Research Article
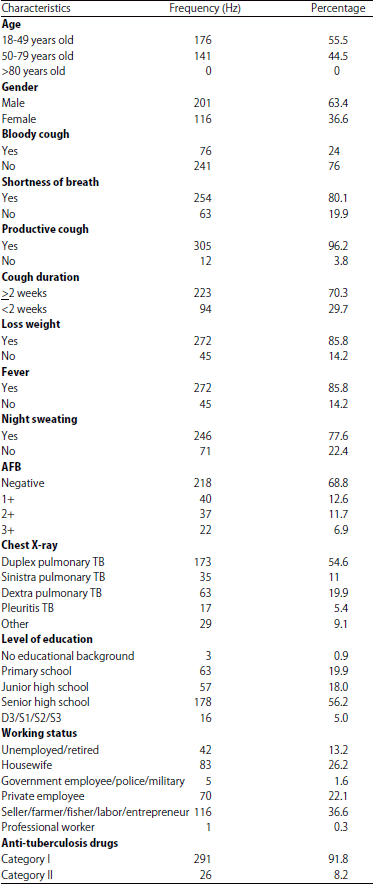
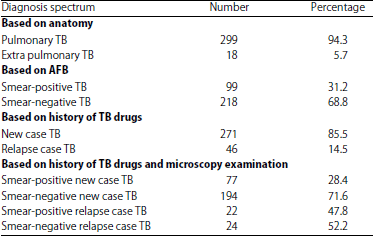
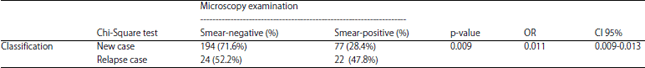
Diagnosis of a Spectrum of Pulmonary Tuberculosis at Islam Hospital Sukapura, Jakarta, Indonesia: A Retrospective Study of 317 Cases
Department of Clinical Pathology, Faculty of Medicine and Health, University of Muhammadiyah Jakarta, Indonesia
Mochammad Hatta
Molecular Biology and Immunology Laboratory, Faculty of Medicine, Hasanuddin University Makassar, Indonesia
LiveDNA: 62.16730
Muhammad Nasrum Massi
Molecular Biology and Immunology Laboratory, Faculty of Medicine, Hasanuddin University Makassar, Indonesia
Indah Pratiwi
Department of Clinical Pathology, Faculty of Medicine and Health, University of Muhammadiyah Jakarta, Indonesia
Muhammad Fachri
Department of Pulmonology and Respiratory Medicine, Faculty of Medicine and Health, University of Muhammadiyah Jakarta, Indonesia
Slamet Sudi Santoso
Department of Medical Education, Faculty of Medicine and Health, University of Muhammadiyah Jakarta, Indonesia
Amir Syarifuddin
Department of Medical Education, Faculty of Medicine and Health, University of Muhammadiyah Jakarta, Indonesia
Ressy Dwiyanti
Molecular Biology and Immunology Laboratory, Faculty of Medicine, Hasanuddin University Makassar, Indonesia
Rizki Amelia Noviyanthi
Molecular Biology and Immunology Laboratory, Faculty of Medicine, Hasanuddin University Makassar, Indonesia