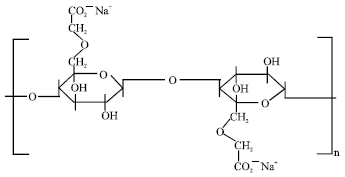
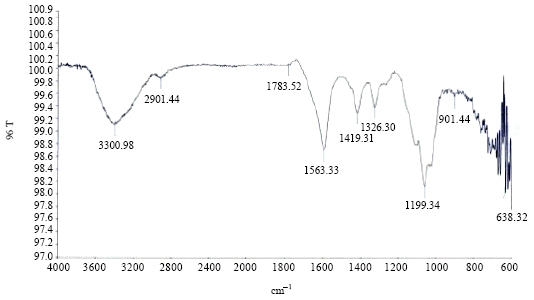
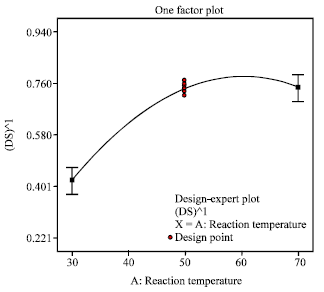
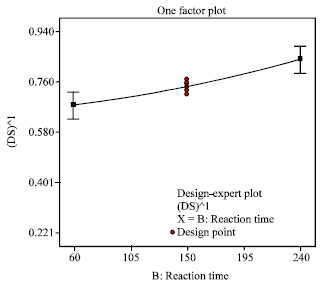
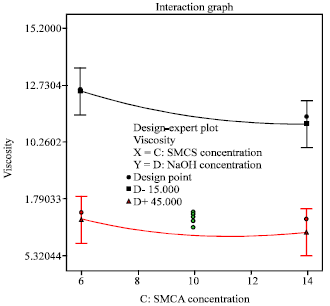
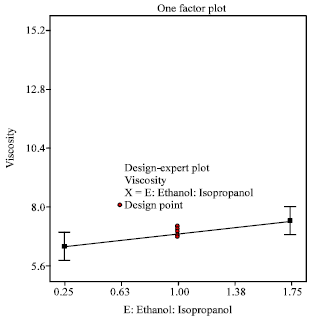
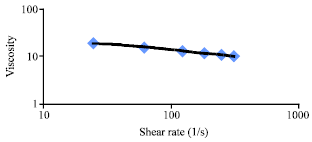
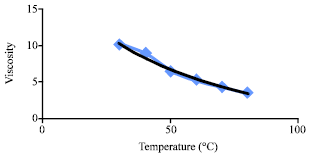
Research Article
Optimization of Reaction Conditions for Preparing Carboxymethylcellulose
Chemical Engineering Programme, School of Engineering and Information Technology, Niversiti Malaysia Sabah, Locked Bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia
A. Bono
Chemical Engineering Programme, School of Engineering and Information Technology, Niversiti Malaysia Sabah, Locked Bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia
A.C.R. Valintinus
Chemical Engineering Programme, School of Engineering and Information Technology, Niversiti Malaysia Sabah, Locked Bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia
S. Nilus
Chemical Engineering Programme, School of Engineering and Information Technology, Niversiti Malaysia Sabah, Locked Bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia
L.M. Chng
Chemical Engineering Programme, School of Engineering and Information Technology, Niversiti Malaysia Sabah, Locked Bag No. 2073, 88999 Kota Kinabalu, Sabah, Malaysia