosa |
However, the immunoreactivity cells were not shown in most duodenal glands (Fig. 6). The immunoreactivity was high and abundant at the endocrine cells of the tunica mucosa and the duct which connects the duodenal glands to the lumen of duodenum (Fig. 7). The immunoreactivity was shown in all parts of the duodenum.
Glucagon immunoreactivity cells were found at endocrine cells of tunica mucosa and duodenal glands of the tunica sub-mucosa. However, the immunoreactivity was low in all parts of the duodenum with moderate background (Fig. 8).
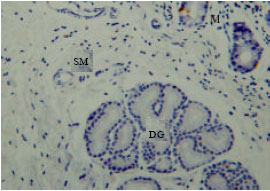 | |
| Fig. 6: | Serotonin immunoreactivity at M: Tunica mucosa MS: and tunica submucosa DG: Duodenal gland |
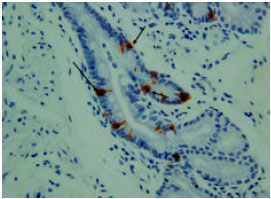 | |
| Fig. 7: | Serotonin immunoreactivity at the duct which connecting the duodenal gland to the lumen of the duodenum |
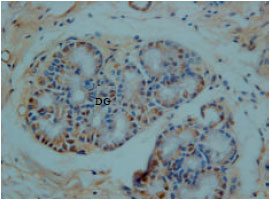 | |
| Fig. 8: | Glucagon immunoreactivity at DG Duodenal gland of tunica submucosa |
DISCUSSION
It is generally accepted that the endocrine cells in the alimentary tract differs remarkably between animal species in term of regional distribution, relative frequency, cell type and each regional part of the gastrointestinal tract (Ham, 2002). This study clarified the immunohistochemical types, distribution and frequency of the endocrine cells along the duodenum of dromedary camel. In the present study, five types of endocrine cells were detected in the duodenum of camel. Some studies stated that the endocrine cells in the gastrointestinal tract of humans, cattle and two-humped camel were numerously distributed at the pyloric gland region and duodenum (Alumets et al., 1977; Kitamura et al., 1985; Eerdunchaolu et al., 2001).
The endocrine cells were dispersed in the tunica mucosa and tunica submucosa of the camel duodenum which synthesizing various types of hormones. The main endocrine cells were enteroendocrine, intestinal glands and duodenal glands. Many researchers reported that some types of endocrine cells were present in the duodenal glands of pig (Ito et al., 1987), cat (Kitamura et al., 1982), horse (Kitamura et al., 1984), cattle (Kitamura et al., 1985), sheep (Calingasan et al., 1984), lesser mouse deer (Agungpriyono et al., 1994), mink (Kawano et al., 1983) and babirusa (Agungpriyono et al., 2000).
Gastrin, somatostatin, insulin, serotonin and glucagon endocrine cells were detected in the duodenum of the camel by light microscopic immunohistochemistry. Similar findings concluded that somatostatin and serotonin have been demonstrated on dromedary camel (Ali et al., 2007). While gastrin, glucagon, somatostatin and serotonin were reported in the two-humped camel (Eerdunchaolu et al., 2001).
Gastrin immunoreactivity cells (G cell) were detected in the duodenum of the camel with high level of expression at duodenal gland along the duodenum. While at the tunica mucosa, endocrine cells were not shown in some areas. Similar findings were reported in the two-humped camel, the calf and adult cow (Eerdunchaolu et al., 2001; Kitamura et al., 1984).
Somatostatin immunoreactivity cells were detected in the duodenum of the camel with very high level of expression at the duodenal glands along the duodenum. While at tunica mucosa, the endocrine cells were not shown in some areas. Similar findings were reported in calf, adult cow, sheep, the two-humped camel and babirusa (Kitamura et al., 1984; Agungpriyono et al., 2000; Eerdunchaolu et al., 2001; Ali et al., 2007). It has been reported that, somatostatin played a role in controlling the secretion of hormones from endocrine cells (Kusumoto et al., 1979). Therefore the presence of somatostatin in one-humped camel may help in the conservation of fluid in this desert animal (Ali et al., 2007) and controlling the secretion of hormones.
Insulin immunoreactivity cells were detected in some duodenal glands and endocrine cells of the tunica mucosa along the duodenum of the camel with low expression. No similar findings were reported in the literature for comparison or contradiction. However, The presence of insulin in the camel duodenum requires further study to explain its role in this region.
Serotonin immunoreactivity cells were detected at enteroendocrine cells of the tunica mucosa and on a very few cells of the duodenal glands with high level of expression especially at the tunica mucosa endocrine cells. Similar results were reported on presence of serotonin with high level of expression at the duodenum in two-humped camel and one-humped camel (Eerdunchaolu et al., 2001; Ali et al., 2007).
Glucagon immunoreactivity cells were detected at endocrine cells of the tunica mucosa and duodenal gland with moderate expression. The study findings agree with those of Eerdunchaolu et al. (2001) who reported low expression of glucagon in duodenum of the two-humped camel, low level of expression in gastrointestinal tract of babirusa (Agungpriyono et al., 2000) and calf while it was moderate in the intestine of adult cow (Kitamura et al., 1984). Therefore the presence of the glucagon is beneficial for the metabolism of the carbohydrate in the duodenum of camel. The heterogeneity and concentration of the endocrine cells in the duodenum may relate to regulation of the secretion of pancreatic juice and bile as well as to control of the function of the stomach and small intestine (Kitamura et al., 1982; Kitamura et al., 1985; Krause et al., 1985). Also many studies seem to suggest that dietary changes can affect the regional distribution of enteroendocrine cells (Sharma and Schumacher, 1996).
CONCLUSIONS
This study reported the distribution of five types of endocrine cells in the duodenum of the camel which showed very high level of somatostatin, high level of serotonin and gastrin coupled with low level of glucagon. However, low level of insulin endocrine cells was reported for the first time in the duodenum of the camel. The results support the important digestive role of endocrine cells in the duodenum of the camel and water preservation. The immunohistochemical findings require further physiological and experimental studies to elucidate the presence, distribution and function of the duodenum endocrine cells in this unique animal (dromedary camel).
ACKNOWLEDGMENTS
The authors take this opportunity to thank The Director, Camel Research Center and his staff members for providing the experimental animals.
REFERENCES
- Agungpriyono, S., A.A. Macdonald, K.Y. Leus, N. Kitamura and I.K. Adnyane et al., 2000. Immunohistochemical study on the distribution of endocrine cells in the gastrointestinal tract of the babirusa, Babyrousa babyrussa (Suidae). Anat. Histol. Embryol., 29: 173-178.
CrossRefPubMedDirect Link - Agungpriyono, S., J. Yamada, N. Kitamura, Y. Yamamoto, N. Said, K. Sigit and T. Yamasita, 1994. Immunohistochemical study of the distribution of endocrine cells in the gastrointestinal tract of the lesser mouse deer (Tragulus javanicus). Acta. Anat., 15: 232-238.
PubMedDirect Link - Ali, M.A., F. Nyberg, S.I. Chandranath, S. Dhanasekaran and S. Tariq et al., 2007. Distribution of neuroendocrine cells in the small and large intestines of the one-humped camel (Camelus dromedarius). Neuropeptides, 41: 293-299.
CrossRef - Alumets J., F. Sundler and R. Hakanson, 1977. Distribution, ontogeny and ultrastructure of somatostatin immunoreactive cells in the pancreas and gut. Cell Tissue Res., 185: 465-479.
CrossRefDirect Link - Azwai, S.M., S.D. Carter, Z. Woldehiwet and U. Wernery, 1996. Serology of Orthopoxvirus cameli infection in dromedary camels: Analysis by ELISA and western blotting. Comp. Immunol. Microbial. Infect. Dis., 19: 65-78.
CrossRefDirect Link - Bell, F.R., 1979. The relevance of the new knowledge of gastrointestinal hormones to veterinary science. Vet. Res. Commun., 2: 305-314.
CrossRef - Bergeron, J.J., R. Rachubinski, N. Searle, D. Borts, R. Sikstrom and B.I. Posner, 1980. Polypeptide hormone receptors in vivo: Demonstration of insulin binding to adrenal gland and gastrointestinal epithelium by quantitative radioautography. J. Histochem. Cytochem., 28: 824-835.
PubMedDirect Link - Brand, S.J. and P.J. Fuller, 1988. Differential gastrin gene expression in rat gastrointestinal tract and pancreas during neonatal development. J. Biol. Chem., 263: 5341-5347.
Direct Link - Brand, S.J. and D. Stone, 1988. Reciprocal regulation of antral gastrin and somatostatin gene expression by omeprazole-induced achlorhydria. J. Clin. Invest., 82: 1059-1066.
CrossRefDirect Link - Brazeau, P., W. Vale, R. Burgus, N. Ling, M. Butcher, J. Rivier and R. Guillemin, 1973. Hypothalamic polypeptide that inhibits the secretion of immunoreactive pituitary growth hormone. Science, 179: 77-79.
CrossRefPubMedDirect Link - Bulbring, E. and M.D. Gershon, 1967. 5-hydroxytryptamine participation in the vagal inhibitory innervation of the stomach. J. Physiol., 192: 823-846.
PubMedDirect Link - Calingasan, N.Y., N. Kitamura, J. Yamada, Y. Oomori and T. Yamashita, 1984. Immunocytochemical study of the gastroenteropancreatic endocrine cells of the sheep. Acta. Anat., 118: 171-180.
PubMedDirect Link - Carla, L., P. De Girolamo, L. Coppola, G. Paino and L. Castaldo, 1999. Postnatal development of intestinal endocrine cell populations in the water buffalo. J. Anat., 195: 439-446.
CrossRefPubMedDirect Link - Chey, W.Y. and T.M. Chang, 2003. Secretin, 100 years later. J. Gastroenterol., 38: 1025-1035.
CrossRefPubMedDirect Link - Cristina, M.L., T. Lehy, P. Zeitoun and F. Dufougeray, 1978. Fine structural classification and comparative distribution of endocrine cells in normal human large intestine. Gastroenterology, 75: 20-28.
PubMedDirect Link - Dharmsathaphorn, K., R.S. Sherwin and J.W. Dobbins, 1980. Somatostatin inhibits fluid secretion in the rat jejunum. Gastroenterology, 78: 1554-1558.
PubMedDirect Link - Mensah-Osman, E., E. Labut, Y. Zavros, M. El-Zaatari, D.J. Law and J.L. Merchant, 2008. Regulated expression of the human gastrin gene in mice. Regul. Pept., 151: 115-122.
CrossRefDirect Link - Eerdunchaolu, D.V.M., K. Takehana, A. Kobayashi, J. Yamada and H. Ueda et al., 2001. Immunohistochemical study of the distribution of endocrine cells in the gastrointestinal tract of the camel (Camelus bactrianus). Eur. J. Morphol., 39: 57-63.
PubMedDirect Link - Eerdunchaolu, K. Takehana, A. Kobayashi, Baiyin and G.F. Cao et al., 1999. Morphological characterization of gland cells of the glandular sac area in the complex stomach of the bactrian camel (Camelus bactrianus). Anatomia Histol. Embryol., 28: 183-191.
CrossRefPubMedDirect Link - El-Salhy, M., E. Winder and M. Lundqvist, 1985. Comparative studies of serotonin-like immunoreactive cells in the digestive tract of vertebrates. Biomed. Res., 6: 371-375.
Direct Link - Facer, P., A.E. Bishop, R.V. L loyd, B.S. Wilson, R.J. Hennessy and J.M. Polak, 1985. Chromogranin: A newly recognized marker for endocrine cells of the human gastrointestinal tract. Gastroenterology, 89: 1366-1373.
PubMed - Kawakita, S., K. Takatsuki, M. Tsukada, M. Yoneda, T. Takano, A. Kawakubo and H. Nagura, 1990. Localization of chromogranin A-immunoreactivity in bovine gastrointestinal endocrine cells with special reference to Grimelius silver stain. Endocrinol. Jpn., 37: 299-308.
CrossRefPubMedDirect Link - Kawano, H., T. Yamashita, J. Yamada and N. Kitamura, 1983. A light microscopic study of the gastro-entero-pancreatic endocrine cells of the mink (Mustela vison). Arch. Histol. Jpn., 46: 559-573.
PubMedDirect Link - Kitamura, N., J. Yamada, N.Y. Calingasan and T. Yamashit, 1984. Immunocytochemical distribution of endocrine cells in the gastrointestinal tract of the horse. Equine Vet. J., 16: 103-107.
PubMedDirect Link - Kitamura, N., J. Yamada, N.Y. Calingasan and T. Yamashita, 1985. Histologic and immunocytochemical study of endocrine cells in the gastrointestinal tract of the cow and calf. Am. J. Vet. Res., 46: 1381-1386.
PubMed - Krause, W.J., J. Yamada and J.H. Cutts, 1985. Quantitative distribution of enteroendocrine cells in the gastrointestinal tract of the adult opossum, Didelphis virginiana. J. Anat., 140: 591-605.
PubMed - Kusumoto, Y., T. Iwanaga, S. Ito and T. Fujita, 1979. Juxtaposition of somatostatin cell and parietal cell in the dog stomach. Arch. Histol. Jpn., 42: 459-465.
PubMedDirect Link - Larsson, L.I., 2000. Developmental biology of gastrin and somatostatin cells in the antropyloric mucosa of the stomach. Microscopy Res. Tech., 48: 272-281.
CrossRefPubMedDirect Link - Theodorakis, M.J., O. Carlson, S. Michopoulos, M.E. Doyle, M. Juhaszova, K. Petraki and J.M. Egan, 2006. Human duodenal enteroendocrine cells: Source of both incretin peptides, GLP-1 and GIP. Am. J. Physiol. Endocrinol. Metab., 290: E550-E559.
CrossRefDirect Link - Munck, L.K., O. Eskerod, M.B. Hansen, K. Bukhave and J. Rask-Madsen, 1994. Failure of tropisetron to inhibit jejunal water and electrolyte secretion induced by 5-hydroxytryptamine in healthy volunteers. Gut, 35: 637-640.
CrossRefDirect Link - Ahlman, H. and O. Nilsson, 2001. The gut as the largest endocrine organ in the body. Ann. Oncol., 12: S63-S68.
CrossRefPubMedDirect Link - Park, S.M., H.R. Lee, J.G. Kim, J.W. Park and G. Jung et al., 1999. Effect of Helicobacter pylori infection on antral gastrin and somatostatin cells and on serum gastrin concentrations. Korean J. Internal Med., 14: 15-20.
PubMedDirect Link - Vergara-Esteras, P., F.A. Harrison and D. Brown, 1990. The localization of somatostatin-like immunoreactivity in the alimentary tract of the sheep with observations on the effect of an infection with the parasite haemonchus contortus. Exp. Physiol., 75: 779-789.
PubMedDirect Link - Pillion, D.J., V. Ganapathy and F.H. Leibach, 1985. Identification of insulin receptors on the mucosal surface of colon epithelial cells. J. Biol. Chem., 260: 5244-5247.
PubMedDirect Link - Rehfeld, J.F., 1998. The new biology of gastrointestinal hormones. Physiol. Rev., 78: 1087-1108.
Direct Link - Rindi, G., R. Buffa, F. Sessa, O. Tortora and E. Solcia, 1986. Chromogranin A, B and C immunoreactivities of mammalian endocrine cells. Distribution, distinction from costored hormones/prohormones and relationship with the argyrophil component of secretory granules. Histochemistry, 85: 19-28.
PubMed - Sandstrom, O. and M. El-Salhy, 2000. Duodenal endocrine cells in mice with particular regard to age-induced changes. Histol. Histopathol., 15: 347-353.
PubMedDirect Link - Schmidt, P.T., L. Hansen, L. Hilsted and J.J. Holst, 2004. Cholecystokinin inhibits gastrin secretion independently of paracrine somatostatin secretion in the pig. Scand. J. Gastroenterol., 39: 217-221.
CrossRefDirect Link - Sharma, R. and U. Schumacher, 1996. The diet and gut microflora influence the distribution of enteroendocrine cells in the rat intestine. Cell. Mol. Life Sci., 52: 664-670.
CrossRefDirect Link - Siriwardena, A., C. Booker, J. Pratt and J. M. Kellum, 1991. Pathways of serotonin-induced electrolyte transport in rat distal colon. Surgery, 110: 411-418.
PubMedDirect Link - Solcia, E., C. Capella, G. Vassallo and R. Buffa, 1975. Endocrine cells of the gastric mucosa. Int. Rev. Cytol., 42: 223-286.
PubMed - Solcia, E., G. Rindi, R. Buffa, R. Fiocca and C. Capella, 2000. Gastric endocrine cells: Types, function and growth. Regul. Pept., 93: 31-35.
CrossRefDirect Link - Sumii, M., K. Sumii, A. Tari, H. Kawaguchi and G. Yamamoto et al., 1994. Expression of antral gastrin and somatostatin mRNA in Helicobacter pylori-infected subjects. Am. J. Gastroenterol., 89: 1515-1519.
PubMedDirect Link - Sweet, L.E., 1965. Camel raiding of North Arabian Bedouin: A mechanism of ecological adapt. Am. Anthropol., 67: 1132-1150.
Direct Link - Ham, T.S., 2002. Regional distribution and relative frequency of gastrointestinal endocrine cells in large intestines of C57BL/6 mice. J. Vet. Sci., 3: 233-238.
PubMedDirect Link - West, S.D. and D.W. Mercer, 2004. Cholecystokinin-induced gastroprotection: A review of current protective mechanisms. Digestive Dis. Sci., 49: 361-369.
CrossRefPubMedDirect Link - Wu, V., K. Sumii, A. Tari, M. Sumii and J.H. Walsh, 1991. Regulation of rat antral gastrin and somatostatin gene expression during starvation and after refeeding. Gastroenterology, 101: 1552-1558.
PubMedDirect Link - Huang, X.G. and X.B. Wu, 2005. Immunohistochemical study on gastrointestinal endocrine cells of four reptiles. World J. Gastroenterol., 11: 5498-5505.
Direct Link - Zavros, Y., S. Rathinavelu, J.Y. Kao, A. Todisco and J. DelValle et al., 2003. Treatment of Helicobacter gastritis with IL-4 requires somatostatin. Proc. Natl. Acad. Sci. USA., 100: 12944-12949.
CrossRefPubMedDirect Link








