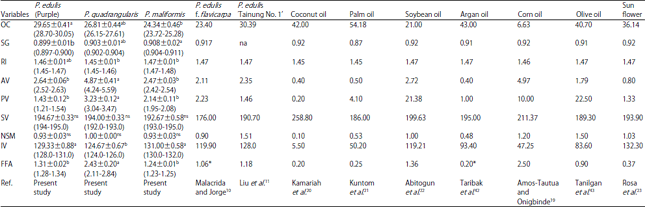
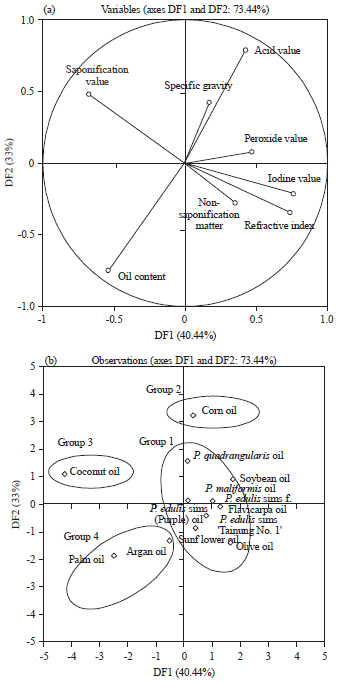
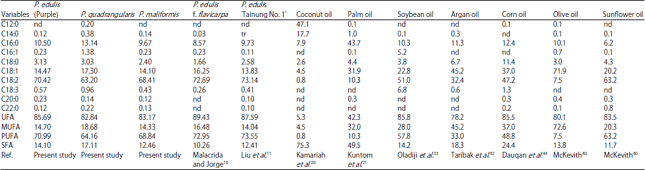
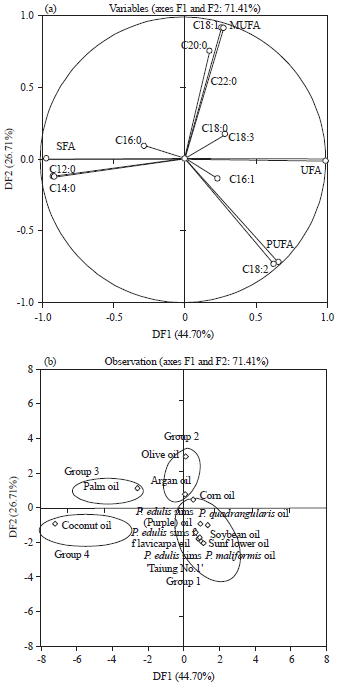
Research Article
Physicochemical, Fatty Acid and Antioxidant Properties of Passion Fruit (Passiflora Species) Seed Oil
Department of Crop Science, Faculty of Agriculture and Food Sciences, Universiti Putra Malaysia, Bintulu Sarawak Campus, 97008 Bintulu, Sarawak, Malaysia
LiveDNA: 60.27181
J.S. Bujang
Department of Biology, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Darul Ehsan, Malaysia
M.H. Zakaria
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Darul Ehsan, Malaysia