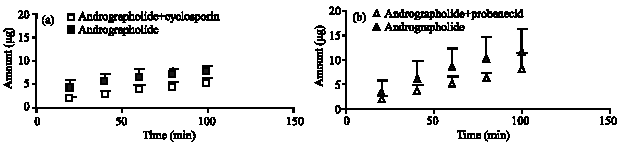
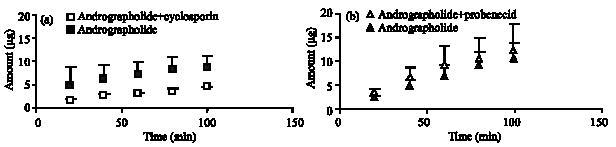Research Article
Membrane Transport of Andrographolide in Artificial Membrane and Rat Small Intestine
Faculty of Pharmaceutical Sciences, Khan Kaen University, Khan Kaen, Thailand
Jinda Wangboonskul
Faculty of Pharmaceutical Sciences, Khan Kaen University, Khan Kaen, Thailand
Kanokwan Jarukamjorn
Faculty of Pharmaceutical Sciences, Khan Kaen University, Khan Kaen, Thailand
Bung-orn Sripanidkulchai
Faculty of Pharmaceutical Sciences, Khan Kaen University, Khan Kaen, Thailand
Teruo Murakami
Faculty of Pharmaceutical Sciences, Hiroshima International University, Kure, Japan