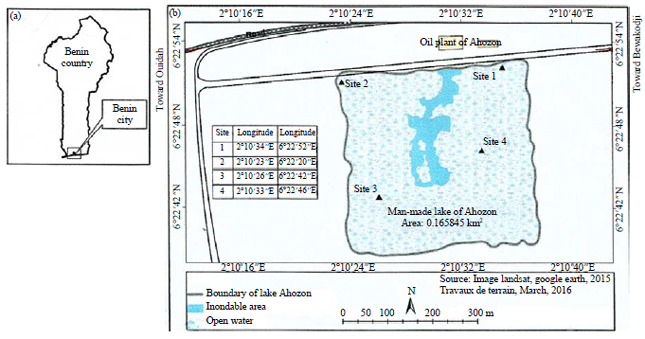
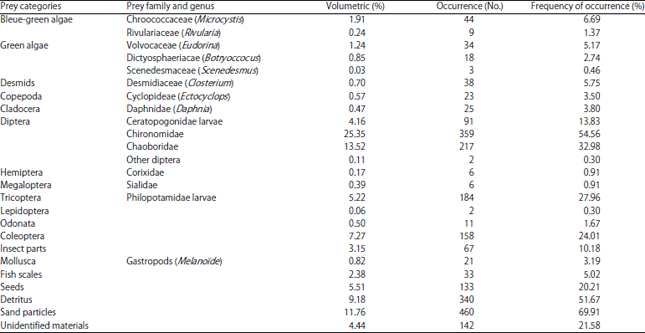
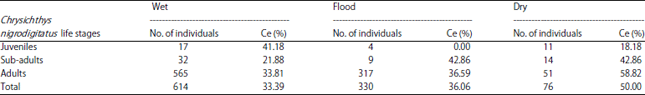
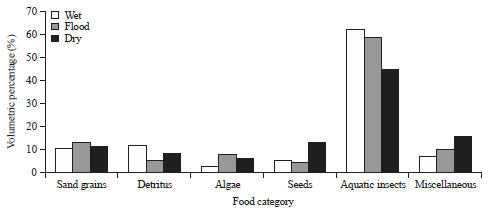
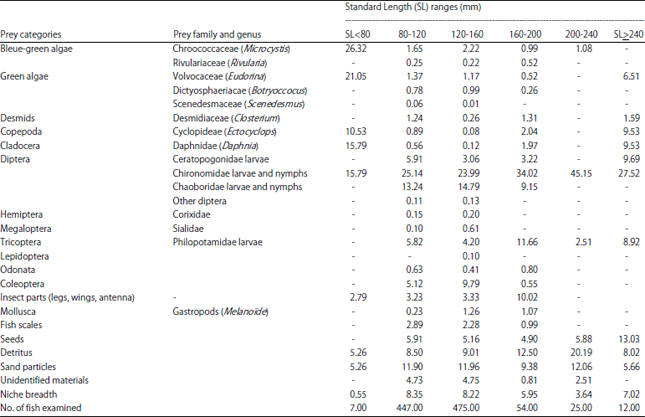
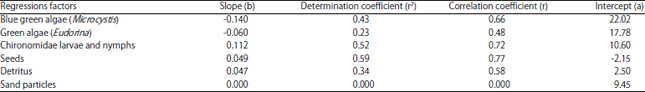
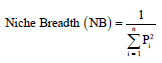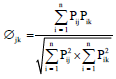Research Article
Trophic Ecology and Establishment of the Silver Catfish, Chrysichthys nigrodigitatus (Pisces: Siluriformes: Claroteidae) Introduced in an Artificial Pond of Benin, West Africa
Laboratoire d`Ecologie et de Management des Ecosyst�mes Aquatiques (LEMEA), Departement de Zoologie, Faculte des Sciences et Techniques, Universite d`Abomey-Calavi, B.P. 526 Cotonou, Benin
Alphonse Adite
Laboratoire d`Ecologie et de Management des Ecosyst�mes Aquatiques (LEMEA), Departement de Zoologie, Faculte des Sciences et Techniques, Universite d`Abomey-Calavi, B.P. 526 Cotonou, Benin
Youssouf Abou
Laboratoire d`Ecologie et de Management des Ecosyst�mes Aquatiques (LEMEA), Departement de Zoologie, Faculte des Sciences et Techniques, Universite d`Abomey-Calavi, B.P. 526 Cotonou, Benin