Research Article
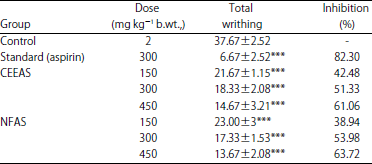
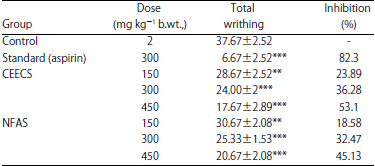
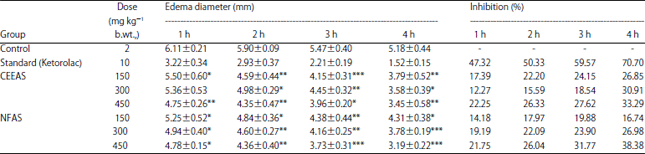
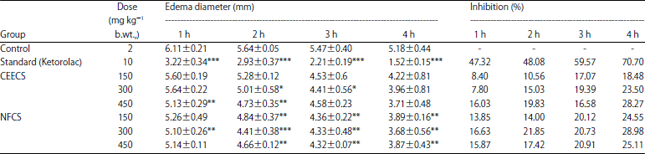
Investigation of in vivo Analgesic, Anti-Inflammatory, in vitro Membrane Stabilizing and Thrombolytic Activities of Atylosia scarabaeoides and Crotalaria spectabilis Leaves
Department of Pharmacy, Southeast University, Dhaka, Bangladesh
Md. Rafiqul Islam
Department of Pharmacy, Southeast University, Dhaka, Bangladesh
Tahmida Khanom Chowdhury
Department of Pharmacy, Southeast University, Dhaka, Bangladesh
Abdur Rahman
Department of Pharmacy, Southeast University, Dhaka, Bangladesh
Md. Sahab Uddin
Department of Pharmacy, Southeast University, Dhaka, Bangladesh
LiveDNA: 880.11658
Sabiha Ferdowsy Koly
Department of Pharmacy, Southeast University, Dhaka, Bangladesh
Md. Shahid Sarwar
Department of Pharmacy, Noakhali Science and Technology University, Noakhali, Bangladesh
LiveDNA: 880.16609