Research Article
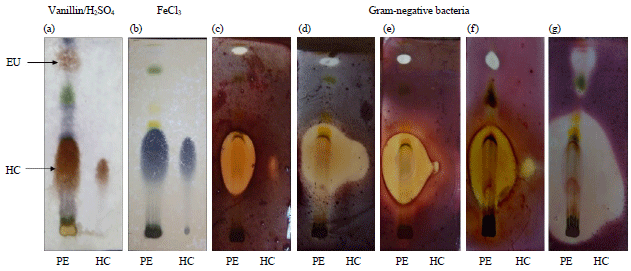
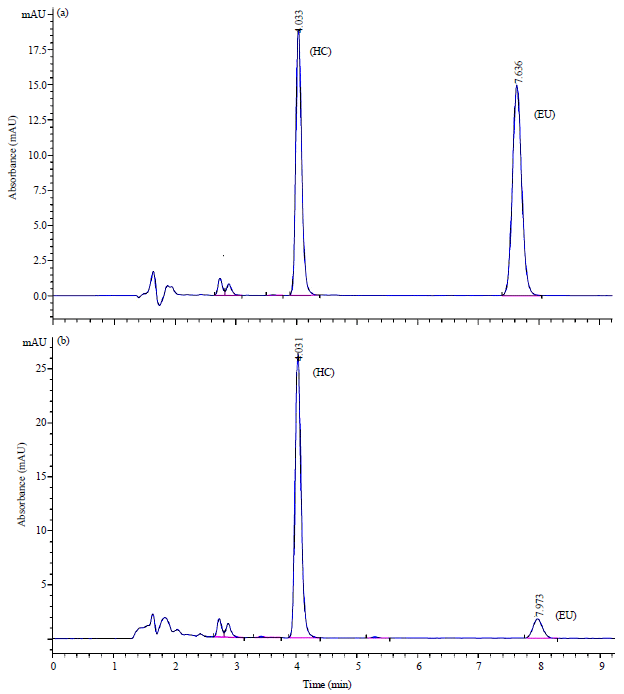
Phytochemical Analysis, Identification and Quantification of Antibacterial Active Compounds in Betel Leaves, Piper betle Methanolic Extract
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
LiveDNA: 60.12587
C.R. Saad
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
M.D. Hassan
Department of Clinical Veterinary Studies, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
Y. Rukayadi
Institute of Bioscience, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
M.H. Norazian
Kulliyyah of Pharmacy, International Islamic University Malaysia, 25200 Bandar Indera Mahkota, Kuantan, Pahang, Malaysia
M.S. Kamarudin
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia