Research Article
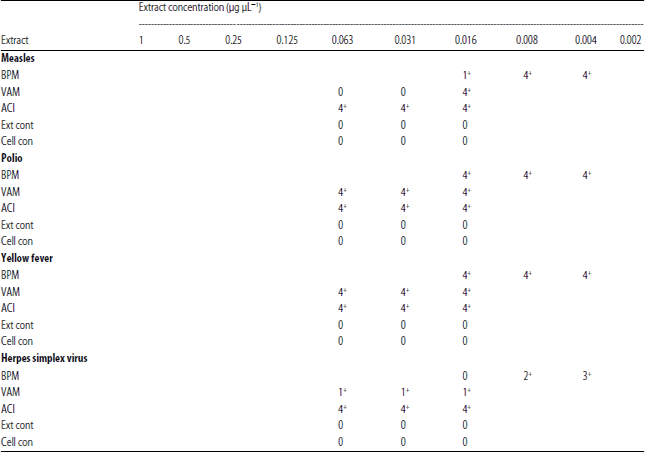
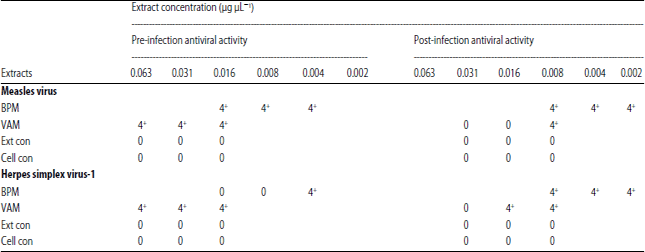
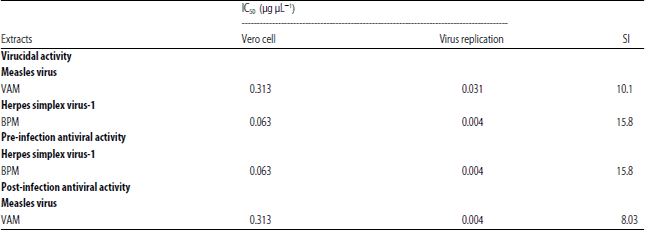
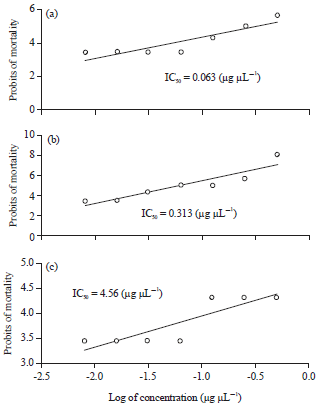
In vitro Antiviral Activities of Bryophyllum pinnatum (Odaa opuo) and Viscum album (Awuruse)
Department of Microbiology, School of Biological Sciences, Federal University of Technology, Owerri, Imo State, Nigeria
LiveDNA: 234.25146
Juliet Adamma Shenge
Department of Virology, College of Medicine, University of Ibadan, Nigeria