Research Article
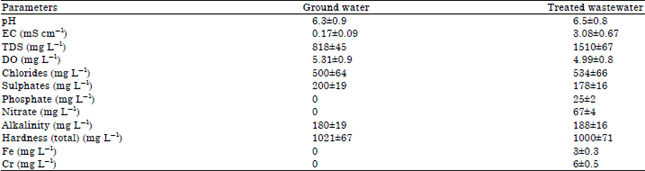
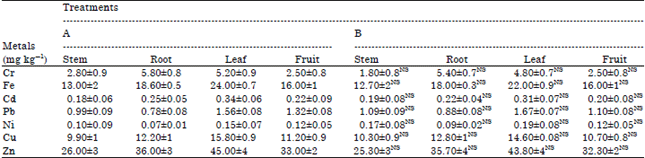
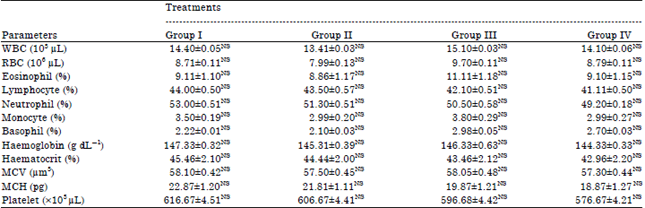
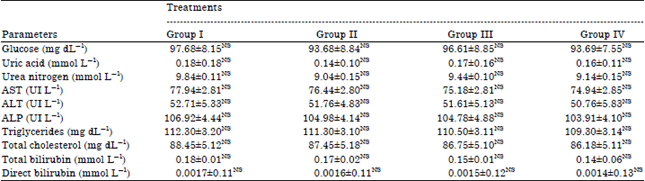
Toxicity Evaluation of Crop Plants Irrigated with Treated Municipal Wastewater
Department of Genetics, University of Delhi South Campus, New Delhi, 110021, India
S.A. Nasim
Department of Medical Elementology and Toxicology, Jamia Hamdard, Hamdard Nagar, New Delhi, 110062, India
S. Nafees
Department of Medical Elementology and Toxicology, Jamia Hamdard, Hamdard Nagar, New Delhi, 110062, India
S. Srivastava
Department of Genetics, University of Delhi South Campus, New Delhi, 110021, India