Research Article
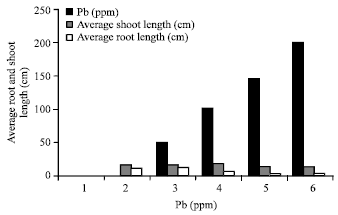
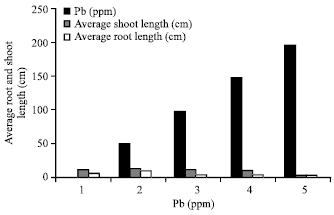
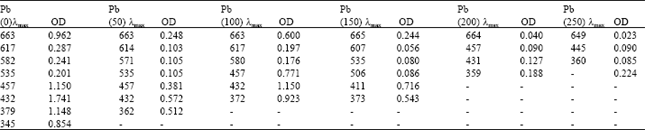
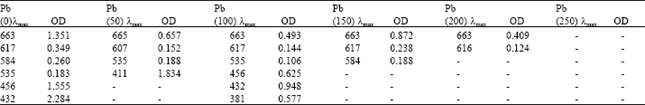
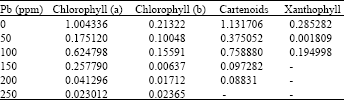
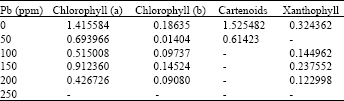
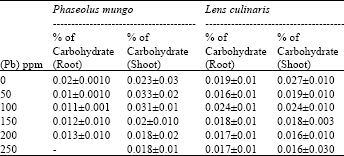
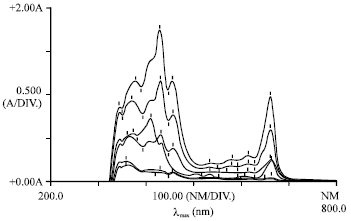
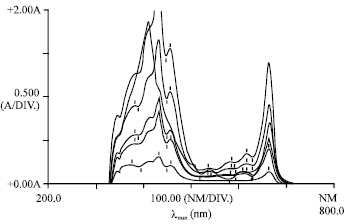
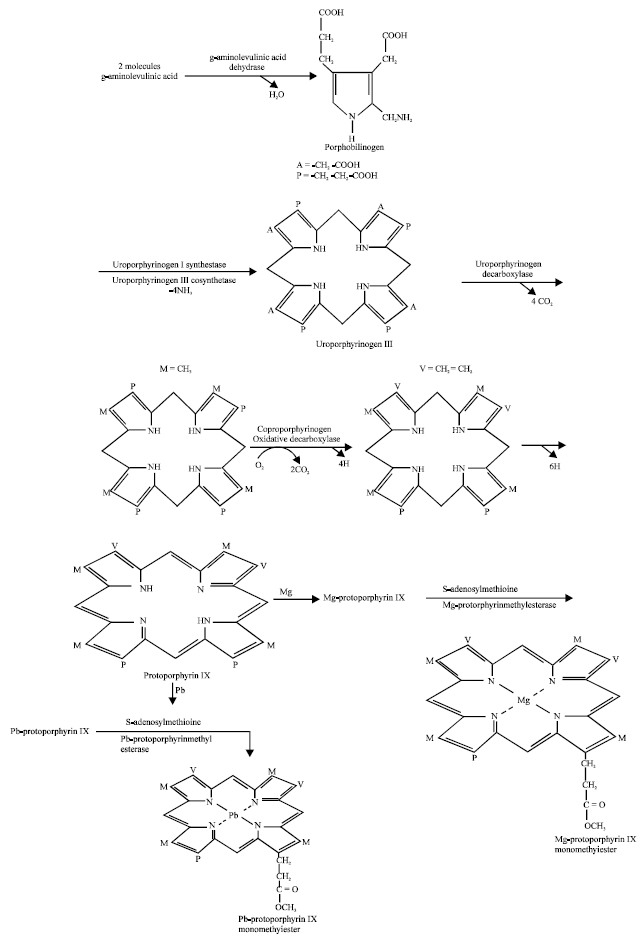
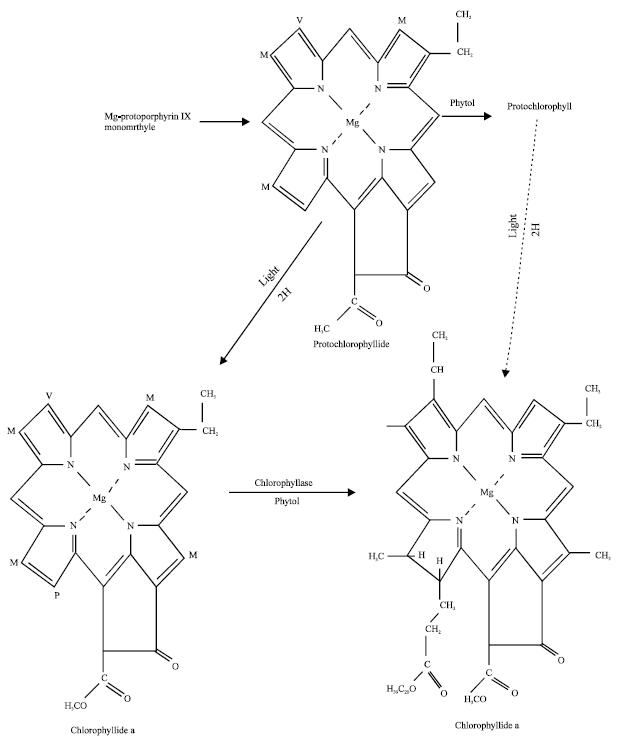
Phytotoxicity of Pb: II. Changes in Chlorophyll Absorption Spectrum due to Toxic Metal Pb Stress on Phaseolus mungo and Lens culinaris
Department of Botany and Jinnah University for Women 5-C, Nazimabad 74600, Karachi,
Salma Kanwal
Department of Chemistry, Jinnah University for Women 5-C, Nazimabad 74600, Karachi,
Fahim Uddin
University of Karachi, Pakistan
Rafia Azmat
Department of Chemistry, Jinnah University for Women 5-C, Nazimabad 74600, Karachi,