Research Article
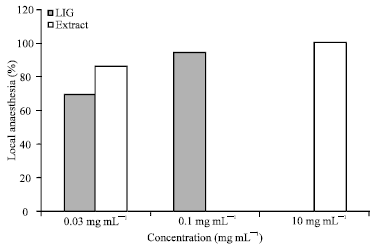
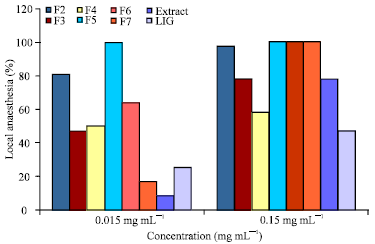
Local Anesthetic and Tissue Effects of the Leaf Extract and Fractions of Sterculia tragacantha Lindl
Department of Veterinary Surgery, University of Nigeria, Nsukka, Nigeria
Uzoma I. Asuzu
Department of Veterinary Physiology and Pharmacology, University of Nigeria, Nsukka, Nigeria
Raphael O.C. Kene
Department of Veterinary Surgery, University of Nigeria, Nsukka, Nigeria
Sunday O. Udegbunam
Department of Veterinary Physiology and Pharmacology, University of Nigeria, Nsukka, Nigeria
Chinaka O. Nwaehujor
Department of Veterinary Physiology and Pharmacology, University of Nigeria, Nsukka, Nigeria