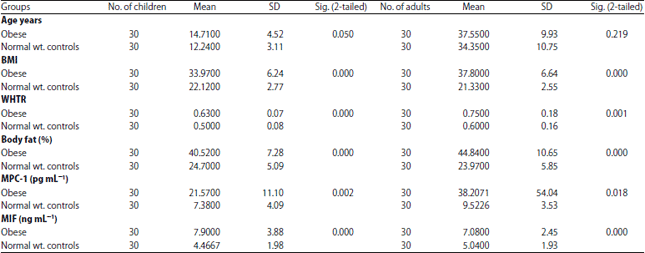
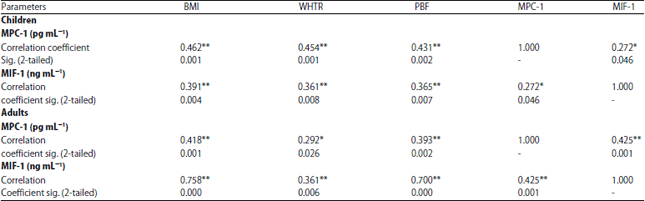
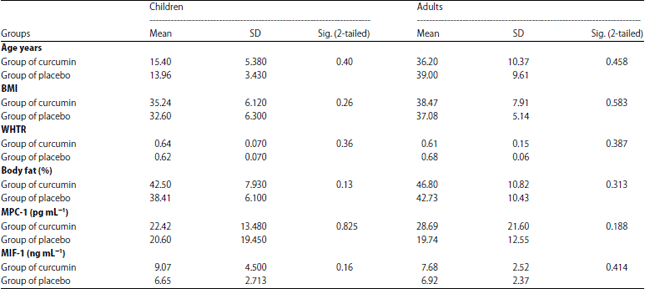
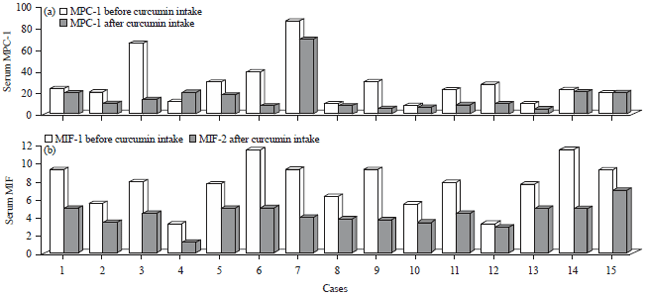
Research Article
Curcumin Intake Could Lower Serum Macrophage Migration Inhibitory Factor and Monocyte Chemoattractant Protein-1 Levels in Obese Subjects
Department of Pediatrics, National Research Centre (NRC), Cairo, Egypt
LiveDNA: 20.2028
Soha M. Abd El Dayem
Department of Pediatrics, National Research Centre (NRC), Cairo, Egypt
Mona Hamed
Department of Clinical Pathology, National Research Centre (NRC), Cairo, Egypt
Dina F. Ayoub
Department of Clinical Pathology, National Research Centre (NRC), Cairo, Egypt
Eman Mahmoud Hassan
Department of Clinical Pathology, National Research Centre (NRC), Cairo, Egypt
Heba Assal
Department of Internal Medicine, National Research Centre (NRC), Cairo, Egypt