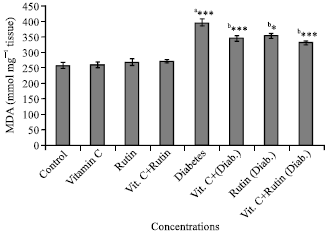
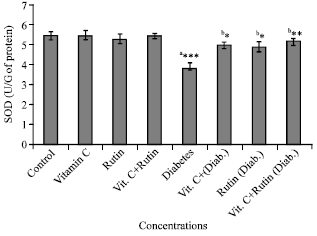
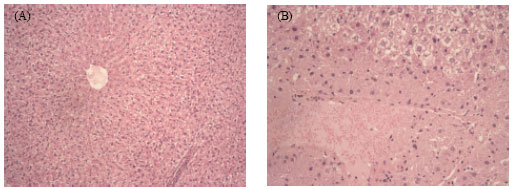
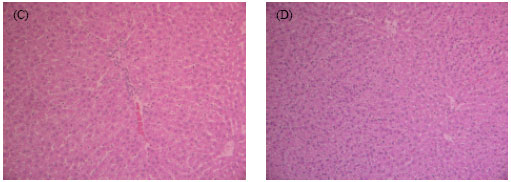
Research Article
Combined Treatment of Rutin and Vitamin C Improves the Antioxidant Status in Streptozotocin-Induced Diabetic Rats
Department of Community Health Sciences, College of Applied Medical Sciences, King Saud University, Riyadh, Saudi Arabia