Research Article
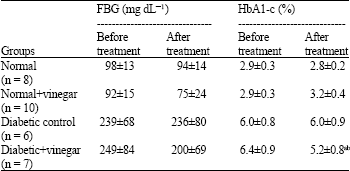
Apple Cider Vinegar Attenuates Lipid Profile in Normal and Diabetic Rats
Department of Nutrition, Para-Medical School, Ahvaz Jundishapour University of Medical Sciences, Ahvaz, Islamic Republic of Iran
A. Mansoori
Department of Nutrition, Para-Medical School, Ahvaz Jundishapour University of Medical Sciences, Ahvaz, Islamic Republic of Iran
A.R. Sarkaki
Department of Physiology, Faculty of Medicine, Ahvaz Jundishapour University of Medical Sciences, Ahvaz, Islamic Republic of Iran
M.T. Jalali
Department of Laboratory Sciences, Para-Medical School, Ahvaz Jundishapour University of Medical Sciences, Ahvaz, Islamic Republic of Iran
S.M. Latifi
Department of Biostatistics, Faculty of Health, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Islamic Republic of Iran









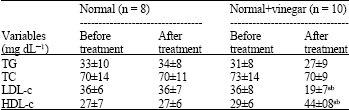

Niz Thaha - Singapore Reply
Very useful paper in our study.
However, we wish to know what is the active ingredient found in the apple juice, and the variety of apple suitable as effective (red or green).
Also if apple vinegar is applicable to Type 1 or 2 diabetic patients.
Further research is highly recommended and we are willing to be partners in your study.
Regards / niz