Research Article
A Description of Some Ectoparasites of the Wall Gecko
Department of Pathology, College of Health Sciences, Usmanu DanFodiyo University, Sokoto, Nigeria
Wall geckos are a group of small lizards which have become established in the vicinity of man in the tropics[1]. In Nigeria these reptiles inhabit dark abode of the home, where they are seen, often crawling across walls and ceilings and other surfaces of building. They move with remarkable agility, usually in search or pursuit of insect preys. Like all vertebrates, these reptiles are also susceptible to parasitism in their niche[2,3]. In this study some ectoparasites recovered from the body of some wall geckos are described. A study of the ectoparasites of the wall gecko is desirable because of the co-habitation of these lizards with man and livestock in the domestic environment. This is important because some parasites of the lizards have the potential to infest mammals[4]. The significance of a possible zoonotic involvement of parasites seen in this study is also indicated.
Specimen of gecko studied were collected from Jos, Plateau State, which is located between 10° 30"-09° 00" North and 09°30"-08°30" East, about Central Nigeria. Animals were caught with the aid of a sweep-net or a locally made straw-basket trap. Captured animals were put in separate clean covered collecting-trays in which some cotton wool sprinkled with chloroform had been placed. Sacrificed animals were transferred to small glass trays and each lizard was examined systematically under the dissecting microscope. Ectoparasites seen were carefully extracted using tweezers, scalpel or fine combs. The location of parasites on the body of host animal was noted and recorded. An extracted parasite was transferred into a drop of paraffin lubricating oil on a microslide and examined under the compound microscope[5]. For the purpose of identification, ectoparasites were preserved in caustic potash, dehydrated in graded series of alcohol and mounted for closer observation of detail structures[6].
Ectoparasites recovered from the wall gecko were all acarines, including ticks and mites. Host lizards were parasitized in a mixed infestation. Parasites seen were generally microscopic or just visible with the naked eye as tiny specks. They possessed membranous tegument and distinct capitulum with no obvious divisions between the cephalothorax and the abdominal region. 381 (73.3%) of the parasites recovered (n = 520), occurred on the back part of 49 (70.0%), out of the 70 host animals examined (Table 1). Parasites also occurred (n = 520) on the head (25 or 4.8%), neck (33 or 6.65%), limbs (46 or 8.85%) and eye-shield (31 or 5.96%) of infested animals (Table 1). In all cases they were found with capitulum partially buried just beneath the scales and were all engorged, obviously with host blood and fluid. This is because host blood cells, as seen under the microscope, were identical with blood cells squeezed from abdomen of ectoparasites extracted from the body of the host animals. Acarines isolated from host gecko were identified[4,7,8] as follows:
(i) | Trombiculid ectoparasites which infested 45 or 76.3% (n = 70) of parasitized host lizards were the only type of mites found on the animals examined (Table 2). Engorged adult and larval mites measured on the average 0.4-0.25 mm and 0.3-0.24 mm on the average (Fig. 1 and 2). They were pale orange-red, almost transparent and velvet-skinned minute acarines with hair-like protuberances covering most of the body. The parasites were identified as species of the genera Trombicula because they had blade-like chelicerae and the coxae of the four-pairs of legs in the adult were incorporated into the ventral surface. |
| Table 1: | Percentage anatomical distribution of acarine on gecko body |
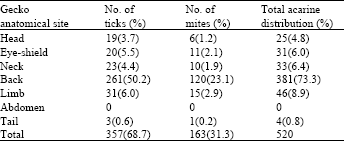 | |
| Figures in brackets are percentages | |
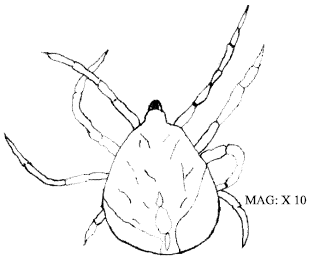 | |
| Fig. 1: | Trombicula spp. (adult) |
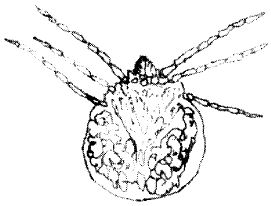 | |
| Fig. 2: | Trombicula spp. (larva) |
| (ii) | Argasid (soft) ticks which were the other ectoparasites seen occurred on 18 or 30.5% (n = 70) of the parasitized geckos (Table 2). At full engorgement parasites were swollen into rotund pale yellow ticks with legs which were short in proportion to the body (Fig. 3). Organisms had inferior capitulum, no scutum and structurally wider than they were long (Fig. 3 and 4). The ectoparasites measured 0.45-0.48 mm on the average and most likely belong to the genera Argas because they had spherical cuticular discs and fine hair-like dorso-integumental protuberances which were indistinct dorsally but regularly spaced at the lateral margin of the body (Fig. 3 and 4). |
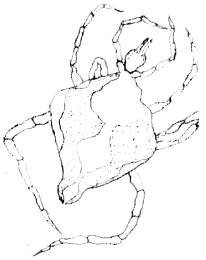
| (iii) | Ixodid (hard) ticks were also found to infest 28 or 47.5% (n = 70) of the geckos examined (Table 2). The parasites appeared translucent and colorless with thin, shiny dorsal shield which was convex rather than broadly rounded at the posterior margin. The capitulum was prominent, with the basis capituli well developed extending laterally, but the anal groove was indistinct and scutal festoons absent (Fig. 5). Organism measured 0.60-0.45 mm on the average and identified as species of the genera Aponoma. Inference was based on organism’s second palpal segment which was much longer than wide (Fig. 5). |
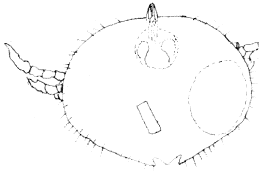 | |
| Fig. 3: | Argas spp. |
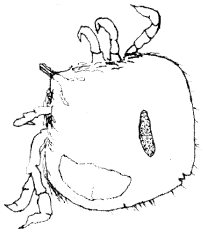 | |
| Fig. 4: | Argas spp. |
| Table 2: | Percentage number of gecko parasitized by acarines |
 | |
| Figures in bracket are percentages | |
 | |
| Fig. 5: | Aponomma spp. |
 | |
| Fig. 6: | Margaropus spp. |
A few of the ixodid ticks found on the geckos were probably species of Margaropus because the pre-anal plates of each organism continued as two prongs, one on either sides of the anal region (Fig. 6). Organism’s 4th pairs of legs were notably enlarged compared with the other pairs of legs.
Ticks and mites are distributed the world over on a wide range of vertebrate hosts[8]. It is therefore inferred from this study that the wall gecko is one of such animal ectoparasitized. Other lizards are commonly infested by an acarine fauna similar in morphology and bio-ecology to those which attack the wall gecko[4,7]. Such similarities seen among the acarines is useful in the understanding of related ectoparasites of medical and veterinary significance[8,9]. A major difference however is that ectoparasites of the gecko in this study were generally minute in size and possessed membranous tegument, as compared with the leathery covering of the larger acarines which infest other higher vertebrates[8]. Nevertheless the gecko acarines resemble those of other vertebrates by their blood ingesting habit. Indeed previous studies had shown that ticks and mites of cold-blooded animals also infested warm-blooded vertebrates[8], including man and livestock[4,9]. For instance Trombicula splendens and T. alfrenddugesi are partial to reptiles, attacking snakes and lizards, but also parasitize birds and domestic animals[7]. It is therefore likely that acarines of the gecko as observed in this study may feed on man and livestock and by the virtue of their blood feeding habit also transmit diseases of the gecko[2,10,11] to man or domestic animals[4,7]. It had been suggested that parasites of the wall gecko are of possible zoonotic significance[12]. The co-habitation of this reptile with man and livestock in a common domestic abode may therefore easily enhance the developmental or parasitic life-cycle of the gecko ectoparasites. In another light, studies have shown that some species of Aponoma ticks live mainly on reptiles in the tropics and also show preference for monotreme mammals[13]. This affinity to such an archaic host may be of additional importance in the understanding of ixodid evolution of parasitism[14,15].