Research Article
Effects of Bay Leaves on the Patients with Diabetes Mellitus
Department of Medical Technology, Faculty of Allied Medical Sciences, Zarqa Private University, P.O. Box 132222, Zarqa, 13132, Jordan
Diabetes Mellitus (DM) is a major risk factor for cardiovascular disease (Jaffe et al., 2006). Diabetes is a chronic disorder of glucose metabolism resulting from dysfunction of pancreatic beta cells and insulin resistance. The incidence of cardiovascular diseases is increased two- to four-fold in people with diabetes (Grundy, 2006). Spices that have been reported to be hypoglycemic include fenugreek, garlic, turmeric, cumin, ginger, mustard, curry and coriander (Srinivasan, 2005). We have also shown that spices such as cinnamon, cloves, bay leaves and turmeric display insulin-enhancing activity in vitro (Khan et al.,1990). Botanical products can improve glucose metabolism and the overall condition of individuals with diabetes not only by hypoglycemic effects but also by improving lipid metabolism, antioxidant status and capillary function (Bailey and Day, 1989). The composition of human diet plays an important role in the management of lipid and lipoprotein concentrations in the blood. The importance of serum lipoprotein disturbances and abnormal lipid metabolism characterized by hyperlipidaemia or hyperlipoproteinmia as etiological factors in the development of coronary heart diseases and potentiating of arteriosclerosis is now supported by a considerable body of evidence amassed from epidemiological and population studies (Arteriosclerosis, 1971; Coronary Drug Project Research Group, 1975; Turpeinen, 1979; Lipid Research Clinics Programme, 1984). Moreover, many studies have now shown that elevated concentration of total or Low Density Lipoprotein (LDL) cholesterol in the blood are powerful risk factors for coronary heart disease, whereas high concentrations of High Density Lipoprotein (HDL) cholesterol or a low LDL (or total) to HDL cholesterol ratio may protect against coronary heart disease (Shaten et al., 1991; Castelli et al., 1992). The use of herbs as medicines has played an important role in nearly very culture on earth, including Asia, Africa, Europe and the Americas Wargovich et al., 2001). Herbal medicine is based on the premise that plants contain natural substances that can promote health and alleviate illness. Bay leaf, Laurus nobilis, belongs to the family Lauraceae and it is on of the most popular culinary spices in Western countries. Bay leaf has been used as herbal medicine and has pharmacological activity which includes antibacterial, antifungal, antidiabetes and antiinflammatory effects (Fang et al., 2005).
The objective of this study was to determine if bay leaves may be important in the prevention and/or alleviation of type2 diabetes.
Data collection: This study design and utilized to show the impact of bay leave supplementation on blood glucose and lipids levels among type2 diabetic. Then comparison between two results to improved the effects of bay leave on blood glucose and lipids levels. The study was conducted in Al Mafraq Governmental Hospital in Jordan from January up to July 2010.
Sixty five individuals with type2 diabetes of both sexes (35 males and 30 females) of age 35 years or older were recruited for participating in the current study. Only those diabetic subjects, who were not taking medicine for other health conditions and whose fasting blood glucose were in the range of 160-300 mg dL-1 and high lipids level were included in the study. The study was approved by Medical Ethical Committee of the Zarqa Private University.
The study was conducted for 4 weeks. Type 2 diabetic individuals were allowed to take their routine diet and usual diabetic medicine. The individuals were told to take 2 capsules of whole bay leave powder immediate after breakfast, lunch, dinner and before sleep for 4 weeks each capsules contain (250 mg) that means 2 g per day, these capsules were prepared by technician of the local pharmacy. The research did not suggest any alterations in other aspects of the subject's medical care, diet, or exercise. Compliance was monitored by contact with the subjects.
Biochemical analysis: Biochemical analysis done by collection of blood samples approximately 8 mL blood samples were taken before breakfast from the vein directly into lithium heparin vacuum tubes for measurements of fasting blood glucose level, triglyceride, total cholesterol, LDL and HDL. The samples were centrifuged within 1 h at 1000xg for 10 min at 4°C, the plasma transferred into separate labeled tubes and transferred immediately by cold boxes filled with ice to the Al Mafraq Governmental Hospital laboratory. All biochemical measurements were carried out by the same team of laboratory technicians using an auto analyzer (IMMULIT, DPC, Los Angeles, CA, USA) each individual on the starting day and at end of week 4. Prior to implementation of the training program, an official permission was obtained from the supervisors of the selected units. This was intended to facilitate data collection and to explain study purpose. At the beginning of the study, participants' were invited to participate in the study. The researcher explained the study purpose and procedures for the randomly selected sample. Potential subjects were further informed that the participation was voluntary and that study findings would be presented group wise and no individual would be recognized.
Statistical analysis: Collected data were tabulated and needed statistical analyses were done using descriptive statistic, means and Standard Deviation (SD) of the means were calculated utilizing the computer data processing (SPSS, version 12). A probability value (p) of <0.05 was considered to be statistically significant.
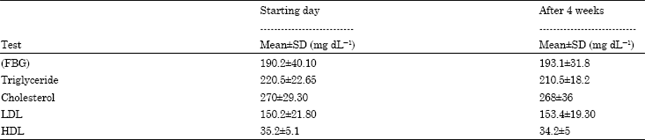
Sixty five subjects of type2 diabetes were randomized into the study their samples had a mean age of 42 years (SD±6). Thirty five patients were male and thirty female. The majorities were married; the mean length of time since diabetes was diagnosed was 9 years (SD±8). Repeated measure ANOVA was used to assess the effectiveness of bay leave among type2 diabetic individuals by examining fasting blood glucose and lipid levels changes across time is shown in Table 1. The fasting blood glucose and lipids values on the starting day indicate of diabetic individuals before the start of bay leave. So these values levels were the control for the study.
On the starting day of the experiment (day 0), the mean fasting blood glucose levels of the diabetic individuals, were (192.2 mg dL-1), Triglyceride (210.5 mg dL-1), Total Cholesterol (273 mg dL-1), LDL (160.1 mg dL-1) and HDL (37.1 mg dL-1). When the diabetic individuals used the doses of bay leave (2 g) for 4 weeks, their mean Fasting Blood Glucose levels dropped to (140.3 mg dL-1), Triglyceride (159 mg dL-1), Total Cholesterol (210 mg dL-1), LDL (122.1 mg dL-1) and HDL (45.3 mg dL-1) . The reduction in the mean Fasting Blood Glucose levels and lipid levels were significant at (p<0.05).
Table 2 shows no significant changes in the placebo group. This conclusion was supported by the repeated measure ANOVA (F) test.
The present study shows that 4 weeks of bay leaves supplementation does improve plasma glucose and lipid profiles in patients with type 2 diabetes. The active components of bay leaves are under study. Of the 81 compounds representing 98.74% of total oil of Laurus nobilis, monocyclic monoterpenes such as 1,8-cineole (58.59%), alpha-terpinyl acetate (8.82%) and terpinene-4-ol (4.25%) are the main components. Bicyclic monoterpenes such as alpha- and beta-pinene (3.39-3.25%) and sabinene (3.32%) are also present. The acyclic monoterpenes, linalool, myrcenol and sesquiterpenes are found at less than 0.5%. o-Cymene (1.30%) and p-cymene (1.83%) are also present and cumin aldehyde, dimethylstyrene, eugenol, methyl eugenol and carvacrol are minor, aromatic compounds of laurel oil (Yalcin et al., 2007).
| Table 1: | Levels of fasting blood glucose and lipid profiles before and after consumption of bay leaves on type 2 diabetic |
 | |
*Significant at (p<0.05) | |
| Table 2: | Effect of placebo on fasting blood glucose and lipid profiles of the type 2 diabetic |
 | |
The active in vitro component of bay leaves is water soluble (Broadhurst et al., 2000). The active component of the bay leaves is likely a polyphenol since more than 80% of the in vitro insulin potentiating activity was removed by polyvinylpyrrolidone (Broadhurst et al., 2000), which binds aromatic hydroxyl groups (Wall et al., 1996). The current study indicate that present of Polyphenols compound in bay leaves have been shown to have effects on insulin sensitivity, glucose uptake and antioxidant status, these funding agree with Anderson (2008). Also, a variety of phenolic compounds, in addition to falvonoids, are found in fruit, vegetables and many herbs. The phenolic compounds (such as caffeic, ellagic and ferulic acids, sesamol and vanillin) inhibit atherosclerosis (Decker, 1995). The present study shows reduction in triglyceride, total cholesterol and LDL related to phonlic compounds supported by Nofer et al. (2002) documented that the role in reverse cholesterol transport, HDL have recently been recognized to have several other important cardioprotective properties including the ability to protect LDL from oxidative modification. Also, Parthasarathy et al. (1990) suggested that HDL may play a protective role in atherogenesis by preventing the generation on an oxidatively modified LDL and the mechanism action of HDL may involve exchange of lipid peroxidation products between the lipoproteins. HDL is the major carrier of cholesteryl ester hydroperoxides, but more than this it appears to have the prolonged capacity to decrease the total amount of lipid peroxides generated on LDL during oxidation. Several enzymes are present on HDL: paraoxonase (an enzyme normally resident on HDL), lecithin, Cholesterol acyl transferase, platelet ctivating factor cetylhydrolase, phospholipase D and protease. Apolipoproteins, such as apolipoprotein AI, could also have enzymic activity (Mackness and Durrington, 1995). The current study shows increasing in HDL after consumption bay leaves for 30 days supported by Mackness et al. (1993) suggested that a direct role for HDL in preventing atherosclerosis probably by an enzymic process which prevents the accumulation of lipid peroxides on LDL. They reported that paraoxonase is an example of an enzyme which might possibly be involved. Oxidative stress is particularly active in brain whose membranes are rich in polyunsaturated, highly peroxidable fatty acids. The antiradical efficiency of bay leaf and the extent of oxidative damage in diabetic rat brain synaptosomes. The diabetic rat brain is defective in neurotransmission that is attributable to oxidative damage (Mooradian and Morin, 1991). This prompted the present study. The total polyphenolic content of bay leaves was found to be 6.7 mg gallic acid equivalents (GAE)/100 g. Bay leaf displayed scavenging activity against superoxide and hydroxyl radicals in a concentration-dependent manner. Further, bay leaves showed inhibition of Fe2+-ascorbate induced lipid peroxidation in both control and diabetic rat (Lakshmi et al., 2007).
The increased level of cholesterol in the diabetic rat brain synaptosomes could arise from a rise in cholesterol biosynthesis. Increased activity of hydroxy methyl glutaryl (HMG CoA) reductase, a rate-limiting enzyme in cholesterol biosynthesis has been reported in diabetic rats (Draznin and Eckel, 1993).
Bay leaf contains linalool as the active component, which may effectively scavenge the free radicals and terminate the radical chain reaction. Linalool has considerable protective effects against H2O2 induced- oxidative stress in brain tissues by decreasing oxidative reaction in unsaturated fatty acids (Celik and Ozkaya, 2002).
In summary, bay leaves reduced, total cholesterol, LDL, triglycerides and glucose and increased HDL levels in people with type 2 diabetes. Additional studies are needed to confirm these results and also to identify the active components.
ACKNOWLEDGMENTS
This research was supported by the Deanship of Research and Graduate Studies, Zarqa Private University, Zarqa, Jordan.