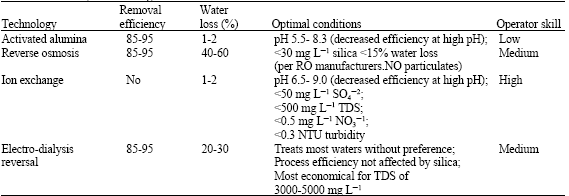
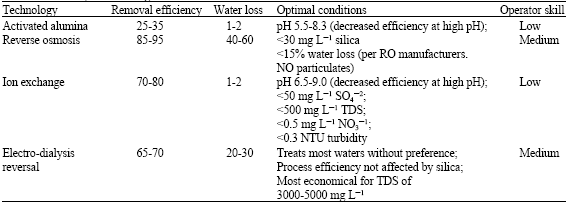
Research Article
Arsenic, Fluoride and Nitrate in Drinking Water: The Problem and its Possible Solution
PCSIR Labs Complex, Karachi-75280, Pakistan
Noor Taj
PCSIR Labs Complex, Karachi-75280, Pakistan
Fahmida Parveen
PCSIR Labs Complex, Karachi-75280, Pakistan
T.H. Usmani
PCSIR Labs Complex, Karachi-75280, Pakistan
Rafia Azmat
Department of Chemistry, Jinnah University for Women, Karachi, Pakistan
Fahim Uddin
Department of Chemistry, Karachi University, Karachi-75270, Pakistan