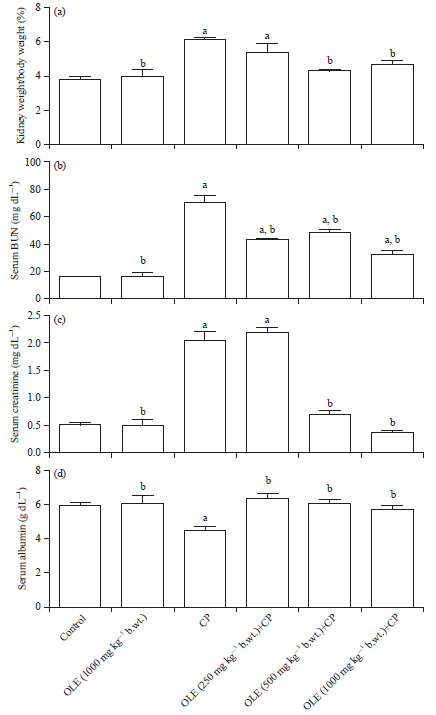
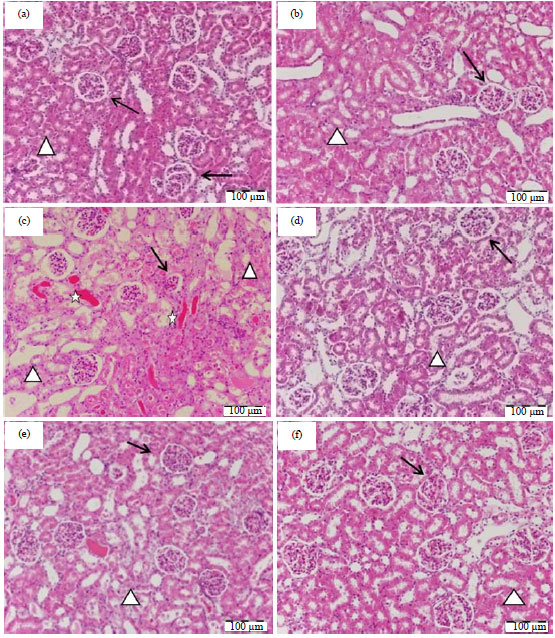
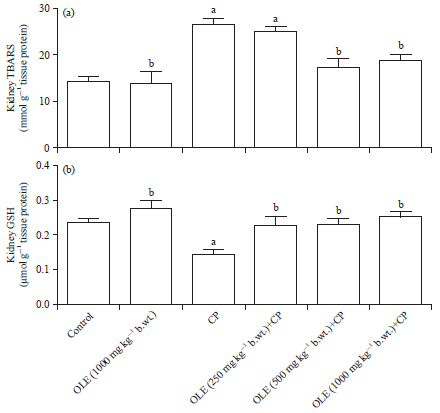
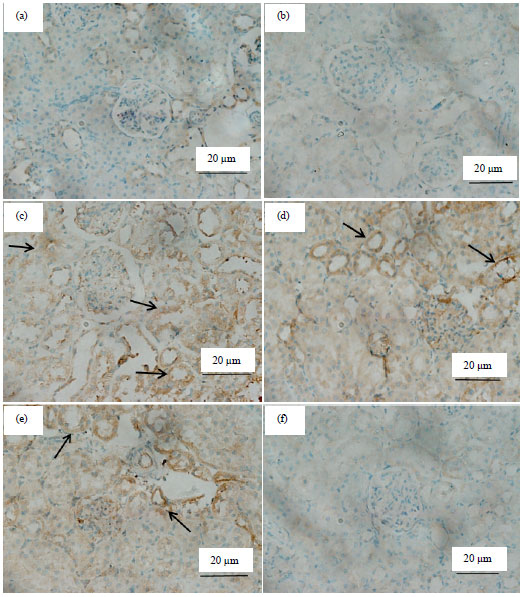
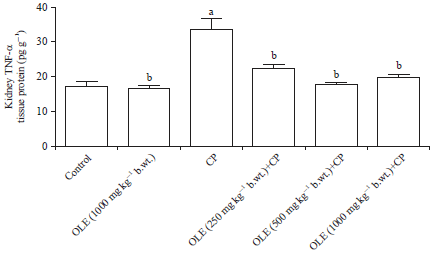
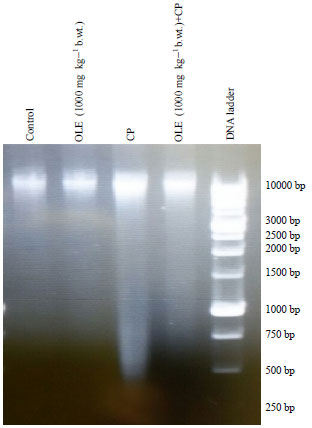
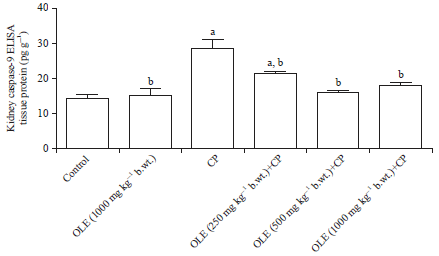
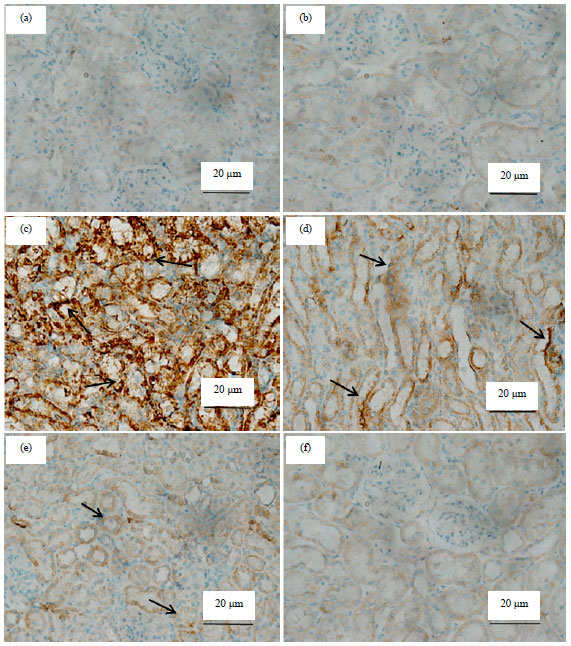
Research Article
Anti-apoptotic and Anti-inflammatory Effects of Olive Leaf Extract Against Cisplatin-induced Nephrotoxicity in Male Rats
Department of Pharmacology and Toxicology, College of Pharmacy, King Saud University, P.O. Box 22452, 11459 Riyadh, Saudi Arabia
Dalia Fouad
Department of Zoology and Entomology, Faculty of Science, Helwan University, EinHelwan, Cairo, Egypt
LiveDNA: 20.4569