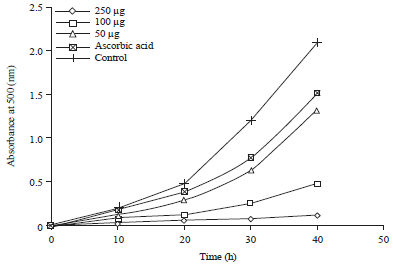
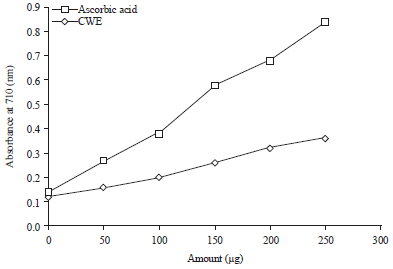
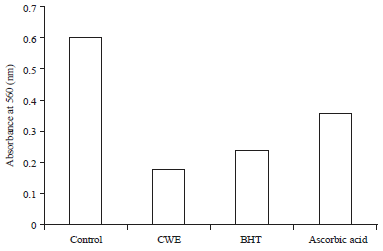
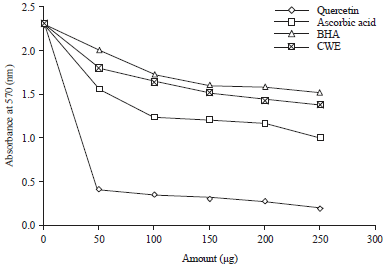
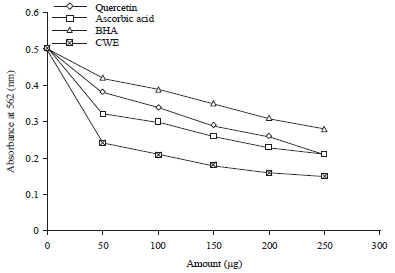
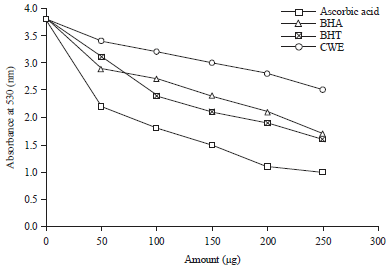
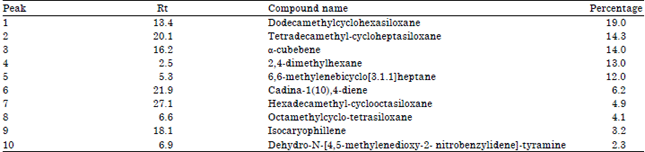
Research Article
In vitro Antioxidant Activities and Antimicrobial Efficacy of Asian Snakewood; Colubrina asiatica (L.) Brong.
Department of Botany, Shri Pancham Khemraj Mahavidyalaya, Sawantwadi, MS, India
U.L. Dethe
Department of Botany, Shri Pancham Khemraj Mahavidyalaya, Sawantwadi, MS, India
D.K. Gaikwad
Department of Botany, Shivaji University, Kolhapur, India