Research Article
Thymoquinone Attenuates Toxicity and Oxidative Stress Induced by Bisphenol A in Liver of Male Rats
Department of Zoology, Faculty of Science, University of Alexandria, Egypt
In the past century, the production of synthetic chemicals as well as the unexpected by-products has imposed adverse health consequences on human and wildlife. Bisphenol A (BPA) is currently the base compound in the manufacture of polycarbonate plastic and epoxy resin. Bisphenol A is a fairly ubiquitous compound, found as a common component in plastic baby bottles, as well as food and beverage containers (Rubin, 2011). It is one of the most abundant chemicals in the environment and can be detected in significant amounts in different foods, with an estimated mean dietary daily intake in adult human populations of 0.4-1.4 μg kg-1 (FAO/WHO, 2010). Humans are prone to a direct risk of exposure to BPA as it can leach out from the polymers to food or water especially upon heating (Koch and Calafat, 2009). Bisphenol A is a xenoestrogen which imitates estrogen and becomes related to an array of health adverse effects, including breast cancer (Pupo et al., 2012), disturbances in reproductive function (Takeuchi et al., 2004) in addition to disturbance in steroidogenesis (Zhang et al., 2011). Independent of its estrogenic activity, BPA has been shown to dysregulate cytokines (Wetherill et al., 2007) and to induce oxidative stress in the brain, liver and kidney (Bindhumol et al., 2003; Kabuto et al., 2004). A close relationship between BPA and cardiovascular disorders, type 2 diabetes and liver enzyme abnormalities has also been reported (Lang et al., 2008).
Liver plays an astonishing array of vital functions. It is involved in almost all the biochemical pathways needed for maintenance and regulation of homeostasis. It is the major organ for the metabolism and detoxification of xenobiotics and therefore is largely prone to BPA more than other organs (Knaak and Sullivan, 1966). Bisphenol A was found to be associated with abnormal liver function and altered insulin homeostasis (Lang et al., 2008). Some studies reported that high doses of BPA decreased the viability of rat hepatocytes, reduced mitochondrial function in hepatocytes and caused formation of multinucleated giant cells in rat hepatocytes (Nakagawa and Tayama, 2000).
Because of the concerns about the side effects of conventional medicine, interests in medicinal plants and their derived natural products has burgeoned. Among the promising medicinal plants, Nigella sativa (Family: Ranunculaceae) is an amazing herb with a rich historical and religious background. Thymoquinone (TQ) is the most potent and pharmacologically active constituent in the volatile oil of seeds of this medicinal plant. It has been reported to have various therapeutic effects such as anti-inflammatory, antibacterial, antifungal, antiparasitic, antiasthmatic and antidiabetic (Houghton et al., 1995). Thymoquinone also protects against chemical-induced carcinogenesis (Worthen et al., 1998). Furthermore, TQ has been reported to possess strong antioxidant properties (Houghton et al., 1995). It prevents oxidative injury in hepatocytes induced by carbon tetrachloride or tertbutyl hydroperoxide in vitro (Daba and Abdel-Rehman, 1998) and in vivo (Nagi et al., 1999).
Most studies on BPA focused on its effect on reproduction and endocrine disruption and limited information considering the toxic effect on liver are available. Consequently, this study aimed to evaluate (1) The hepatotoxic effect of BPA on liver of male albino rats, (2) The possible role of thymoquinone in alleviating the detrimental effects of BPA on the selected organ.
Chemicals: Bisphenol A (purity: 99%) and thymoquinone were purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). All other chemicals used in the present study were of analytical grade. Kits were the products of Biodiagnostic Co. (Egypt), Biosystems Co. (Spain) and Randox Co (England).
Animals and experimental design: Thirty two male Sprague-Dawley rats (initially weighting 180-200 g) were used in the present study. They were obtained from the animal house of the Medical Research Institute, Alexandria University, Egypt. Rats were housed in conventional stainless steel cages at 23±2°C, 60±10% humidity and a 12 h light/12 h dark photoperiod. Animals were allowed standard laboratory rat food and water ad libitum. They were acclimatized to the laboratory environment for 2 weeks prior to the experiments and then were divided into 4 groups (n = 8). Rats of the first group were administered 0.3 mL olive oil and served as control. Bisphenol A at a dose of 10 mg kg-1 day-1 dissolved in 0.3 mL olive oil was given to rats of the second group for 4 weeks. This dose is higher than the current No Observed Adverse Effect Level (NOAEL). Animals of the third group received TQ at a dose of 10 mg kg-1 day-1 for 5 weeks. Rats of the fourth group received the same previous doses of BPA and TQ. Doses were given orally by gavage once daily. The BPA was given orally because this route of exposure is more relevant to human exposure. The TQ continued to be administered for 1 more week after finishing the 4 weeks of BPA administration.
Body and liver weight: The initial and final body weights were recorded at the onset and at the end of the experiment for the calculation of body weight change (%). After the experimental duration, the liver was excised and its relative weight (%) was determined in different experimental groups.
Blood sampling and preparation of serum: After 24 h of the last drugs administration, rats were sacrificed by cervical decapitation under light anesthesia with diethyl ether and the blood was collected into non-heparinized tubes. Serum was separated from blood by centrifugation at 5000 rpm for 10 min and was stored at -20°C for analysis.
Tissue sampling and preparation of liver homogenate: The abdominal cavities of the anesthetized rats were immediately opened and the livers were excised, perfused with normal saline to remove blood, blotted between filter papers and used for the preparation of tissue homogenate. About 0.5 g of each liver was homogenized in 4.5 mL of phosphate-buffered saline (PBS, pH 7.0, containing 1 mM EDTA). The crude tissue homogenate was then centrifuged at 8000 rpm for 30 min and the supernatant was collected and stored at 4°C for further analyses.
Estimation of lipid peroxidation level in liver homogenate: Lipid peroxidation (LPO) was determined by measuring thiobarbituric acid reactive species (TBARS) according to the method of Ohkawa et al. (1979). 1, 1, 3, 3-tetraethoxypropane, a precursor of malonaldehyde (MDA) was used as a standard for this assay. The amount of TBARS formed was measured spectrophotometrically at 532 nm and expressed as nmol of MDA equivalent/mg protein.
Determination of total hepatic antioxidant capacity: Total Antioxidant Capacity (TAC) was measured in serum by mean of commercial kit (Randox Co, England) according to the method of Rice-Evans and Miller (1994). The assay is based on the incubation of 2, 2-azino-di-(3-ethylbenzthiazoline sulphonate) (ABTS) with a peroxidase and hydrogen peroxide to produce the radical cation (ABTS+) which has a relatively stable blue-green color. It absorbs at 600 nm. The suppression of the color is compared with that of the Trolox which is widely used as a traditional standard for TAC measurement assays. Results are expressed as mmol equivalent Trolox/L.
Determination of non-enzymatic and enzymatic antioxidants in liver homogenate: Reduced glutathione (GSH) was analyzed spectrophotometrically at 405 nm using Elman’s reagent (5', 5'dithiobis 2-nitrobenzoic acid, DTNB) that becomes converted to 2-nitro-S-mercaptobenzoic acid per one mole of glutathione (Ellman, 1959). Results are expressed as nmol mg-1 protein. The activity of glutathione peroxidase (GPx) was determined as described by Paglia and Valentine (1967) where the GPx catalyzes the oxidation of glutathione. In the presence of glutathione reductase and NADPH the oxidized glutathione is immediately converted to the reduced form with a concomitant oxidation of NADPH to NADP+. The decrease in the absorbance (A340) is directly proportional to the GPx activity in the sample. Results were expressed in U/mg protein. The activity of glutathione S-transferase (GST) was determined spectrophotometrically at 340 nm using 1-cholro-2, 4 dinitrobenzene (CDNB) as a substrate and monitoring the change in absorbance due to thioester formation (Habig et al., 1974). Results are expressed as nmol mg-1 protein. The superoxide dismutase (SOD) activity in liver tissue was analyzed using nitroblue tetrezolium (NBT) as a substrate and phenazine methosulphate. The color intensity of the chromogen produced was measured at 560 nm. Results are expressed as U/mg protein (Kakkar et al., 1984). Catalase (CAT) activity was assayed according to the method of Aebi (1984) using H2O2 as substrate that can be decomposed by CAT enzyme. The reaction is stopped exactly after one min with CAT inhibitor. In the presence of peroxidase, the remaining H2O2 reacts with 3, 5-dichloro-2-hydroxybenzene sulfonic acid (DHBS) and 4-aminophenazone (AAP) to form a complex with color intensity that is inversely proportional to the amount of CAT in the original sample. Results were expressed in U/mg protein.
Liver function biomarkers in the serum: Transaminases namely alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were determined depending on incubation of substrate with the enzyme to produce phenylhydrazine which absorbs at 546 nm. The amount of phenylhydrazine formed is directly proportional to the enzyme quantity (Reitman and Frankel, 1957). Alkaline phosphatase (ALP) was assayed according to the method of Belfield and Goldberg (1971) where phenyl phosphate is transformed into phenol and phosphate in the presence of the enzyme. The liberated phenol is measured at 520 nm in the presence of 4-aminophenazone and potassium ferricyanide. Gamma Glutamyl Transferase (GGT) activity was determined by the method of Rosalki et al. (1970) using γ-glutamyl-p-nitroanilide as substrate. Total bilirubin was measured according to the method of Walter and Gerade (1970) depending on the reaction between bilirubin in the sample and the diazonium salt of sulphanilic acid to produce azobilirubin which shows a maximum absorption at 535 nm in an acid medium.
Serum chemistry: Serum biomarkers were determined using the assay kits and were performed according to the manufacturers’ protocols and instructions. Levels of total cholesterol and triglycerides were assayed according to the methods of Richmond (1973), Fossati and Prencipe (1982), respectively. High Density Lipoproteins (HDL) were estimated according to Warnick et al. (1982). Low-Density Lipoproteins (LDL) and very Low-Density Lipoprotein (vLDL) were calculated mathematically using the equation of Friedewald et al. (1972). Total protein and albumin concentrations were assayed according to the methods of Lowry et al. (1951) and Doumas et al. (1971), respectively.
Statistical analysis: Data is expressed as Mean±SEM. Data was analyzed using Statistical Package for Social Science (SPSS/Version 17.0) software. Significance between experimental groups was determined using one-way analysis of variance (ANOVA) followed by Least Signifcant Difference (LSD) test for comparison between two groups. The p-values less than 0.05 were considered statistically signifcant.
Effect of BPA on body and liver weight: Changes in body weight and relative weight of liver in the control and treated groups are presented in Fig. 1. No statistically significant changes in body weight were recorded between different experimental groups when compared to the control group.
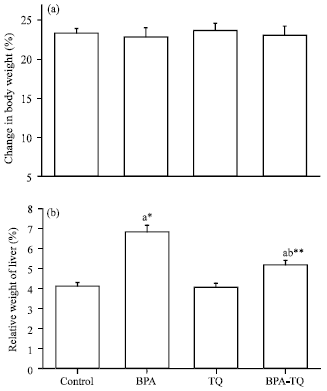 | |
| Fig. 1(a-b): | Effect of oral administration of bisphenol A (BPA) or thymoquinone (TQ) or both on (a) Body weight change (%) and (b) Relative weight of liver in male rats. Each value represents the Mean±SEM for each experimental group (n = 8), aTreated groups vs. the control, bBPA-TQ versus BPA group,*p<0.05, **p<0.01 |
Mean values were +22.81, +23.65 and +23.03% in BPA, TQ and BPA-TQ groups, respectively compared to +23.29% in the control group (Fig. 1a). The relative weight of liver was found to be 6.82% in BPA-treated rats which is significantly (p<0.05) higher compared to 4.10% in the control group. In contrary, a significant (p<0.01) decrease by -23.9% was observed in the relative weight of liver in rats administered TQ together with BPA, compared to BPA-treated rats although the value did not reach the normal value of the control group. Daily administration of TQ alone induced non-significant change in liver weight (Fig. 1b).
Effect of BPA and/or TQ on the level of LPO in hepatic tissue: The effect of BPA on LPO in the liver is presented in Table 1. According to this table, the level of TBARS (a marker for LPO) in BPA-treated rats was 11.33 nmol mg-1 protein which was more than four folds higher than the control animals. Administration of TQ for 5 weeks to BPA-treated rats resulted in a significant (p<0.05) decrease in the level of TBARS compared to the BPA group. The value was found to be 4.10 nmol mg-1 proteins compared to 11.33 nmol mg-1 protein in the BPA group. In contrast, non signifcant change in the level of TBARS was observed in animals treated with TQ alone compared to the control group.
Influence of BPA or TQ or both on the antioxidative status of the liver: According to Table 1, administration of BPA induced a marked suppression in the antioxidative status of the liver. Significant (p<0.05) decreases in the TAC (by -65.5%), the non-enzymatic antioxidant GSH (by -45.3%) and the enzymatic antioxidants namely GPx, GST, SOD and CAT (by -43.8, -28.7, -39.7 and -42.4%, respectively) were observed in the liver of BPA-treated rats. Supplementation with TQ at a dose of 10 mg kg-1 for 5 weeks significantly (p<0.05) normalized the suppressed antioxidants in the BPA-treated rats and to a large extent restored their values toward that of the control group (Table 1). It is worth to note that administration of TQ alone has a non-significant effect on the antioxidative status of the liver compared to the control group.
Effect of BPA and TQ on serum biomarkers related to hepatic dysfunction: The activities of liver marker enzymes and the level of total bilirubin in the serum of control and treated groups are shown in Table 2. Exposure to BPA was found to be associated with hepatic dysfunction as indicated by significant (p<0.05) increase in the activities of AST, ALT, ALP, GGT and in the level of total bilirubin. The mean values increased by +83.6%, +115.8, +45.8, +101.7 and +98.3%, respectively compared to the control group value. The results also revealed non-significant changes in these biomarkers in rats treated with TQ alone for 5 weeks as compared to the control. Administration of TQ together with BPA significantly (p<0.05) improved the liver functioning although it could not bring values of these indices to that of the control group.
Effect of BPA or TQ or both on serum chemistry: The changes in lipid profile and protein contents in the serum of the control and experimental groups are illustrated in Table 3.
| Table 1: | Effect of BPA or TQ or both on the oxidative/antioxidative status in liver of rats |
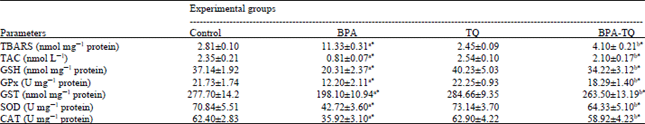 | |
| Each value represents the Mean±SEM for each experimental group (n = 8). aTreated groups vs. the control, bBPA-TQ vs. BPA group, *p<0.05, TBARS: Thiobarbituric acid reactive species, TAC: Total antioxidant capacity, GSH: Reduced glutathione, GPx: Glutathione peroxidase, GST: Glutathione S-transferase, SOD: Superoxide dismutase, CAT: Catalase | |
| Table 2: | Liver function biomarkers in the serum of different experimental groups |
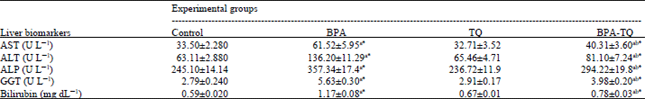 | |
| Each value represents the Mean±SEM for each experimental group (n = 8). aTreated groups vs. the control, bBPA-TQ vs. BPA group, *p<0.05, AST: Aspartate aminotransferase, ALT: Alanine aminotransferase, ALP: Alkaline phosphatase, GGT: γ-glutamyl transferase | |
| Table 3: | Effect of BPA or TQ or both on serum chemistry |
 | |
| Each value represents the Mean±SEM for each experimental group (n = 8). aTreated groups vs. the control, bBPA-TQ vs. BPA group, *p<0.05, **p<0.01. HDL: High density lipoproteins, LDL: Low density lipoproteins, vLDL: Very low density lipoproteins | |
Significant (p<0.05) increases in the level of total cholesterol (+39.7%), triglyceride (+45.1%), LDL (+92.7%) and vLDL (+45.1%) were recorded while that of HDL was significantly (p<0.05) decreased (-55.8%) in BPA-treated rats compared to the control animals. Administration of TQ to the BPA-treated rats resulted in a significant (p<0.01) decrease in the level of total cholesterol, triglycerides, LDL and vLDL compared to the BPA group while HDL level was significantly (p<0.01) increased although not reaching the control group value. On other hand, rats administered TQ alone for 5 weeks showed a non-significant change in these parameters compared to the control. Concerning total proteins and albumin, their values in the serum were significantly (p<0.01) decreased (by -38.8 and -32.3%, respectively) in response to BPA administration compared to the control group. TQ administration along with BPA could not improve the altered protein contents as indicated by the significant (p<0.01) difference between values in this group and that of the control group. Level of total proteins and albumin in rats treated with TQ alone showed non-significant changes when compared to the control animals (Table 3).
DISCUSSION
To date, there is a controversy about the toxicity of BPA. Although FDA has labeled BPA as a safe agent, newly emerging data has stirred discussion on the need of more studies about human health risk assessment of BPA exposure (Hugo et al., 2008). The present study revealed a disturbance in the oxidative/antioxidative status in rat liver with a concomitant impairment in its proper functioning as a result of BPA administration. Supplementation with TQ, the main bioactive constituent in the oil of Nigella sativa seed, protected the liver against BPA-induced hepatotoxicity and improved its antioxidative status.
Body and organ weights are fundamental benchmarks for toxicological studies. In the present study, body weight in BPA-treated rats did not show significant changes. Similar results were obtained with the administration of BPA for 30 days (Bindhumol et al., 2003). Also, Tyl et al. (2002) did not detect an effect for BPA on body weight. On the other hand, Yamasaki et al. (2002) found a statistically significant decrease in body weight in rats treated with BPA at a dose ≥466 mg kg-1 day-1. The present study revealed a significant increase in the relative weight of liver. Since liver is the primary site for getting rid of xenobiotics, the observed increase in its weight in the present study may represent a homeostatic mechanism to deliver more BPA into the liver for detoxification. Schulte-Hermann (1979) mentioned that liver enlargement may be due to a combination of hepatocyte hypertrophy and smooth endoplasmic reticulum proliferation which is presumed to stimulate a hepatic physiological adaptation to an increased workload demand.
Liver function tests are routinely used as diagnostic markers for hepatotoxicity. Aminotransfersases (ALT and AST), alkaline phosphatase (ALP) and γ-glutamyltransferase (GGT) are the major markers in monitoring the functional status of liver. The ALT activity is an important index to measure the degree of cell membrane damage while AST is an indicator of mitochondrial damage. The activity of ALP is often employed to assess the integrity of plasma membrane. The activity of GGT is a marker of hepatic injury. Increased levels of hepatic marker enzymes indicate the permeability and/or necrosis of hepatocytes (Goldberg and Watts, 1965). In the current study, daily administration of BPA for 4 weeks resulted in hepatic injury as indicated by elevation in the activity of ALT, AST, ALP, GGT and the level of total bilirubin in the serum. Elevated levels of these markers are familiar indicators for excess cellular leakage and loss of membrane integrity of hepatocytes (Dortman and Lawhorn, 1978). This suggests that BPA may act as a plasma membrane labilizer. These results are in a good agreement with previous studies with similar increases in serum indices for liver damage (Yamasaki et al., 2002; Korkmaz et al., 2010; Hassan et al., 2012). Moon et al. (2012) mentioned that BPA can induce hepatic damage and mitochondrial dysfunction by increasing oxidative stress in the liver. Treatment with TQ at a dose of 10 mg kg-1 significantly suppressed the BPA-induced elevation in these hepatic biomarkers. The efficacy of TQ in protecting hepatic enzyme leakage may be related to its ability to preserve the structural and functional integrity of the liver against the adverse effects of BPA as well as repair of hepatic tissue damage caused by BPA. These results come in concert with studies which indicated the heaptoprotective effect of TQ in models of liver injury in vitro (Daba and Abdel-Rehman, 1998) and in vivo (Alsaif, 2007). Thymoquinone was found to reduce hepatic dysfunction through its membrane stabilization properties against reactive oxygen species (ROS)-mediated hepatocellular injury (Basiglio et al., 2009).
The correlation between oxidative stress, antioxidant potential and development of chronic diseases has become evident. Oxidative stress adversely alters many crucial biological molecules leading to loss of form and function. In the present study, BPA elicited a marked increase in the level of TBARS which indicates enhanced LPO. Bisphenol A has been proposed to induce hepatic damage and mitochondrial dysfunction through induction of oxidative stress (Bindhumol et al., 2003; Moon et al., 2012). It generates ROS and induces cellular apoptosis in hepatocytes leading to an increase in the level of TBARS (Korkmaz et al., 2010). Previous studies have also illustrated elevated levels of TBARS in the brain, testes and kidneys of male rats exposed to BPA (Aydogan et al., 2008, 2010). Bisphenol A decomposes into many metabolites probably including BPA radical by reactions of radical oxygen (Saki, 2001). Increased LPO can be counteracted by administrating antioxidant molecules. In the present study, treatment of rats with TQ significantly reduced the BPA-induced oxidative stress as indicated by the reduction in TBARS production in hepatic tissue. The protective effect of TQ can be attributed to its strong scavenging properties and its ability to reduce free radicals production. The antioxidant activity of TQ has been previously reported (Houghton et al., 1995; Nagi and Mansour, 2000). Quinone reductase is an enzyme that catalyses the reduction of quinones to hydroquinones and prevents their subsequent participation in the generation of ROS (Lind et al., 1982). Thymoquinone, as a substrate for quinone reductase, showed a significant induction in the activity of this enzyme in hepatic tissue (Nagi et al., 1999).
Cells are endowed with adequate antioxidants which represent the cell’s buffering systems to prevent the propagation of free radical reactions. In the current study, a reduction in the total antioxidant capacity in the serum of BPA-treated rats was reported. The GSH which is considered the most prevalent intracellular non-enzymatic antioxidant, has a crucial role as a ROS scavenger. In the current study, GSH content was significantly reduced due to BPA administration. This could be explained by the increased consumption of GSH in scavenging the BPA-generated ROS. This result is consistent with previous studies which reported similar decrease in hepatic GSH in rats administered BPA (Korkmaz et al., 2010; Hassan et al., 2012). The reduction of GSH content indicates failure of primary antioxidative system to act against free radicals. GPx, GST, SOD and CAT constitute a mutually supportive team of antioxidant enzymes which provide a defense system against ROS. Damage at the cellular level by oxidants is attenuated by these antioxidant enzyme. The current study showed that there was a significant decline in hepatic activity of these endogenous antioxidant enzymes in BPA-treated rats. These results are in concordance with Hassan et al. (2012) and Sangai et al. (2012). These results reflect the inability of the liver to eliminate free radicals and indicate that BPA induces oxidative stress by disturbing the balance between radicals and antioxidant defense system in liver. Reduction in the activity of these enzymes might be due to reduced enzyme synthesis by the injured hepatic cells or depletion of these enzymes by the ROS overproduction. Upon supplementation with TQ, the activities of the antioxidant enzymes in rat hepatic tissue were significantly restored. It is therefore suggested that TQ might increase the bioavailability of these enzymes possibly by reducing the damage to the hepatic cells as found in this study. Thymoquinone was found to upregulate the GST, GPx and CAT genes with the consequent elevation in their activities to overcome oxidative stress (Ismail et al., 2010).
Liver is the principle organ for maintaining the body’s internal environment. It plays a key role in the control of lipid metabolism. Results of the present study revealed a disturbance in lipid profile as reflected by the significant increase in the level of total cholesterol, triglycerides, LDL, vLDL with a concomitant decrease in HDL. The liver has a complex network of nuclear receptors that coordinately regulates the expression of enzymes involved in lipid metabolism, from fatty acid oxidation and uptake to triacylglycerol synthesis, accumulation and/or secretion (Nguyen et al., 2008). Bisphenol A has the potential to affect lipid and glucose homeostasis in various tissues by interfering with different nuclear receptors involved in regulation of metabolism (Casals-Casas and Desvergne, 2011). Bisphenol A has also found to stimulate lipid accumulation and up-regulate genes involved in lipid metabolism in adipocytes (Masuno et al., 2002). The present study showed that TQ supplementation had favorably modified serum lipid profile in rats with significant decreases in total cholesterol, triglycerides, LDL, vLDL and increased HDL. Thymoquinone was reported to have the ability to significantly reduce the lipid profile (Bamosa et al., 2002; Zaoui et al., 2002). The mechanism underlying this hypolipidemic effect is not fully understood. The antilipidemic action of TQ may reside in its ability to stimulate insulin secretion and action (Fararh et al., 2002). It was also suggested that the hypolipidimic activity of TQ may be attributed to inhibition of oxidative stress (Sinha et al., 2008).
Serum proteins are invariably synthesized by liver. In the present study, BPA was found to affect the synthetic function of the liver as indicated by the significant decrease in the level of total proteins and albumin. The reduction in serum proteins with impaired liver enzymes are indications of impaired synthetic function of the liver. The observed decrease in biosynthesis and secretion of proteins might be due to formation of BPA (or its metabolites) adducts with DNA or RNA. Izzotti et al. (2009) confirmed the ability of BPA to form DNA adducts both in vitro and in vivo in rodent liver. Bisphenol A is converted to bisphenol O-quinone which might be the ultimate DNA binding metabolite (Atkinson and Roy, 1995). This binding might prevent RNA polymerase from transcribing the DNA and can inhibit the formation of mRNA. A failure in mRNA formation results in an inhibition of protein synthesis, which may be considered to be the cause of the liver cell necrosis (Korkmaz et al., 2010). Supplementation with TQ to BPA-treated rats could not improve the reduction in the levels of total proteins and albumin. This can be attributed to inadequate dose or duration for TQ administration to improve the synthetic function of the liver.
Based on the data of the current study, it may be concluded that BPA has detrimental effect on liver of male rats. Also it supports the possibility that thymoquinone plays a notable role in protecting the liver against BPA-induced toxicity and oxidative stress in male rats. The strong antioxidative activity could partially be the mechanism underlying the protective effects of thymoquinone. The versatility and potency of thymoquinone make it potential candidates for therapeutic and preventive drugs for hepatotoxicity resulting from toxicants, including BPA.