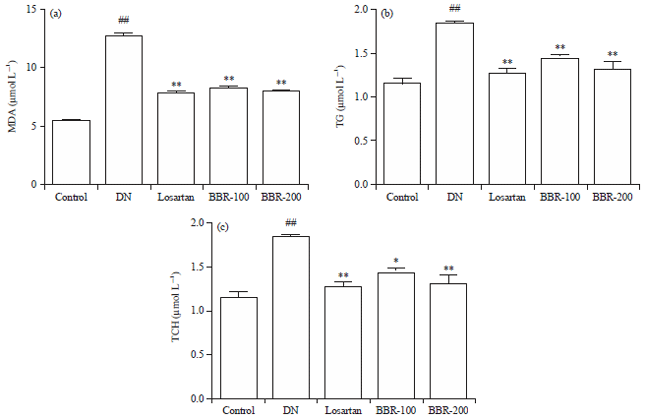
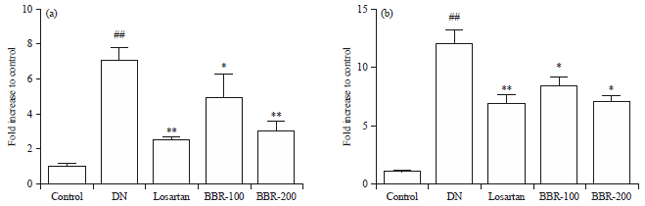
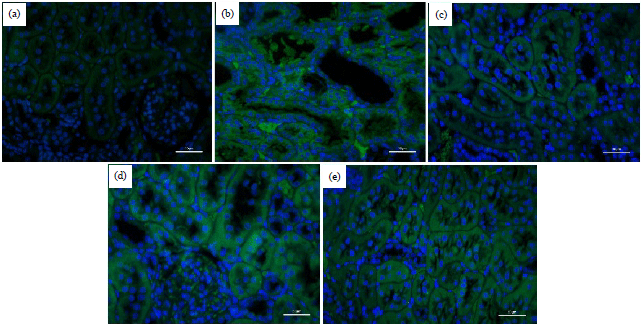
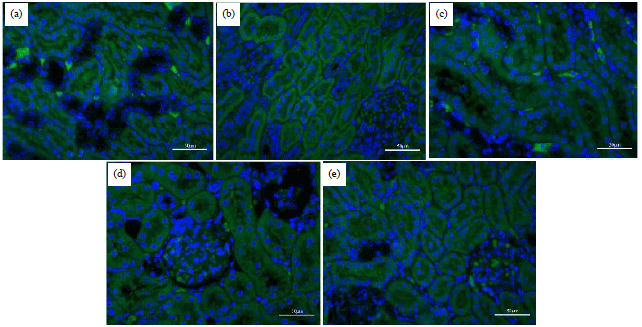
Research Article
Renoprotective Effect of Berberine on Streptozotocin-induced Diabetic Nephropathy Rats
Department of Pharmacy, The First Affiliated Hospital of Jinan University, 510630 Guangzhou, China
LiveDNA: 86.16523
Zhanxiong Xie
Department of Pharmacy, Shenzhen Longhua District Central Hospital, 518110 Shenzhen, China
Jianghong Liu
Department of Pharmacy, Shenzhen Longhua District Central Hospital, 518110 Shenzhen, China
Qunhua Wu
Department of Pharmacy, Shenzhen Longhua District Central Hospital, 518110 Shenzhen, China
Xiaokang Wang
Department of Pharmacy, Shenzhen Longhua District Central Hospital, 518110 Shenzhen, China