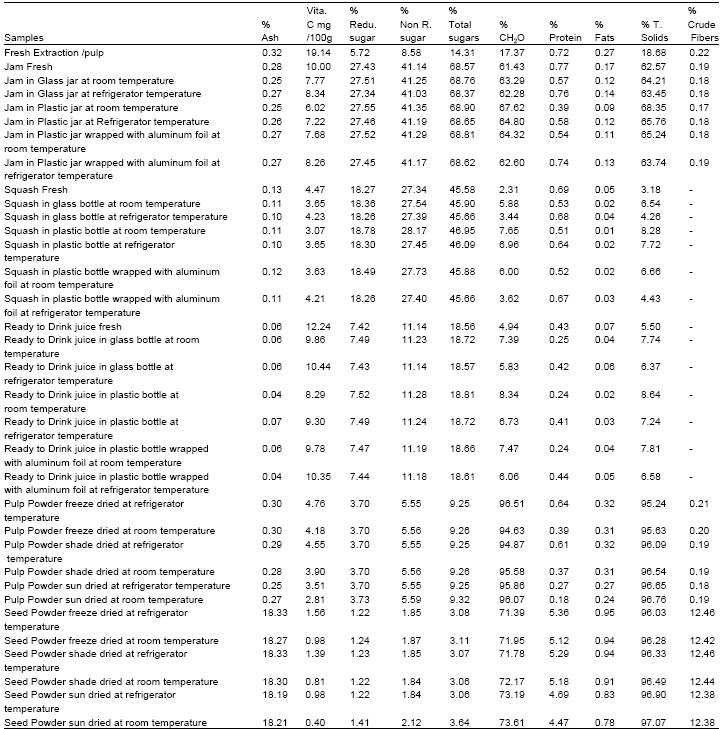
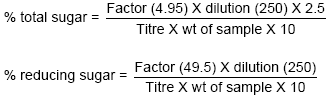Research Article
Determination of Nutritive Values of Jamun Fruit (Eugenia jambolana) Products
Institute of Food Sciences and Technology, Sindh Agriculture University, Tandojam, Pakistan
Saghir Ahmed Sheikh
Institute of Food Sciences and Technology, Sindh Agriculture University, Tandojam, Pakistan
S. M. Nizamani
Institute of Food Sciences and Technology, Sindh Agriculture University, Tandojam, Pakistan