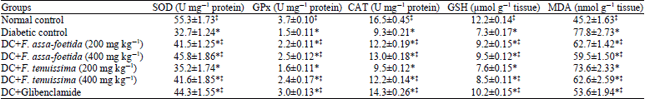
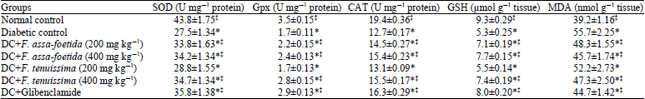
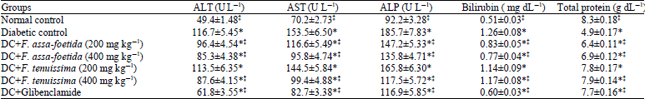
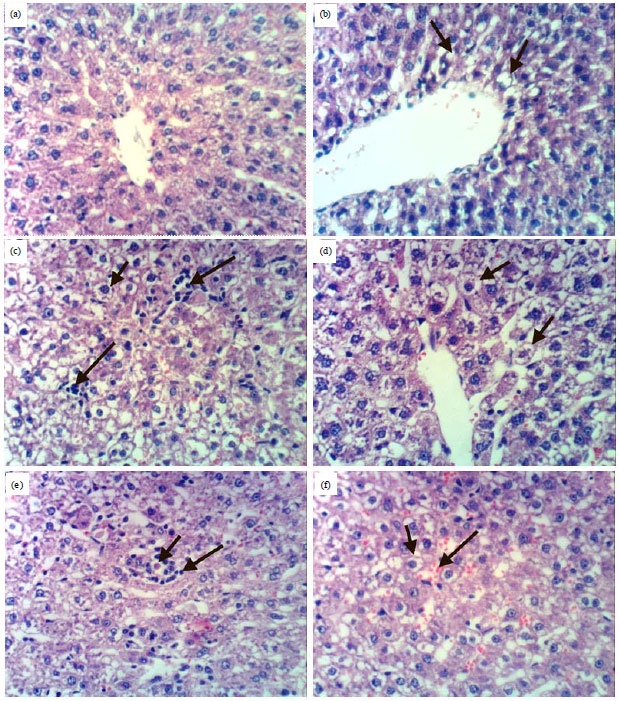
Research Article
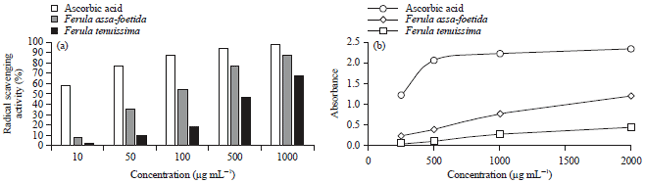
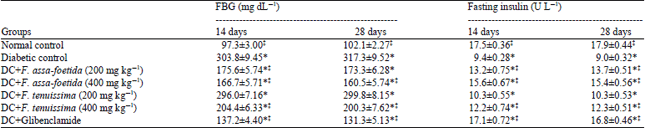
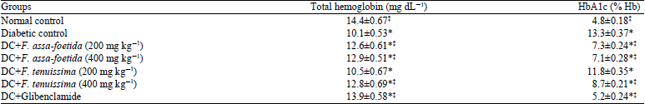
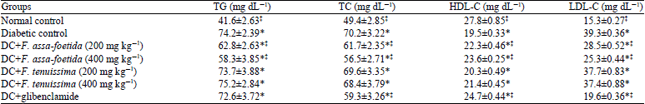
Antihyperglycemic and Antihyperlipidemic Effects of Ferula assa-foetida and Ferula tenuissima Extracts in Diabetic Rats
Department of Pharmacognosy, College of Pharmacy, Prince Sattam Bin Abdulaziz University, Al-Kharj, Kingdom of Saudi Arabia
Gamal A. Soliman
Department of Pharmacology, College of Veterinary Medicine, Cairo University, Egypt
Rehab F. Abdel- Rahman
Department of Pharmacology, National Research Centre, Cairo, Egypt
Maged Saad Abdel-Kader
Department of Pharmacognosy, College of Pharmacy, Prince Sattam Bin Abdulaziz University, Al-Kharj, Kingdom of Saudi Arabia
Majid A. Ganaie
Department of Pharmacology, College of Pharmacy, Prince Sattam Bin Abdulaziz University, Al-Kharj, Kingdom of Saudi Arabia
Erdal Bedir
Department of Bioengineering, College of Engineering, Ege University, Izmir, Turkey
Sura Baykan Erel
Department of Pharmaceutical Botany, College of Pharmacy, Ege University, Izmir, Turkey
Bintug Ozturk
Department of Pharmaceutical Botany, College of Pharmacy, Ege University, Izmir, Turkey