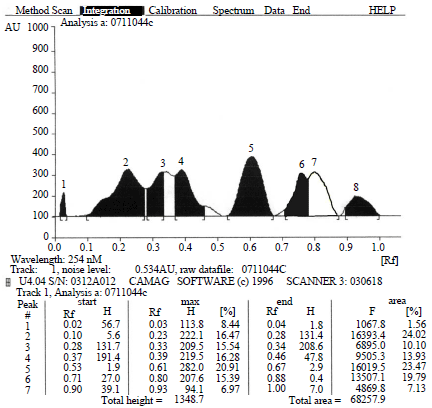
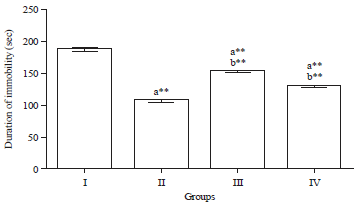
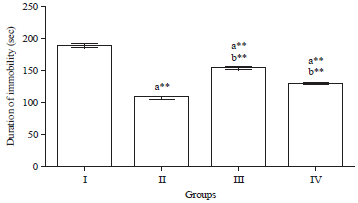
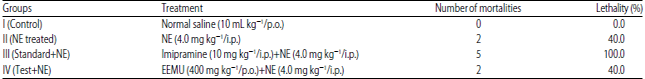Research Article
Anti-depressant Activity of Standardized Macrotyloma uniflorum Extract in Experimental Models of Depression in Rats
Department of Geriatric, The Second Affiliated Hospital of Zhengzhou University, Zhengzhou, 450000 Henan Province, China
Xiaoxiao Li
Department of Geriatric, The Second Affiliated Hospital of Zhengzhou University, Zhengzhou, 450000 Henan Province, China
Weiqin Yang
Departement of Pediatric, Zhengzhou Huiji District People`s Hospital, Zhengzhou, 450000 Henan Province, China
He Jia
Department of Geriatric, The Second Affiliated Hospital of Zhengzhou University, Zhengzhou, 450000 Henan Province, China
Chunling Liu
Department of Neurology, The Second Affiliated Hospital of Zhengzhou University, Zhengzhou, 450000 Henan Province, China
Yash Prashar
Rayat Institute of Pharmacy, 144533 Railmajra, India
LiveDNA: 91.15949
Souravh Bais
Rayat Institute of Pharmacy, 144533 Railmajra, India
LiveDNA: 91.5733