Review Article
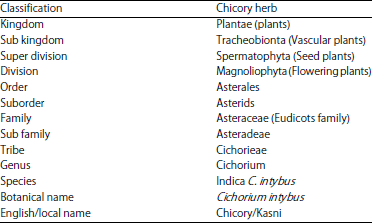
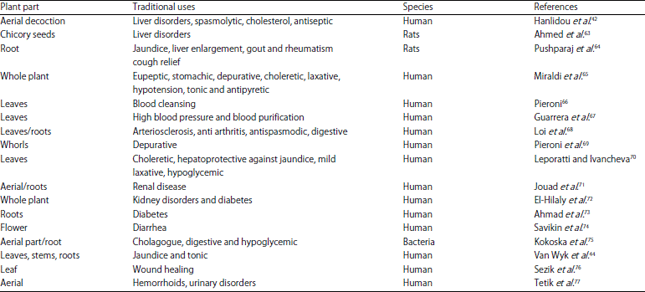
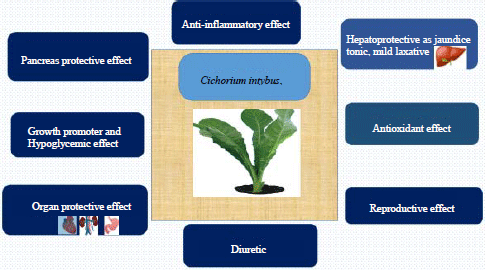
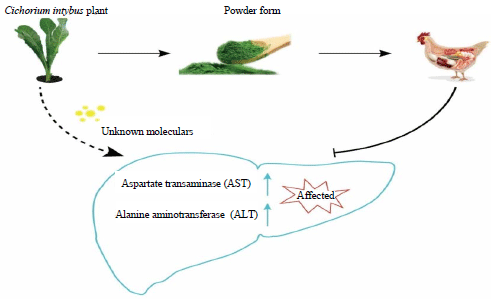
Chicory (Cichorium intybus) Herb: Chemical Composition, Pharmacology, Nutritional and Healthical Applications
College of Animal Sciences and Technology, Northwest A and F University, 712100 Yangling, China
LiveDNA: 92.10398
Mohamed E. Abd El-Hack
Department of Poultry, Faculty of Agriculture, Zagazig University, 44511 Zagazig, Egypt
LiveDNA: 20.7210
Mahmoud Alagawany
Department of Poultry, Faculty of Agriculture, Zagazig University, 44511 Zagazig, Egypt
LiveDNA: 20.7214
Muhammad A. Arain
College of Animal Sciences and Technology, Northwest A and F University, 712100 Yangling, China
LiveDNA: 92.16093
Muhammad Arif
Department of Animal Sciences, College of Agriculture, University of Sargodha, 40100, Pakistan
Muhammad A. Mirza
Institute of Animal Nutrition and Feed Technology, University of Agriculture, Faisalabad, Pakistan
Muhammad Naveed
Department of Clinical Pharmacy, School of Basic Medicine and Clinical Pharmacy, China Pharmaceutical University, Nanjing, Jiangsu, 211198, China
Sun Chao
College of Animal Sciences and Technology, Northwest A and F University, 712100 Yangling, China
Muhammad Sarwar
G.M. College for Science and Technology, 38090 Faisalabad, Pakistan
LiveDNA: 92.92
Maryam Sayab
ACE College for Women, Sargodha Road, Faisalabad, Pakistan
LiveDNA: 92.14416
Kuldeep Dhama
Division of Pathology, ICAR-Indian Veterinary Research Institute, Izatnagar, 243122 Uttar Pradesh, India
LiveDNA: 91.4710










fida hussain Reply
very informative