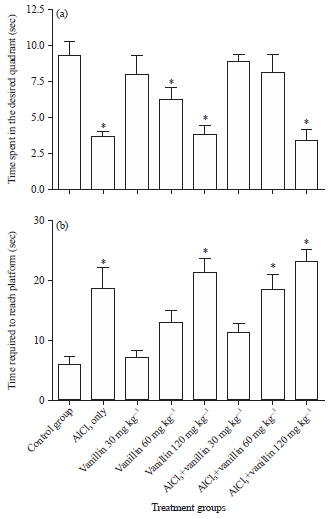
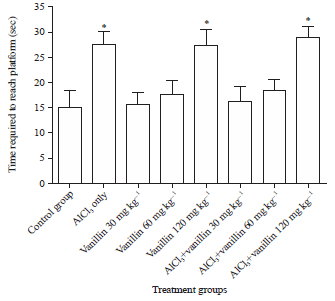
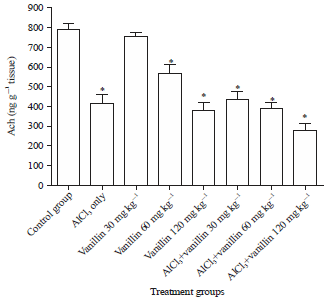
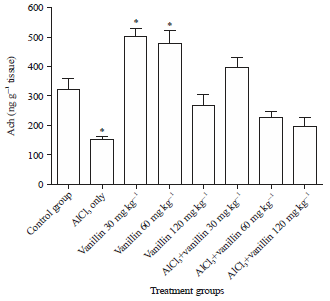
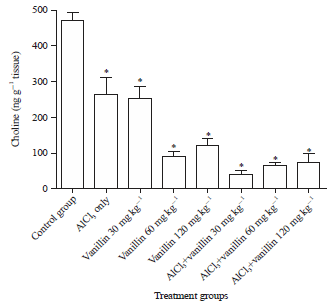
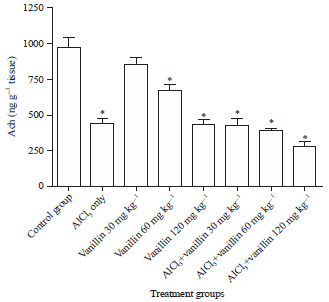
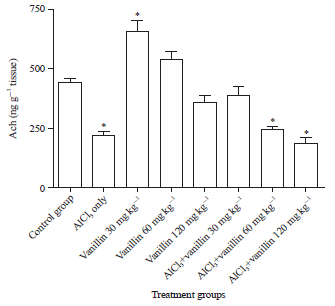
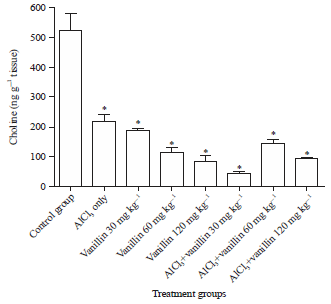
Research Article
Behavioral and Neurochemical Alterations Induced by Vanillin in a Mouse Model of Alzheimer’s Disease
College of Pharmacy, Al Ain University of Science and Technology, Al Ain-Abu Dhabi, United Arab Emirates
LiveDNA: 962.16138
Duaa Thalji
Faculty of Pharmacy, Al-Isra University, Amman, Jordan
Naji Abuirmeileh
Department of Nutritional Sciences, Jordan University of Science and Technology, Irbid, Jordan
Anas Bahnassi
Coram CVS Specialty Infusion Services, Coram Health Services, Denver, CO, United State of America
Izzedin Salahat
Department of Medicinal Chemistry, Faculty of Pharmacy, University of Tabuk, Tabuk, Kingdom of Saudi Arabia
Amjad Abuirmeileh
Faculty of Pharmacy, Al-Isra University, Amman, Jordan