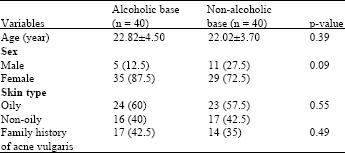
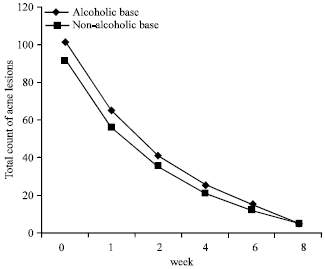
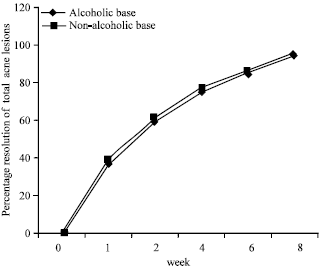
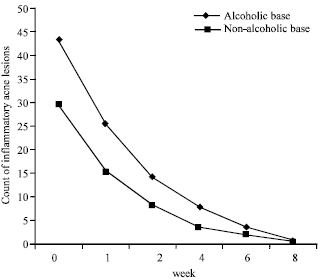
Research Article
Efficacy of 10% Azelaic Acid Gel with Hydro-alcoholic or Alcohol-free Bases in Mild to Moderate Acne Vulgaris; the First Clinical Trial
Department of Dermatology, Sina Hospital, Tabriz University of Medical Sciences, Tabriz, Iran
Shahla Babaeinejad
Department of Dermatology, Sina Hospital, Tabriz University of Medical Sciences, Tabriz, Iran
Mehdi Amirnia
Department of Dermatology, Sina Hospital, Tabriz University of Medical Sciences, Tabriz, Iran
Javad Shokry
Department of Dermatology, Sina Hospital, Tabriz University of Medical Sciences, Tabriz, Iran
Elham Razzagh Karimi
Department of Dermatology, Sina Hospital, Tabriz University of Medical Sciences, Tabriz, Iran
Daniel F. Fouladi
Drug Applied Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
Kamran Sedaghat
School of Medicine, Tabriz University of Medical Sciences, Tabriz, Iran