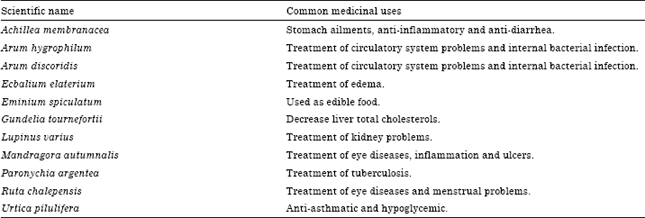
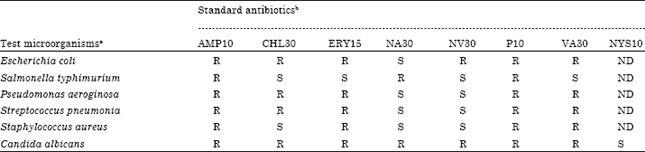
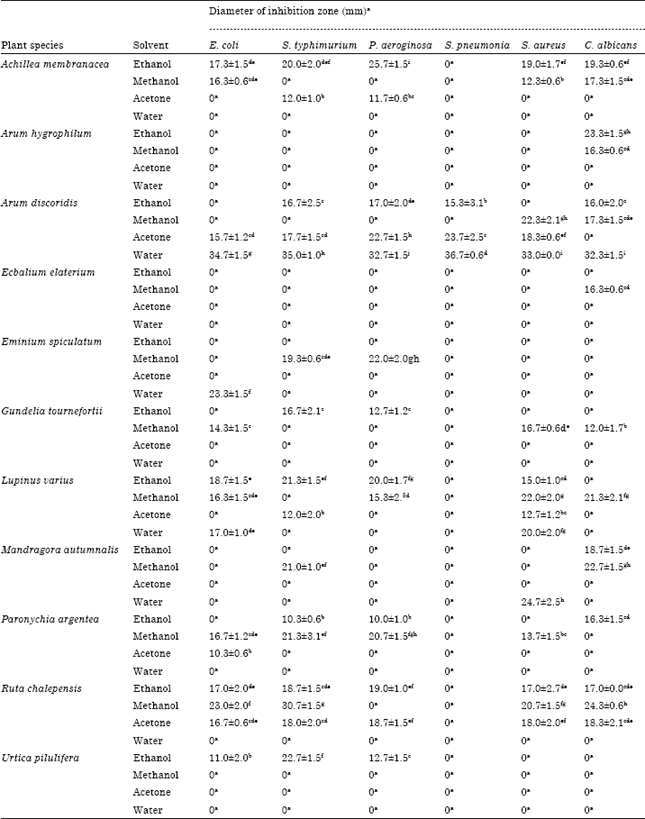
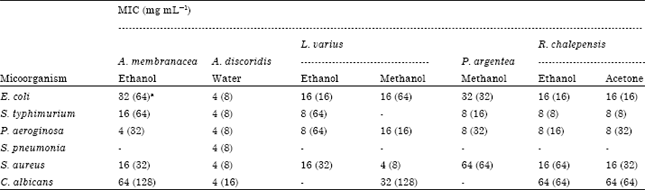
Research Article
Antimicrobial Activity of Crude Extracts of Some Plant Leaves
Department of Biotechnology, Faculty of Agricultural Technology, Balqa� Applied University, 19117, Al-Salt, Jordan
Mohamad Shatnawi
Department of Biotechnology, Faculty of Agricultural Technology, Balqa� Applied University, 19117, Al-Salt, Jordan
Mohammad Al-alawi
Department of Plant Production and Protection, Faculty of Agricultural Technology, Balqa� Applied University, 19117, Al-Salt, Jordan
Enas Al-Zu`bi
Department of Biotechnology, Faculty of Agricultural Technology, Balqa� Applied University, 19117, Al-Salt, Jordan
Hanee Al-Dmoor
Department of Nutrition and Food Processing, Faculty of Agricultural Technology, Balqa� Applied University, 19117, Al-Salt, Jordan
Maisa Al-Qudah
Department of Allied Medical Sciences, Faculty of Al-Zarqa, Balqa� Applied University, Al-Zarqa, Jordan
Jafar El-Qudah
Department of Nutrition and Food Processing, Faculty of Agricultural Technology, Balqa� Applied University, 19117, Al-Salt, Jordan
Ismael Otri
Department of Biotechnology, Faculty of Agricultural Technology, Balqa� Applied University, 19117, Al-Salt, Jordan