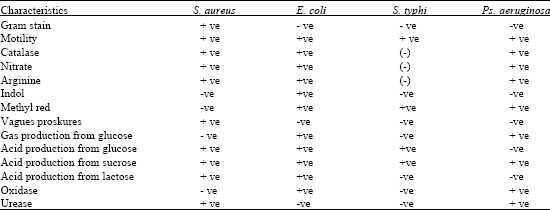
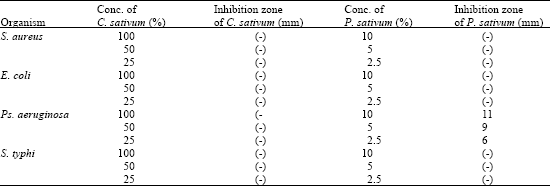
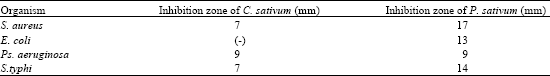
Research Article
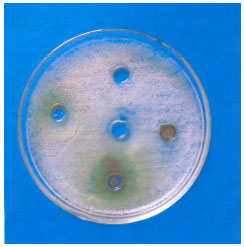
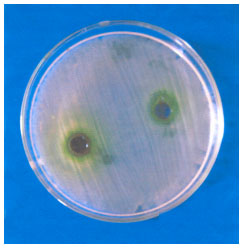
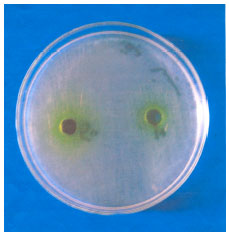
Antimicrobial Activity of Petroselinum sativum and Coriandrum sativum Seeds
Collage of Veterinary Medicine and Animal Production, Sudan University of Science and Technology, P.O. Box 204, Khartoum North, Sudan
Sabna D. Mohammed
Faculty of Science and Technology, El Neelain University, P.O. Box 12702, Khartoum, Sudan
Samia, M.A. El Badwi
Department of Veterinary Medicine, Pharmacology and Toxicology, University of K bartoum, P.O. Box 32, Khartoum North, Sudan
Warda, S. Abdel Gadir
Food Research Center, Ministry of Science and Technology, P.O. Box 213, Khartoum North, Sudan
Abdel-Gadir H. Alkhatim
Faculty of Science and Technology, El Neelain University, P.O. Box 12702, Khartoum, Sudan
S.E. I. Adam
Faculty of Science and Technology, El Neelain University, P.O. Box 12702, Khartoum, Sudan