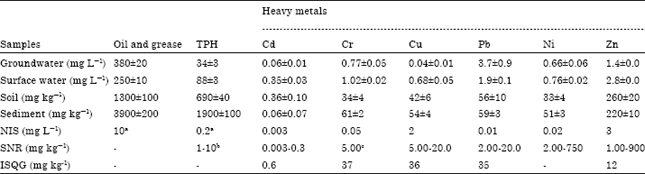
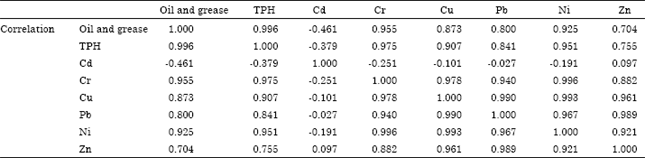
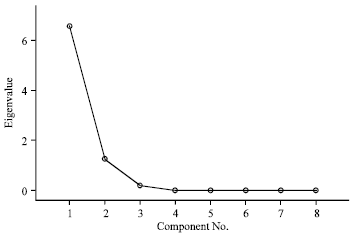
Research Article
Source Identification of Chemical Contaminants in Environmental Media of a Rural Settlement
Department of Chemistry, University of Ibadan, Ibadan, Nigeria
A.T. Etchie
Department of Chemistry, University of Ibadan, Ibadan, Nigeria
G.O. Adewuyi
Department of Chemistry, University of Ibadan, Ibadan, Nigeria