Research Article
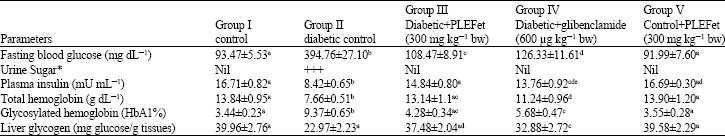
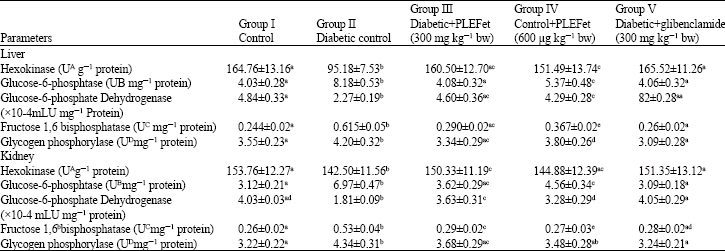
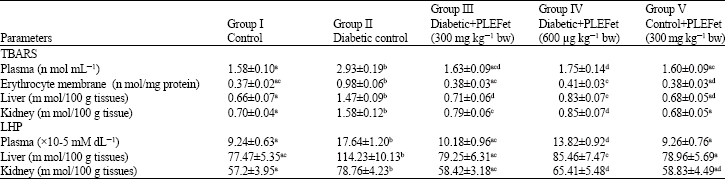
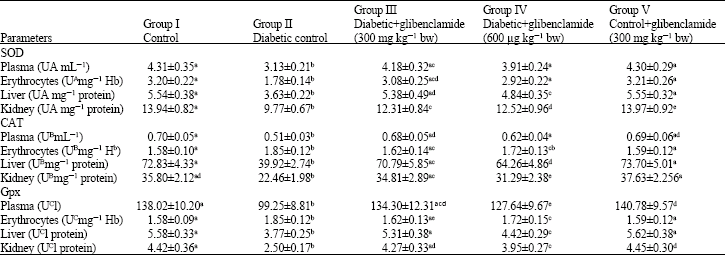
Antihyperglycemic and Antilipidperoxidative Effects of Piper longum (Linn.)Dried Fruits in Alloxan Induced Diabetic Rat
Department of Biochemistry, Faculty of Science, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India
Simon Silvan
Department of Biochemistry, Faculty of Science, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India
Krishnamoorthi Vasudevan
Department of Zoology, Faculty of Science, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India
Subramanian Balakrishnan
Department of Biochemistry, Faculty of Science, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India