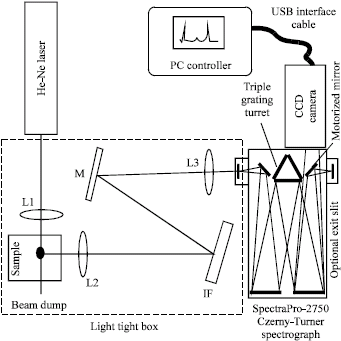
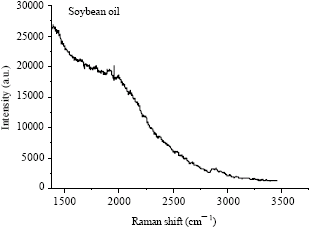
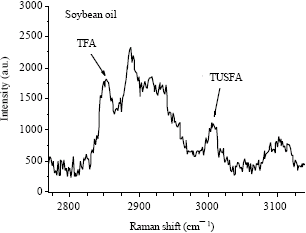
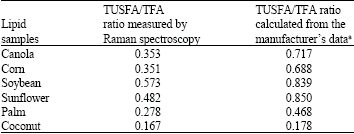
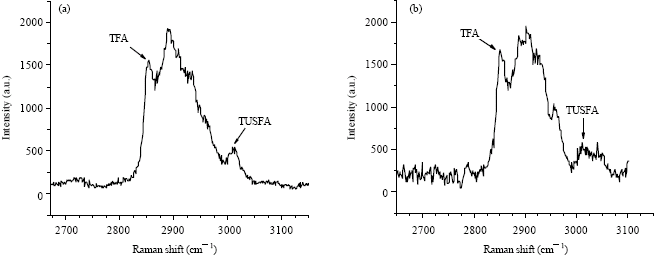
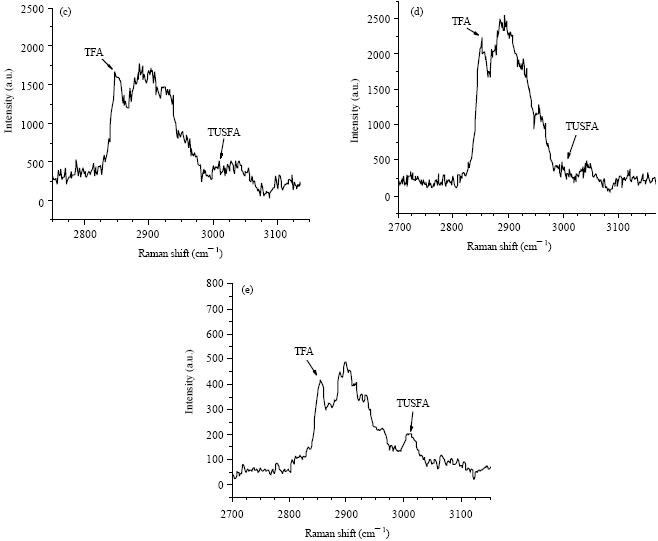
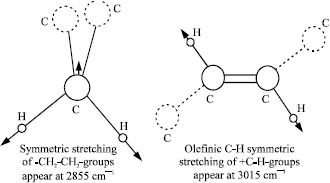
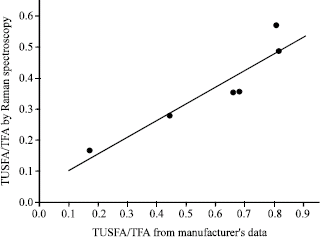
Research Article
Determination of Ratio of Unsaturated to Total Fatty Acids in Edible Oils by Laser Raman Spectroscopy
Industrial Physics Division, BCSIR Laboratories, Dhaka-1205, Bangladesh
K.M. Abedin
Department of Physics, University of Dhaka, Dhaka-1000, Bangladesh
M. Rafiqul Islam
Department of Physics, University of Dhaka, Dhaka-1000, Bangladesh
Aminul I. Talukder
Department of Physics, University of Dhaka, Dhaka-1000, Bangladesh
A.F.M.Y. Haider
Department of Physics, University of Dhaka, Dhaka-1000, Bangladesh