Mini Review
Antimicrobial Activity of Plant Secondary Metabolites: A Review
Texas A and M International University, Laredo, Texas, 78041, USA
R.A. Ynalvez
Texas A and M International University, Laredo, Texas, 78041, USA
There is increased prevalence of antibiotic resistant bacteria emerging from the extensive use of antibiotics. This renders current antimicrobial agents insufficient to control at least some bacterial infections (Riffel et al., 2002). Plants are used for the treatment of diseases and novel drugs continue to be developed through research from plants. There are more than 20,000 plant species used in traditional medicines and they are prospective reservoirs for new pharmaceuticals. Traditional medicinal plants have received significant attention because their phytochemicals may lead to new drug discoveries (Amor et al., 2009).
For several years, there has been an increased interest in the study of different extracts obtained from traditional medicinal plants as potential sources of new antimicrobial agents. The search for new antimicrobial agents is an important line of research because of the resistance acquired by several pathogenic microorganisms (Hossain et al., 2012; Riffel et al., 2002). From previous research studies, several secondary metabolites have been revealed as potential antibacterial agents; therefore, this study will discuss the different secondary metabolites produced by plants and their potential antibacterial activities.
In many developing countries, plant materials have been known to be used as traditional medicine. There is the lack of healthcare in these countries. Thus plants become readily accessible and economical sources available for the treatment of many infections and diseases. In recent years, medicinal plants have started to increase its appeal to pharmaceutical companies and the scientific research community. This was due to evidences that these plant-derived compounds have a potential for many biological activities which include antimicrobial activity (Savoia, 2012). Scientists’ aim is to extract and characterize active phytocompounds found in plants, since they have provided high activity profile drugs (Vaghasiya et al., 2011).
Ethnopharmacology has recently become an area of interest because of the use of these medicinal plants against infectious diseases. In this study, the raw materials from a plant are collected and further analyzed for the bioactive constituents exhibiting the biological activity. Thus in the future, they can be used as pharmaceutical drugs (Savoia, 2012). These natural products are chemical compounds derived from any living organism, including plants. Primary metabolites are necessary compounds produced by the plant for growth and metabolism; they include carbohydrates, proteins and lipids. On the other hand, secondary metabolites are compounds that are products of primary metabolism and are known for plant defense mechanisms (Ramawat, 2007). Drugs derived from natural products are usually secondary metabolites and their derivatives. Phytochemical screening of different plants has revealed numerous bioactive compounds including alkaloids, tannins, flavonoids, glycosides and saponins. These plant secondary metabolites serve as defense mechanisms against many microorganisms, insects and herbivores (Vaghasiya et al., 2011).
SECONDARY METABOLITES
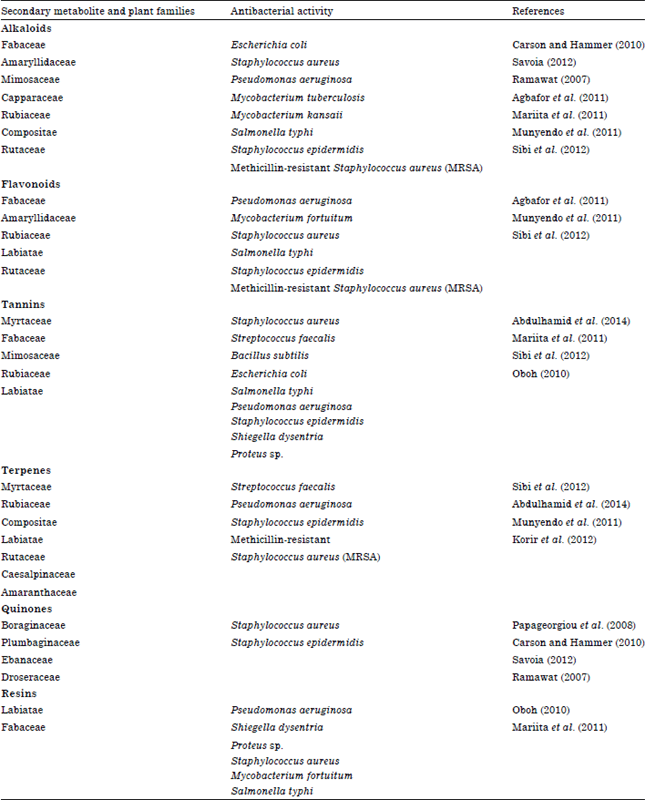
Secondary metabolites are being the subject for many research studies because these compounds exhibit many biological activities. These include antimicrobial, antifungal, anticancer and anti-inflammatory activities. For this study, only the antimicrobial activity of different secondary metabolite compounds from different plants will be discussed. In addition, the evidences of the presence of antimicrobial biological activity reported in recent studies will be presented. Table 1 summarizes these different secondary metabolites and their antibacterial activities.
ALKALOIDS
Alkaloids are organic heterocyclic nitrogen compounds that are basic-forming water-soluble salts. They contain nitrogen, which is usually derived from an amino acid. Several classes of flavonoids include phenylalkylamines, pyrrolidines, tropane alkaloids, pyrrolizidines and purine alkaloids (Carson and Hammer, 2010; Savoia, 2012; Ramawat, 2007). These metabolic compounds are grouped into three classes, depending on the precursors and final structure of the molecule. The classes include; (1) True alkaloids, which are basic and contain nitrogen in a heteroyclic ring-includes nicotine, (2) Pseudoalkaloids, which are also basic but they are not derived from amino acids-includes caffeine and (3) Protoalkaloids, which are amino acid derivatives, basic, but the nitrogen is not in a heterocyclic ring-includes phenylethylamine derived alkaloids including mescaline. Furthermore, alkaloids have been shown to have analgesic effects; morphine alkaloids are pain relievers that are used as narcotics (Agbafor et al., 2011).
Tamarindus indica (Fabaceae) aqueous pulp extract was tested for antimicrobial activity against Escherichia coli, Staphylococcus aureus, Salmonella typhi and Pseudomonas aeruginosa. Phytochemical screening of the aqueous extract revealed that the main constituents found in higher concentrations were alkaloids (4.32%). The T. indica pulp extract was found to have antibacterial effects against all microorganisms tested, except S. typhi (Abukakar et al., 2008). Moreover, Zapoteca portoricensis (Fabaceae) leaves were collected and extractions were done using four different solvents namely, water, methanol, ethylacetate and diethylether. These plant extracts were tested for antibacterial activity against E. coli, S. aureus, Streptococcus pyogenes, Klebsiella pneumonia and P. aeruginosa. After performing phytochemical analysis, it was shown that the aqueous plant extract was abundant with alkaloids. Therefore, these constituents may be exhibiting the antibacterial activity of the Z. portoricensis leaves, which can potentially be used for skin infection treatment, since the aqueous extract exhibited a large zone of inhibition against P. aeruginosa.
| Table 1: | Secondary metabolites from different plant families and their reported antibacterial activities |
 | |
The discovery of these secondary metabolites being active in inhibiting a gram-negative bacterium is astounding since secondary metabolites are usually known to more active against gram-positive bacterium and mostly inactive against gram-negative bacterium (Agbafor et al., 2011).
In another experimental study, methanol extracts were prepared from eight different plant species, which included: Thylachium africanum (Capparaceae), Boscia angustifolia (Capparaceae), Cissus quadrangularis (Vitaceae), Grewia simi (Tiliaceae), Acacia etbaica (Mimosaceae), Scadoxus multiflorus (Amaryllidaceae), Commiphora africana (Burseraceae) and Acacia nilotica (Mimosaceae). These extracts were tested against Mycobacterium tuberculosis, Mycobacterium kansaii, Mycobacterium smegmatis, Mycobacterium fortuitum, S. typhi, K. pneumonaie, P. aeruginosa, E. coli and S. aureus to determine their potential antibacterial activity. Mariita et al. (2011) used different chemical tests to identify the phytochemicals present in the plant extracts. It was revealed that there were a few promising antibacterial activities, including: (1) S. multiflorus methanol extract was found to be active against M. tuberculosis, (2) B. angustifolia presented inhibition against M. kansaii and M. tuberculosis compared to dimethyl sulphoxide (negative control), (3) A. nilotica displayed a strong antimicrobial activity against S. aureus and P. aeruginosa and (4) B. angustifolia and S. multiflorus presented strong antimicrobial activity against S. aureus and S. typhi (Mariita et al., 2011).
Furthermore, from the phytochemical analysis it was revealed that from the methanol extracts that had a potential for antimicrobial activity, alkaloids were present in high concentrations for the plant species S. multiflorus and A. nilotica, while B. angustifolia was found to have a trace of alkaloids. The presence of these alkaloids with additional phytochemicals possibly contribute to the antibacterial activity that has been discussed (Mariita et al., 2011). In a similar study, plant materials-whole leaves, roots and stem of Morinda citifolia (Rubiaceae) were used to prepare ethanol extracts. These extracts were tested against several bacterial isolates, which included: Staphylococcus aureus, Staphylococcus epidermidis, Serratia marcescens, Streptococcus pyogenes, Escherichia coli, Pseudomonas aeruginosa and Klebsiella pneumonia. After performing a qualitative phytochemical analysis, the extracts presented different phytochemicals depending on the plant material. All plant materials had alkaloids present, but they differed in quantity; the leaf extract has the highest concentration of alkaloids present, followed by the stem and then the root. Root extracts exhibited significant antibacterial activity with larger zone of inhibitions, then the leaf and root extracts; all extracts were found to be susceptible only to P. aeruginosa and S. epidermidis. Therefore, these alkaloids along with other phytoconstituents likely attribute to the antimicrobial activity (Sibi et al., 2012).
Additionally, Aspilia mossambicensis (Compositae), Ocimum gratissimum (Labiatae) and Toddalia asiatica (Rutaceae) were examined for antimicrobial activity against methicillin-resistant S. aureus and P. aeruginosa. Munyendo et al. (2011) prepared several extracts with different solvents (n-hexane, ethyl acetate and methanol) and different plant parts (leaves, stem bark and root). From this study, the methanol plant extracts had promising results including: (1) More bactericidal activity against the gram positive bacteria (MRSA) than gram-negative bacteria (P. aeruginosa), (2) Striking finding of the plant extract having higher antimicrobial activity levels than Gentamycin, the antibiotic positive control and (3) Highest antibacterial activity was seen in the stem bark of T. asiatica. After a phytochemical screening of different metabolites, it was evident that all the different plants and respective parts possessed different phytocompounds. The leaf and root extract of A. mossambicensis, leaf and root extract of O. gratissimum and all plant materials from the extract of T. asiatica displayed an abundance of alkaloids, in addition to other phytochemicals (Munyendo et al., 2011).
FLAVONOIDS
Flavonoids are phenolic structures found abundantly in photosynthesizing cells. They are usually found in many common edible plant parts such as: Fruits, vegetables, nuts and seeds. Flavonoid compounds have a structural feature of the 2-phenyl-benzopyrane or flavine nucleus, which consists of two benzene rings linked through a heterocyclic pyrane ring. To date, there have been 14 classes of flavonoids identified; they differ based on the chemical nature and position of substituents on the different rings (Carson and Hammer, 2010; Savoia, 2012; Ramawat, 2007).
Many flavonoids have been known to have antioxidant, anti-inflammatory and antitumor activity. In one experimental study, these secondary metabolites were also thought to contribute to antibacterial activity. The phytochemicals flavonoids were found in the aqueous plant extract of Zapoteca portoricensis leaves. From this research, it was demonstrated that these secondary metabolites could be partaking in the anti-Pseudomonas aeruginosa activity (Agbafor et al., 2011).
In addition, in a similar study, the methanol extract of S. multiflorus exhibited anti-M. fortuitum, S. aureus and S. typhi activities, after phytochemical analysis, it was found that this methanol extract was present in high concentrations of flavonoids. Therefore, these flavonoids, with the presence of other phytochemical could contribute to the different antibacterial activities (Mariita et al., 2011). Likewise, plant materials (leaf, root, whole leaves) of Morinda citifolia (Rubiaceae) were examined for antibacterial activity. The ethanol root extract exhibited antimicrobial activity to P. aeruginosa and S. epidermidis. After phytochemical analysis, it was shown that both the leaf and root ethanol extracts had a high concentration of flavonoids present, consequently revealing this biological activity (Sibi et al., 2012).
Moreover, Aspilia mossambicensis (Compositae), Ocimum gratissimum (Labiatae) and Toddalia asiatica (Rutaceae) were examined for antimicrobial activity against methicillin-resistant S. aureus and P. aeruginosa. It was found that the ethanol extract had the highest antibacterial activity against MRSA; flavonoids were revealed to be found in abundant numbers in all plant materials (leaves, stem bark and root) tested for the O. gratissimum extracts, while only a trace amount of flavonoids were found in all T. asiatica plant extracts. The flavonoids, along with other secondary metabolites could contribute to this biological activity (Munyendo et al., 2011).
TANNINS
Tannins are polymeric phenolic substances found in nearly all plant parts. In previous studies, tannins have been known to display different biological activities including antifungal and antibacterial (Carson and Hammer, 2010; Savoia, 2012; Ramawat, 2007). For instance, the antibacterial activity of ethanol and aqueous stem bark extracts from Psidium guajava (Myrtaceae) was evaluated against Staphylococcus aureus, Streptococcus faecalis, Bacillus subtilis, Escherichia coli and Salmonella sp. (Abdulhamid et al., 2014). Tannins were present in high concentrations for both the ethanol and aqueous extracts. Ethanol and aqueous extract of the bark of Psidium guajava exhibited antimicrobial activities against all microorganisms tested; however, the ethanol extract displayed larger zones of inhibitions than the aqueous extract for the most susceptible microorganism being S. faecalis and the least susceptible to Salmonella sp. (Abdulhamid et al., 2014).
In another study, the methanol extract of S. multiflorus exhibited anti-Mycobacterium fortuitum, S. aureus and S. typhi activities, after phytochemical analysis, it was found that this methanol extract was present in high concentrations of tannins. In addition, the methanol extract of A. nilotica, which exhibited anti-Staphylococcus aureus and Pseudomonas aeruginosa activities, presented a high concentration of tannins as well. Therefore, these tannins, with the presence of other phytochemicals, could potentially exhibit these antibacterial activities (Mariita et al., 2011).
Moreover, plant materials (whole leaves, roots and stem) of Morinda citifolia (Rubiaceae) were tested for antimicrobial activity. The ethanol leaf extract exhibited antimicrobial activity to P. aeruginosa and S. epidermidis. After phytochemical analysis, it was shown that both the leaf and root ethanol extracts had a high concentration of tannins present, consequently revealing antimicrobial activity (Sibi et al., 2012). In addition, in a similar study, Oboh (2010) collected Ocimum gratissimum L. (Labiatae) leaves because they are widely distributed in Nigeria and is commonly used as an herbal medicine for the treatment of many respiratory infections, headaches and skin diseases. He prepared an ethanol extract and tested it against several microorganisms including: Proteus species, Pseudomonas aeruginosa, Shigella dysentria, Escherichia coli, Klebsilla species and Staphylococcus aureus. It was found that the O. gratissimum extract exhibited antimicrobial activity against P. aeruginosa, S. dysentria, Proteus sp. and S. aureus. After phytochemical screening, it was revealed that the leaves possess several secondary metabolites, including tannins; therefore, these secondary metabolites can contribute to their antimicrobial activities (Oboh, 2010).
TERPENES
Terpenes form the largest group of natural compounds found, with currently over 30,000 known structures. The number of isoprene units they possess can identify them; therefore, 1 isoprene unit is called hemiterpene, 2 isoprene units are called monoterpenes and so forth. Mostly, essential oils are made up of monoterpenes or sesquiterpenes (3 isporene units) (Carson and Hammer, 2010; Savoia, 2012; Ramawat, 2007). Monoterpenes are formed when two five-carbon isoprene units are joined, having a molecular structure of C10H16. The structures of monoterpenes can be cyclic, linear, regular or irregular. Some derivatives of monoterpenes include alcohols, esters, phenols, ketones and aldehydes (Carson and Hammer, 2010; Savoia, 2012; Ramawat, 2007). The biosynthesis of terpenes occurs via., two different biological pathways: (1) Mevalonic-acid pathway, which mostly produces sesquiterpenes, sterols and ubiquinones, (2) Methyl-erythritol phosphate pathway, which the majority of the compounds synthesized are hemiterpenes, monoterpenes and diterpenes (Carson and Hammer, 2010; Savoia, 2012; Ramawat, 2007).
One example of terpene derivatives is terpenoids. Terpenoids were extracted from the bark of Psidium guajava and it was discovered that these compounds were found in high concentrations for the ethanol extract, but was not detected in the aqueous extract. These compounds, along with other phytochemicals found in the bark, exhibited anti-Streptococcus faecalis activity (Abdulhamid et al., 2014). In addition, after phytochemical analysis, plant materials of Morinda citifolia (Rubiaceae) displayed a high concentration of terpenoids present in the leaf ethanol extract. This extract was found to have anti-P. aeruginosa and S. epidermidis activities (Sibi et al., 2012). Similarly, Aspilia mossambicensis (Compositae), Ocimum gratissimum (Labiatae) and Toddalia asiatica (Rutaceae) were examined for antibacterial activitiy against methicillin-resistant S. aureus (MRSA) and P. aeruginosa. From this study, it was discovered that the ethanol extract had the highest antibacterial activity against MRSA. Terpenoids were revealed to be found in abundant numbers in all plant materials (leaves, stem bark and root) tested for the O. gratissimum extracts, while only a trace amount of terpenoids were found in all A. mossambicensis and T. asiatica plant extracts. These secondary metabolites, along with other phytochemicals could be involved in this antibacterial activity (Munyendo et al., 2011).
In addition, Korir et al. (2012) examined the antimicrobial activity of two plant species: Senna didymobotrya (Caesalpinaceae) and Cyanthula polycephala (Amaranthaceae). For each plant species, three solvents were used for extraction; they include hexane, dichloromethane and methanol. These plant extracts were tested against five different microorganisms, which included S. aureus, MRSA, E. coli, P. aeruginosa and K. pneumonia. In this experimental study, zone of inhibitions’ diameters were used to measure antimicrobial activity (Korir et al., 2012). Methanol and dichloromethane extracts had the highest activity compared to the water and hexane extracts. Using thin layer chromatography as their phytochemical analysis method, Korir et al. (2012) revealed that the both plant extracts had a presence of phenol compounds. A striking discovery found in this study was that the methanol extract of C. polycephala was found to be most active against MRSA, a bacterium that has become resistant to antibiotics. The zone of inhibition of this extract was 16.0 mm compared to the positive control Gentamicin, which had a zone of inhibition of 9.0 mm. Furthermore, the methanol extract exhibited anti-bacterial activity against S. aureus and P. aeruginosa with zone of inhibitions of 14 and 10 mm, respectively (Korir et al., 2012).
QUINONES
Quinones are compounds that contain aromatic rings with two ketone substitutions. In the past years, researchers have been able to identify at least 400 naturally occurring quinones, which are found in all plant organs. The class naphthoquinones are one of the largest groups of plant secondary metabolites with a wide range of biological activities. They can be found in about 20 plant families including Plumbaginaceae, Ebanaceae and Boraginaceae; examples of natural naphthoquinones compounds are plumbagin, shikonin, alkannin and their derivatives (Carson and Hammer, 2010; Savoia, 2012; Ramawat, 2007). The naphthoquinone plumbagin is an active constituent in the plant families Plumbagenaeace, Droseraceae and Ebenceae. Depending on the quinone, plants synthesize these metabolites via., several pathways including: (1) Ortho-succinyl benzoic acid pathway for lawsone and juglone, (2) Para-hydroxybenzoic acid-mevalonic acid pathway for shikonin and (3) Acetic acid-mevalonic acid pathway for plumbagin (Ramawat, 2007).
Papageorgious et al. (2008) performed an antimicrobial study on naphthoquinones, specifically the enantiomers alkannins and shikonins. Alkannins and shikonins can be found in at least a hundred and fifty species that belong mainly to the Boraginaceae-including the genera Lithospermum. Animal and clinical studies have demonstrated the effectiveness of alkannins and shikonin esters; therefore, it has resulted in the approval of the pharmaceutical ointment known as Histoplastin Red. Clinical studies have revealed that these esters formulated in Histoplastin Red, displayed wound healing properties with both anti-inflammatory activities and antimicrobial activities against ulcer development (Papageorgiou et al., 2008).
Furthermore, Histoplastin Red was tested for anti-S. aureus and S. epidermis activity. It was found that the enantiomer alkannin inhibited both Staphylococci growth. In addition, β-dimethylacryl, an ester of alkannin, also effectively inhibited all Staphylococci growth (Papageorgiou et al., 2008). Looking at the clinical evidence, it has been determined that there is a broad antimicrobial spectrum for napthoquinones derivatives, alkannins and shikonins enantiomers (Papageorgiou et al., 2008).
RESINS
Resins are non-volatile plant products, which can be classified as either a surface or internal resin. They are insoluble in water but are soluble in organic solvents. Resins are usually found in plants as mixtures of compounds, commonly including flavonoids, terpenoids and fatty substances. One class found under the resin group is essential oils; they are volatile oils, soluble in organic solvents and are produced in plant oil glands (Carson and Hammer, 2010; Savoia, 2012; Ramawat, 2007).
Taxol, a present anticancer drug is composed of phytochemicals known a diterpenes. This drug was first isolated from the bark of Taxus brevifolia. Two sesquiterpene precursors coupled make a triterpene, which contains 30 carbons. Several of these compounds occur in plants as glycosides, or saponins. These saponins are molecules that are made up of carbohydrates linked to steroids or triterpenes (Carson and Hammer, 2010; Savoia, 2012; Ramawat, 2007). Oboh (2010) examined the antimicrobial activity of an ethanol extract made from Ocimum gratissimum (Labiatae) leaves. It was revealed that the ethanol extract exhibited antibacterial activity against Pseudomonas aeruginosa, Shiegella dysentria, Proteus sp. and Staphylococcus aureus. From the phytochemical analysis, it was displayed that the leaves from O. gratissimum has several phytochemicals, including cardiac glycosides (Oboh, 2010).
The aqueous pulp extract of Tamarindus indica was subjected to phytochemical analysis and it was revealed that some phytochemicals found in this pulp extract included resins, specifically saponins (2.2%) and glycosides (1.59%). The pulp extract exhibited the highest sensitivity to S. aureus, while not having any sensitivity to S. typhi (Abukakar et al., 2008). Likewise, after phytochemical analysis, it was found that the methanol extract of S. multiflorus was present in high concentrations of saponins. The methanol extract exhibited anti-M. fortuitum, S. aureus and S. typhi activities. As a result, these secondary metabolites, with the presence of other phytochemicals could potentially exhibit these biological activities (Mariita et al., 2011). In a related experimental study, Oboh (2010) examined the antimicrobial activity of an ethanol extract made from Ocimum gratissimum (Labiatae) leaves. It was revealed that the ethanol extract exhibited antibacterial activity against P. aeruginosa, S. dysentria, Proteus sp. and S. aureus. From the phytochemical analysis, it was displayed that the leaves from O. gratissimum has several phytoconstituents, including saponins (Oboh, 2010).
From this study, it is evident that plant cells produce a variety of compounds, mainly secondary metabolites for defense mechanisms against bacteria. As shown, these phytochemicals can possess antibacterial activities. It is clear that many different phytoconstituents have different mechanisms of inhibiting the bacteria, since they differ within the same plant species, different parts of the plants, such as leaves or bark, have been shown to possess different quantities of these metabolites. In addition, the different plant families that have been tested demonstrate that different families have a unique combination of secondary metabolites; therefore, exhibiting different antibacterial activities. Several secondary metabolites exhibited highly potent biological activities that have been attributed to antimicrobial activities; in some cases, the plant extracts that possessed these phytochemicals had higher zone of inhibitions than an antibiotic, demonstrating promising antibacterial activities for these compounds. The emerging research on identifying, purifying and testing antimicrobial activities of different secondary metabolites is underway and further investigations are encouraged to further our knowledge about these compounds. Secondary metabolites can help to treat infectious diseases that have increased resistance to current antibiotics. They could provide alternative medical treatment for many, especially in developing countries where people may not have access to healthcare.
FUTURE PERSPECTIVES
Over the years, many scientists have performed research on different plant families to be able to identify antimicrobial activities and secondary metabolites. Therefore, in the future, there should be experimental studies to test the biological activities of different plant families. These antibacterial properties are important for future pharmacological uses. Future research is to be able to identify and learn about the mechanisms these secondary metabolites undergo. In addition, future researchers should isolate all compounds from plants, to be able to determine exactly which compound is contributing to the antibacterial activity. In many studies that have been published, a plant extract will have antibacterial potential, so they test it for different secondary metabolites; then several secondary metabolites will be abundant, so it is not certain which components are exhibiting this activity or if it is a synergy among different compounds. Lastly, the future of ethnopharmacology seems very fascinating, there is hope to eliminate these antibiotic-resistant microorganisms by eliminating them with phytochemicals.