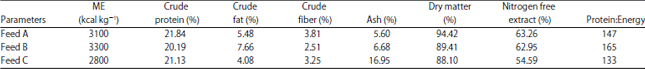
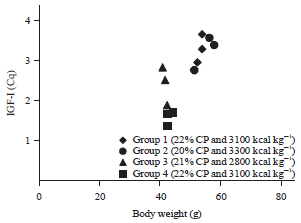
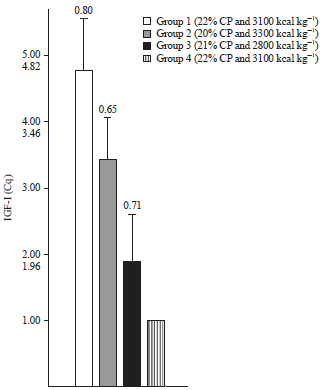Research Article
Effect of Varying Level of Crude Protein and Energy on Insulin-like Growth Factor-I Expression Level in Indonesian Hybrid Chicken
Laboratory of Genetics and Breeding, Faculty of Biology, Universitas Gadjah Mada, Jalan Teknika Selatan Sekip Utara, 55281 Yogyakarta, Indonesia
H.T. S.S.G. Saragih
Laboratory of Animal Structure and Development, Faculty of Biology, Universitas Gadjah Mada, Jalan Teknika Selatan Sekip Utara, 55281 Yogyakarta, Indonesia
B. S. Daryono
Laboratory of Genetics and Breeding, Faculty of Biology, Universitas Gadjah Mada, Jalan Teknika Selatan Sekip Utara, 55281 Yogyakarta, Indonesia