Research Article
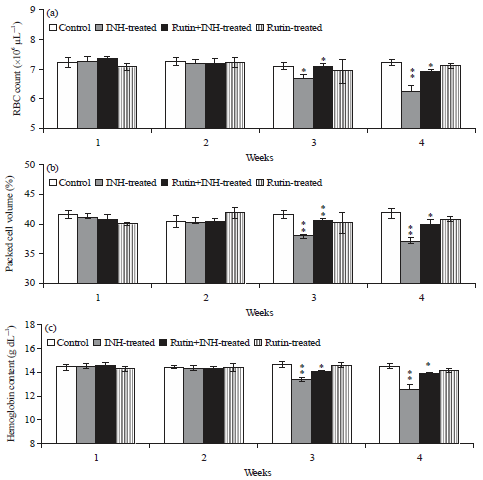
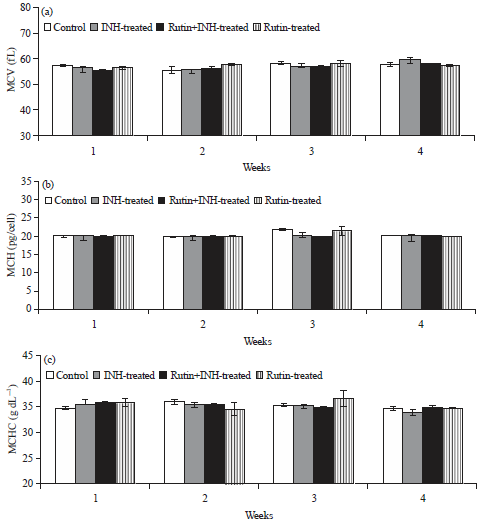
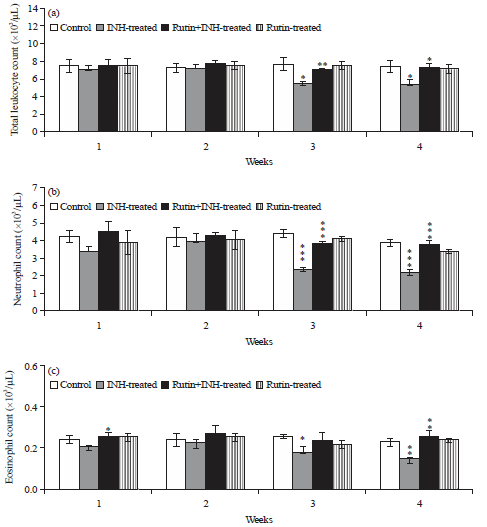
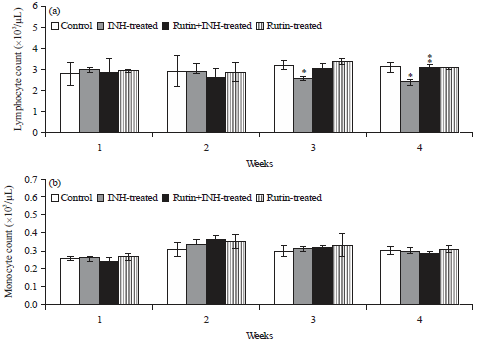
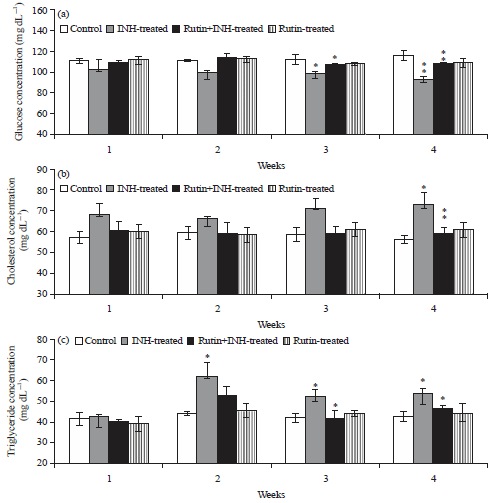
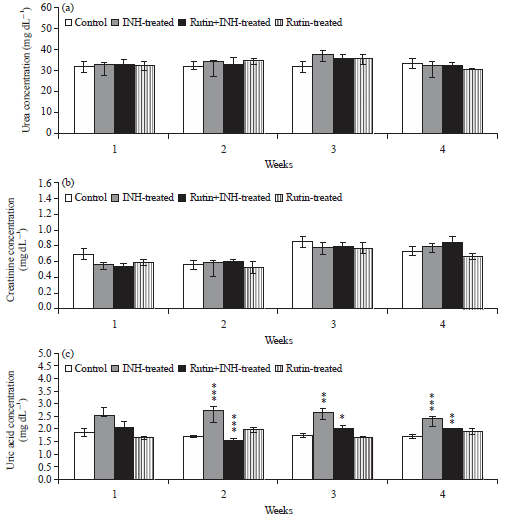
Ameliorative Effect of Rutin Against Isoniazid-induced Alterations in Certain Hematological and Biochemical Parameters of Albino Rats
Division of Physiology, Department of Zoology, Faculty of Science, Cairo University, Giza, Egypt
Salwa Thabet Mahmoud
National Organization for Drug Control and Research (NODCAR), Agouza, Giza, Egypt
Azza Ali Said
Division of Physiology, Department of Zoology, Faculty of Science, Fayoum University, Fayoum, Egypt
LiveDNA: 20.16161
Fatma Abdel-Azeem Youssef Sanad
National Organization for Drug Control and Research (NODCAR), Agouza, Giza, Egypt