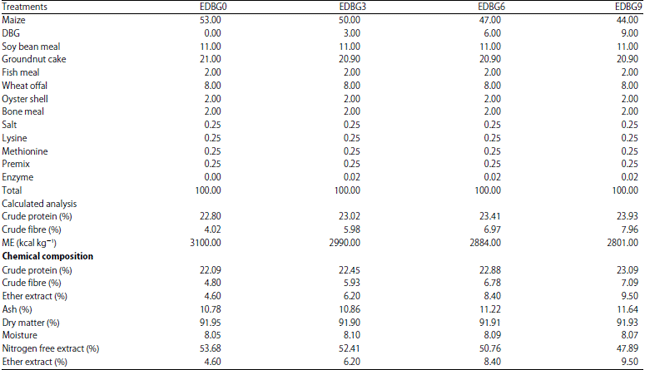
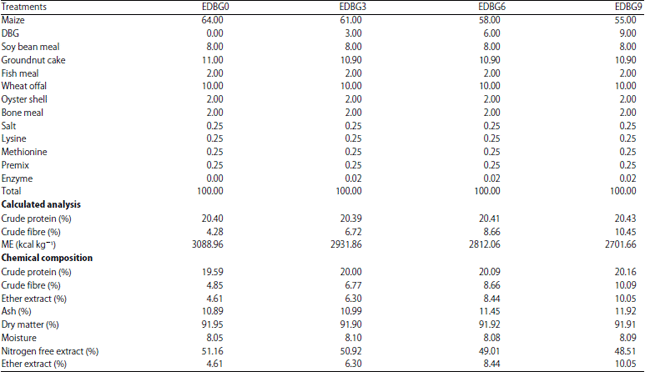
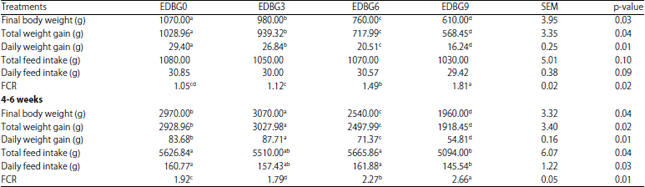
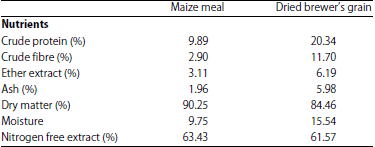Research Article
Blood Bio-Markers, Growth Traits, Carcass Characteristics and Income Over Feed Cost of Broiler Birds Fed Enzyme Fortified Dried Brewer’s Grain
Department of Livestock and Pasture Science, University of Fort Hare, Private Bag X1314, Alice 5700, South Africa
LiveDNA: 234.29280
C.L. Ugwuanyi
Department of Animal Science, Federal Collage of Agriculture, Ishiagu, Ebonyi State, Nigeria
E. Onwujiariri
Department of Animal Science, Federal Collage of Agriculture, Ishiagu, Ebonyi State, Nigeria
C.O. Osita
Department of Animal Science, University of Nigeria, Nsukka, Nigeria
E.A. Akuru
Department of Livestock and Pasture Science, University of Fort Hare, Private Bag X1314, Alice 5700, South Africa
A.O. Ani
Department of Animal Science, University of Nigeria, Nsukka, Nigeria
E. M. Idamokoro
Department of Livestock and Pasture Science, University of Fort Hare, Private Bag X1314, Alice 5700, South Africa
LiveDNA: 234.25028
A.B. Falowo
Department of Livestock and Pasture Science, University of Fort Hare, Private Bag X1314, Alice 5700, South Africa