Research Article
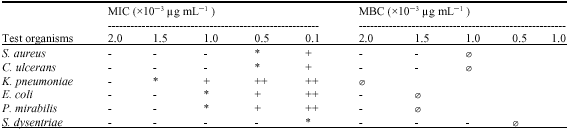
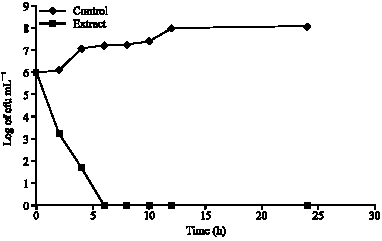
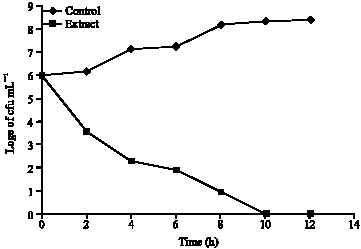
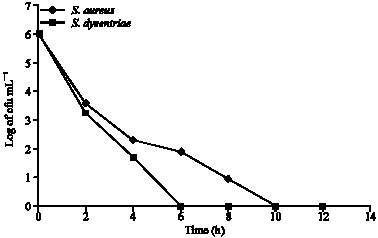
Phytochemical and Antibacterial Studies of Root Extract of Cochlospermum tinctorium A. Rich. (Cochlospermaceae)
Department of Microbiology,Ahmadu Bello University, Zaria, Nigeria
I.A. Bello
Department of Chemistry,Ahmadu Bello University, Zaria, Nigeria
A.B. Aliyu
Department of Chemistry,Ahmadu Bello University, Zaria, Nigeria
T. Olurishe
Department of Pharmacology and Clinical Pharmacy,Ahmadu Bello University, Zaria, Nigeria
S.M. Maidawa
Department of Veterinary Anatomy,Ahmadu Bello University, Zaria, Nigeria
J.D. Habila
Department of Chemistry,Ahmadu Bello University, Zaria, Nigeria
E.O. Balogun
Department of Biochemistry, Ahmadu Bello University, Zaria, Nigeria