Research Article
Diosmin Protects Against Cyclophosphamide-induced Liver Injury Through Attenuation of Oxidative Stress, Inflammation and Apoptosis
Department of Biology, Faculty of Science, Aljouf University, Sakaka, Saudi Arabia
Drug-induced liver injury occurs after exposure to any naturally occurring or manufactured chemical compound. It can lead to acute liver failure or fulminant hepatic failure that causes hepatic encephalopathy and coagulopathy within 26 weeks of the onset of illness1. Cyclophosphamide (CP) is an alkylating agent nitrogen mustard commonly used in cancer chemotherapy. It has an immunosuppressive activity and is used for renal and bone marrow transplantations and in the treatment of autoimmune diseases2. However has a wide spectrum of clinical uses, CP causes multiple adverse effects including hepatotoxicity in humans as well as experimental animals3-6.
The precise mechanism by which CP induces hepatotoxicity is poorly understood. Recent studies have suggested that Reactive Oxygen Species (ROS) like hydrogen peroxide (H2O2), superoxide anion and hydroxyl radical are implicated in CP-induced liver injury4,6-8. During metabolism, CP is converted to phosphoramide mustard and acrolein. Acrolein is a highly reactive metabolite that may be responsible for CP-induced liver injury9. Inflammation has also been reported to play a crucial role in CP-induced hepatotoxicity4,6. In this context, pro-inflammatory cytokines like tumor necrosis factor alpha (TNF-α) and interleukin 1 beta (IL-1β) have been demonstrated in a rat model of CP-induced hepatotoxicity6. The increased pro-inflammatory cytokines could be explained by CP-induced activation of nuclear factor-kappa B (NF-κB)4. The TNF-α and IL-1β contribute to the pathological complications observed in several diseases and activates the pro-apoptotic caspase cascade10 leading to apoptosis of hepatocytes. Therefore, a compound with antioxidant and anti-inflammatory activities could offer a protection against CP-induced hepatocyte damage.
Diosmin or diosmetin-7-O-rutinoside is a naturally occurring flavone glycoside found abundantly in the pericarp of various citrus11. Diosmin has been reported to exert multiple beneficial effects against myocardial infarction12, diabetes mellitus13, hepatic and renal injuries14,15 and hepatocarcinogenesis16. Moreover, diosmin has been found to improve lymphatic drainage, increase the venous tone and reduce the capillary hyperpermeability, thereby, leading to declined release of the inflammatory mediators17. Yet, its impact on CP-induced liver oxidative stress, inflammation and apoptosis has not been previously explored. Thus, the present study aimed to investigate the potential protective actions of diosmin in CP-induced hepatotoxicity in rats.
Chemicals: Diosmin, reduced glutathione (GSH), pyrogallol, 5,5-dithiobis-(2-nitrobenzoic acid) (DTNB), thiobarbituric acid (TBA), trichloroacetic acid (TCA) and 1,1,3,3 tetramethoxypropane were purchased from Sigma (USA). Cyclophosphamide (CP, endoxan) was supplied by Baxter oncology (Düsseldorf, Germany). All other chemicals and reagents were of analytical grade and obtained from standard commercial supplies.
Animals and treatments: Twenty four male albino rats (Rattus norvegicus), weighing about 140-160 g, obtained from the animal center at the Faculty of Pharmacy, Aljouf University (Saudi Arabia) were used as in the present investigation. The animals were housed in standard polypropylene cages (4 rats/cage) and maintained under controlled room temperature (22±2°C) and humidity (55±5%) with 12 h light and 12 h dark cycle and were fed a standard diet of known composition and water ad libitum. The animals used were maintained in accordance with the recommendations of the Animal Ethics Committee of Aljouf University (Saudi Arabia).
The animals were divided into four groups, each comprising six rats (N = 6) designated as follows:
| • | Group I (Control): Rats received the vehicle 0.5% carboxymethylcellulose (CMC) orally for 15 days and a single intraperitoneal dose of normal saline at day 16 |
| • | Group II (CP): Rats received 0.5% CMC orally for 15 days and a single intraperitoneal dose of CP (150 mg kg–1) at day6 16 |
| • | Group III (CP+50 mg diosmin): Rats received 50 mg kg–1 diosmin18 dissolved in 0.5% CMC orally for 15 days and a single intraperitoneal dose of CP (150 mg kg–1) at day 16 |
| • | Group IV (CP+100 mg diosmin): Rats received 100 mg kg–1 diosmin18 dissolved in 0.5% CMC orally for 15 days and a single intraperitoneal dose of CP (150 mg kg–1) at day 16 |
At the end of the experiment (day 18), blood samples were collected, allowed to coagulate at room temperature and then centrifuged at 3000 rpm for 10 min to separate serum. Collected sera were stored at -20°C until used. Immediately after sacrifice, livers were quickly excised and washed in cold saline. A sample from each liver was homogenized (10% w/v) in cold Phosphate Buffered Saline (PBS). The homogenate was centrifuged at 3000 rpm for 15 min at 4°C and the clear supernatant was stored at -20°C. Other liver samples were either fixed in 10% buffered formalin for histological processing or kept frozen at -80°C for RNA isolation.
Determination of liver marker enzymes: Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were assayed in serum of rats according to the method of Schumann and Klauke19 using reagent kit purchased from Spinreact (Spain). Serum alkaline phosphatase (ALP) was measured according to the method of Wenger et al.20 using Spinreact (Spain) reagent kit.
Determination of TNF-α and IL-1β): Liver TNF-α and IL-1β were determined using specific ELISA kits purchased from R and D systems (USA) following the manufacturer’s instructions.
Determination of oxidative stress and antioxidant status: Lipid peroxidation was assayed by measuring malondialdehyde (MDA) following the method of Preuss et al.21. Nitric oxide level was estimated as nitrite according to the method of Montgomery and Dymock22 using reagent kit purchased from biodiagnostics (Egypt). The GSH content was determined following the method of Beutler et al.23. Activity of the antioxidant enzymes superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx) were determined according to the methods of Marklund and Marklund24, Cohen et al.25 and Matkovics et al.26, respectively.
RNA isolation and quantitative reverse transcription-polymerase chain reaction (qRT-PCR): Gene expression analysis of TNF-α, IL-1β, BAX and Bcl2 was determined in liver of rats according to the method of Mahmoud 4. Total RNA was isolated from frozen liver samples using TRIzol reagent (Invitrogen), purified and quantified and at 260 nm. The RNA samples with A260/A280 ratios ≥1.7 were selected. Reverse transcription was performed with 2 μg RNA using high-capacity cDNA reverse transcription kit with RNase inhibitor (Applied biosystems, USA). The cDNA was amplified using SYBR green master mix (Applied Biosystems, USA) in a total volume of 20 μL using the primer set listed in Table 1. The PCR reaction included initial denaturation at 95°C for 10 min, 35 cycles of denaturation at 95°C for 30 sec, annealing at Tm-5°C for 30 sec and extension at 72°C for 60 sec. Fluorescent values were converted into threshold cycle (CT) and data were analyzed following the 2–ΔΔCt method27 and normalized to β-actin.
| Table 1: | Primers used for qRT-PCT |
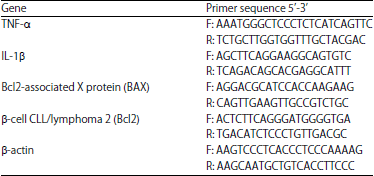 | |
Histopathological study: Liver samples were fixed in 10% neutral buffered formalin for 24 h. The fixed samples were processed and 4 μM thick paraffin sections were prepared. Slides were stained with hematoxylin and eosin (H and E) and then examined by light microscopy.
Immunohistochemical staining of BAX and Bcl-2: Immunolocalization staining of BAX and Bcl2 was performed according to Pedrycz and Czerny28. In brief, 4 μM liver sections were incubated with mouse anti-BAX or anti-Bcl2 (Santa Cruz Biotechnology, USA) diluted 1:200 in tris-buffered saline with 1% bovine serum albumin for 60 min. Sections were washed and incubated with a biotinylated anti-mouse secondary antibody (Dako Cytomation, USA) for 30 min followed by horseradish peroxidase conjugated with streptavidin for further 15 min incubation. After washing the sections, 3-amino-9-ethylcarbasole (Dako Cytomation, USA) was added for 15 min. The sections were counterstained with hematoxylin for 1 min, mounted using the aquatex fluid (Merk KGaA, Germany) and then examined by light microscopy.
Statistical analysis: Statistical analysis was performed using GraphPad prism 5 software (GraphPad software, San Diego, CA, USA). Results were expressed as Mean±Standard Error (SEM) and all statistical comparisons were made by means of the one-way ANOVA test followed by Tukey’s test post hoc analysis. A p-value <0.05 was considered significant.
Disosmin prevents CP-induced liver injury: Figure 1 show the effect of diosmin pre-treatment on serum ALT, AST and ALP activities, the main liver activity biomarkers, in CP-intoxicated rats. Administration of CP produced significant (p<0.001) increases in serum ALT, AST and ALP activities when compared with the control rats. Pre-treatment with 50 mg kg–1 diosmin significantly (p<0.001) decreased serum ALT, AST and ALP in CP-intoxicated rats.
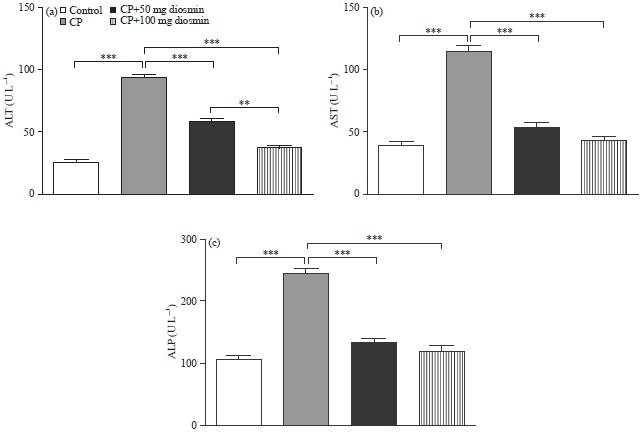 | |
| Fig. 1(a-c): | Effect of diosmin on serum (a) ALT, (b) AST and (c) ALP activities in CP-intoxicated rats. Data are Mean±SEM (N = 6), **p<0.01 and ***p<0.001 |
Similarly, pre-treatment of the CP-intoxicated rats with 100 mg kg–1 diosmin significantly (p<0.001) ameliorated liver marker enzymes when compared with CP-administered rats. Of note, the higher diosmin dose produced a significant (p<0.01) decrease in serum ALT activity as compared to the lower one.
Histopathological examination of H and E-stained liver sections of the normal control rats showed normal histological structure of the hepatocytes (Fig. 2a). On the other hand, sections in the liver of rats treated with CP showed focal areas with massive degeneration, cytoplasmic vacuolations, necrosis and inflammatory cellular infiltration (Fig. 2b). Liver of CP-induced rats pre-treated either with 50 mg (Fig. 2c) or with 100 mg kg–1 diosmin (Fig. 2d) revealed marked improvement of liver histological structure and apparently normal liver tissue.
Disosmin attenuates CP-induced inflammation in liver of rats: The CP administration produced a significant (p<0.001) increase in liver levels of the pro-inflammatory cytokine TNF-α, as represented in Fig. 3a. Oral supplementation with either dose of diosmin 15 days before CP administration significantly (p<0.01) decreased levels of TNF-α in liver of rats. In comparison with the lower dose, diosmin at 100 mg kg–1 dose significantly decreased liver TNF-α levels in CP-induced rats.
Liver TNF-α mRNA expression showed a similar pattern. Administration of CP significantly (p<0.001) up-regulated TNF-α mRNA expression when compared with the control rats, as depicted in Fig. 3b. Pre-treatment with 50 mg or 100 mg kg–1 diosmin produced a significant (p<0.01) down-regulation of TNF-α mRNA expression in liver of CP-induced rats.
The IL-1β exhibited a significant (p<0.001) elevation in liver of CP-induced rats when compared with the corresponding control rats (Fig. 3c). Pre-treatment of the CP-intoxicated rats with 50 mg kg–1 diosmin significantly (p<0.001) decreased liver IL-1β levels. In addition, the 100 mg kg–1 diosmin dose produced a marked (p<0.001) decrease in liver IL-1β levels in CP-induced rats.
Liver IL-1β mRNA expression showed a significant (p<0.001) increase following CP administration. Pre-treatment with 50 mg kg–1 diosmin significantly (p<0.01) decreased liver IL-1β mRNA expression in CP-induced rats. The higher diosmin dose (100 mg kg–1) produced a more potent effect and markedly (p<0.001) improved liver IL-1β mRNA expression when compared with CP control rats, as represented in Fig. 3d.
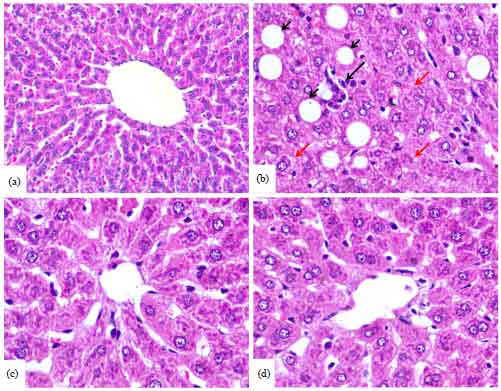 | |
| Fig. 2(a-d): | Photomicrographs of H and E-stained liver sections of (a) Control rats, (b) CP-intoxicated rats showing focal areas with massive degeneration, necrosis and inflammatory cellular infiltration, (c) CP-intoxicated rats pre-treated with 50 mg kg–1 diosmin showing marked improvement in liver histological structure and (d) CP-intoxicated rats pre-treated with 100 mg kg–1 diosmin showing apparently normal liver tissue |
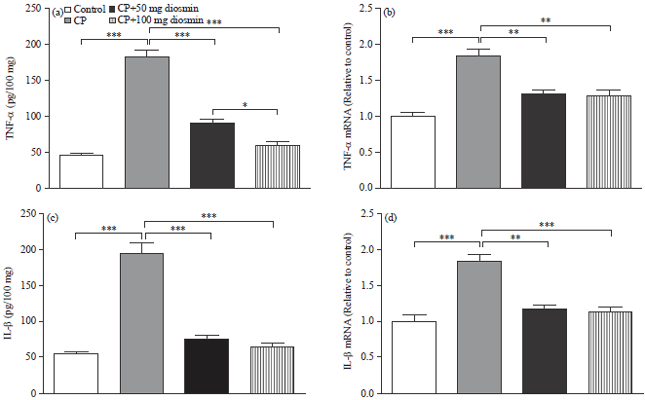 | |
| Fig. 3(a-d): | Effect of diosmin on liver, (a) TNF-α, (b) TNF-α mRNA expression, (c) IL-1β and (d) IL-1β mRNA expression in CP-intoxicated rats. Data are Mean±SEM (N = 6), *p<0.05, **p <0.01 and ***p<0.001 |
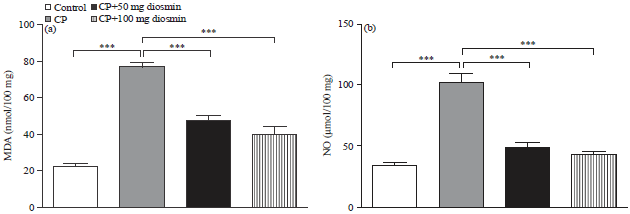 | |
| Fig. 4(a-b): | Effect of diosmin on liver (a) Lipid peroxidation and (b) Nitric oxide levels in CP-intoxicated rats. Data are Mean±SEM (N = 6), ***p<0.001 |
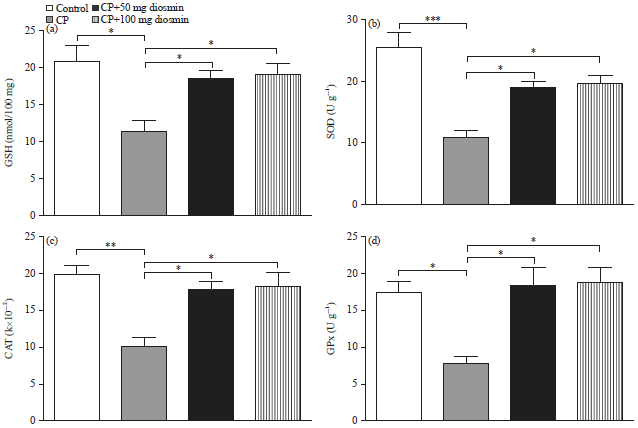 | |
| Fig. 5(a-d): | Effect of diosmin on liver, (a) GSH, (b) Superoxide dismutase, (c) Catalase and (d) Glutathione peroxidase in CP-intoxicated rats. Data are Mean±SEM (N = 6), *p<0.05, **p<0.01 and ***p<0.001 |
Disosmin mitigates CP-induced oxidative stress in liver of rats: Figure 4a, b show the effects of diosmin on lipid peroxidation and nitric oxide levels in liver of CP-induced rats. Rats received CP showed a significant (p<0.001) increase in liver lipid peroxidation, indicated by the elevated MDA levels, when compared with the control group. Both doses of diosmin significantly (p<0.001) attenuated lipid peroxidation in liver of CP-intoxicated rats.
Similarly, nitric oxide exhibited a significant (p<0.001) elevation in liver of CP-induced rats when compared with the control rats. Pre-treatment with either dose of diosmin potentially (p<0.001) ameliorated nitric oxide levels in the liver of CP-administered rats.
To further explore the protective effect of diosmin against CP-induced oxidative stress, liver GSH and antioxidant enzymatic defenses were determined. The CP-administered rats showed a significant (p<0.05) decrease in liver GSH content, as represented in Fig. 5a. Treatment with diosmin at 50 or 100 mg kg–1 significantly (p<0.05) restored hepatic GSH content in CP-administered rats. More or less similar, the CP-induced rats exhibited significant declined activity of liver SOD (p<0.001, Fig. 5b), CAT (p<0.01, Fig. 5c) and GPx (p<0.05, Fig. 5d) when compared with the corresponding control rats. Pre-treatment of the CP-induced rats with 50 or 100 mg kg–1 diosmin significantly (p<0.05) alleviated activity of SOD, CAT and GPx.
Disosmin mitigates CP-induced apoptosis in liver of rats: To investigate the anti-apoptotic effects of diosmin in liver of the CP-intoxicated rats, gene and protein expression levels of the pro-apoptotic (BAX) and anti-apoptotic (Bcl2) mediators were determined using qRT-PCR and immunohistochemistry, respectively.
The CP-administered rats exhibited a significant (p<0.001) increase in mRNA expression of liver BAX, as depicted in Fig. 6a. On the other hand, CP-induced rats pre-treated with 50 as well as 100 mg kg–1 diosmin showed a significant (p<0.001) down-regulation in liver BAX mRNA expression levels.
Immunohistochemical staining of BAX revealed marked up-regulation in liver of the CP-induced rats (Fig. 7b) when compared with control rats (Fig. 7a). Pre-treatment either with 50 (Fig. 7c) or with 100 mg kg–1 diosmin (Fig. 7d) markedly decreased BAX expression in liver of CP-induced rats.
Conversely, Bcl2 mRNA expression exhibited marked (p<0.01) down-regulation in liver of CP-administered rats when compared with the corresponding controls (Fig. 6b). Diosmin pre-treatment produced significant (p<0.05) up-regulation in Bcl2 mRNA in liver of CP-administered rats. In accordance, immunohistochemical staining of Bcl2 revealed decreased expression in liver of CP-administered rats (Fig. 8b) when compared with the control group (Fig. 8a). Diosmin pre-treatment, 50 (Fig. 8c) and 100 mg kg–1 (Fig. 8d), produced a noticeable alleviation in liver Bcl2 protein levels in CP-induced rats.
Cyclophosphamide is a cytotoxic chemotherapeutic drug that acts against various cancers. However, most of its therapeutic uses are often restricted because of its adverse side effects and toxicity. Therefore, the present study aimed at investigating the protective effects of diosmin against CP-induced hepatotoxicity focusing on oxidative stress, inflammation and apoptosis.
The CP administration induced liver injury as showed by the elevated liver marker enzymes, ALT, AST and ALP, in serum of rats. Hepatocytes are rich in these enzymes and thus used commonly to check the liver injury29. The elevated ALT, AST and ALP levels could be a direct result of CP-induced hepatocyte damage. In accordance, we have reported elevated transaminases in serum of CP-induced hepatotoxicity in rats5. More recently, the studies of Mahmoud4 and Mahmoud and Al Dera6 revealed similar findings. The CP-induced liver injury was further confirmed by the recorded histological alterations, including necrosis, inflammatory cell infiltrations, fatty degenerations and other manifestations as previously reported6. Interestingly, diosmin pre-treatment at either dose significantly reduced serum ALT, AST and ALP levels and prevent CP-induced histopathological alterations.
Accordingly, Tahir et al.15 demonstrated that diosmin decreases serum transaminases in alcohol-induced hepatic injury. In addition, Tanrikulu et al.30 reported the ameliorative effect of diosmin on serum transminases in ischaemia/reperfusion induced liver injury. These findings suggest a cell membrane stabilizing property of diosmin and thus hepatoprotective efficacy against CP-induced liver damage.
Oxidative stress has been reported to play an important role in mediating CP-induced liver injury as evidenced from previous studies5,31.
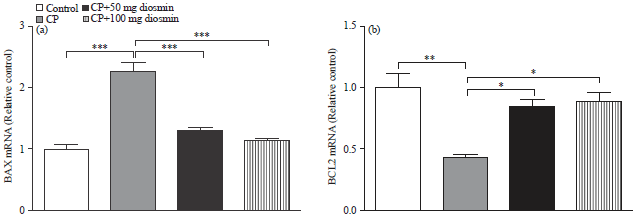 | |
| Fig. 6(a-b): | Effect of diosmin on liver (a) BAX and (b) Bcl2 mRNA expression in CP-intoxicated rats. Data are Mean±SEM (N = 6), *p<0.05, **p<0.01 and ***p<0.001 |
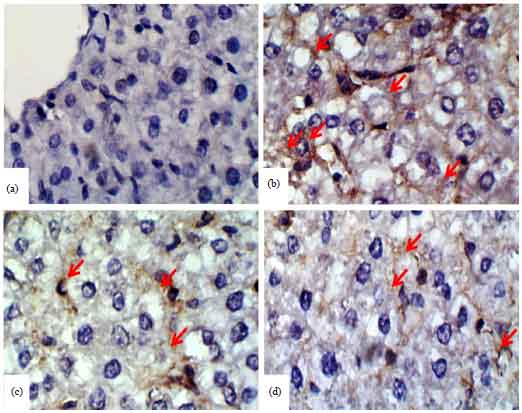 | |
| Fig. 7(a-d): | Photomicrographs of BAX immunostained liver sections (a) Control rats, (b) CP-intoxicated rats, (c) CP-intoxicated rats pre-treated with 50 mg kg–1 diosmin and (d) CP-intoxicated rats pre-treated with 100 mg kg–1 diosmin |
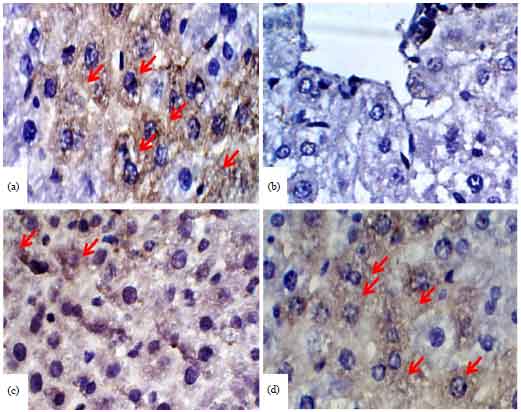 | |
| Fig. 8(a-d): | Photomicrographs of Bcl2 immunostained liver sections, (a) Control rats, (b) CP-intoxicated rats, (c) CP-intoxicated rats pre-treated with 50 mg kg–1 diosmin and (d) CP-intoxicated rats pre-treated with 100 mg kg–1 diosmin |
In the present investigation, CP administration produced a significant increase in liver lipid peroxidation and nitric oxide levels. Accordingly, the studies of Mahmoud 4, Germoush and Mahmoud 5 and Mahmoud and Al Dera6 have reported elevated lipid peroxidation and nitric oxide levels in liver of CP-administered rats. The metabolism of CP produces ROS and free radicals due to acrolein formation32. It was reported that these radicals are involved in the CP-induced oxidative stress33. Lipid peroxidation has been linked with decreased membrane fluidity, increased nonspecific permeability to ions, inactive membrane-bound receptors and enzymes and impaired membrane functioning34. In addition, the produced nitric oxide could react with superoxide producing the potent oxidant peroxynitrite35 and stimulate the production of inflammatory cytokines through the activation of NF-κB in Kupffer cells36. The elevated nitric oxide levels have been explained by the increased expression of inducible nitric oxide synthase (iNOS) in CP-administered rats6. Pre-treatment with diosmin markedly protected rats against CP-induced lipid peroxidation and elevated nitric oxide, indicating antioxidant and radical scavenging activities.
The GSH on the other hand, showed a marked decrease in CP-administered rats, confirming the oxidative stress status. The GSH is an important antioxidant that plays a fundamental role in cellular defense against ROS through acting as cofactor for antioxidant enzymes taking part in cellular redox reactions37. In addition, it reacts with and neutralizes toxic endogenous and exogenous metabolites and maintains cell integrity34,38. Moreover, activities of the antioxidant defense enzymes, SOD, CAT and GPx were significantly reduced in liver of CP-induced rats. These findings were in accordance with the previous study5 and other investigations4,6. Interestingly, pre-treatment of the CP-induced rats with diosmin significantly prevented CP-induced reduction in GSH and antioxidant enzymes. Diosmin has been previously reported to possess potent antioxidant and free radical scavenging activity. These findings are in agreement with earlier studies15 and they highlight the premise that the antioxidant efficacy of diosmin is implicated in the alleviation of CP-induced liver injury. The antioxidant action of diosmin has been ascribed to scavenging superoxide anions and hydroxyl radicals12. Therefore, by counteracting oxidative stress and rejuvenation of antioxidant defenses, diosmin was able to protect against CP-induced liver injury.
Disturbed intracellular hepatic oxidant/antioxidant status due to the administration of CP can activate the inflammatory pathway. Other investigators have recently reported that CP-induced oxidative stress activates NF-κB, leading to increased production of TNF-α and IL-1β5,6. In the present study, CP-administration significantly increased TNF-α and IL-1β gene expression and release in the liver of rats. Pre-treatment of rats with both doses of diosmin potentially protected against CP-induced inflammation through down-regulation of TNF-α and IL-1β gene expression and attenuation of their production. Accordingly, Tahir et al.15 reported that diosmin protects against ethanol-induced hepatic injury via alleviation of inflammation and down-regulation of TNF-α and NF-κB activation.
The CP-administration induced apoptosis of hepatocytes as indicated by down-regulation of gene as well as protein expression of the antiapoptotic protein Bcl2 and the up-regulated pro-apoptotic marker BAX. The observed apoptosis might be explained by the CP-induced production of pro-inflammatory cytokines. Through binding to their receptors, TNF-α and IL-1β can activate the pro-apoptotic caspase cascade10, resulting in apoptosis. Hepatocytes express both TNF receptor 1 (TNFR1) and TNF receptor 2 (TNFR2)39. On binding TNF-α, TNFR1 recruits the adaptor protein tumor necrosis factor receptor-associated death domain (TRADD)40, leading to recruitment of tumor necrosis factor receptor associated protein (TRAF-2) and receptor interacting protein 1 (RIP1). These events result in activation of NF-κB. The TRADD then dissociates from the ligated receptor, recruits FADD and procaspase 8 to initiate apoptotic signaling41,42. Therefore, diosmin can protect the liver against CP-induced apoptosis by diminishing the pro-inflammatory cytokine production. The anti-apoptotic effects of diosmin were confirmed by its modulatory effect on BAX and Bcl2 both gene and protein expression. The Bcl2 plays a central role in protecting cells from apoptosis43. It functions to inhibit the apoptosis during cell proliferation44. Takahashi et al.45 reported that over-expression of Bcl2 produced by the transfection of Bcl2 gene into the vascular smooth muscle cell inhibited their apoptosis. Thus, the increased expression of Bcl2 and decreased BAX and pro-inflammatory cytokines by diosmin reflected its anti-apoptotic activity.
The present study reveals several mechanisms by which CP lead to hepatotoxicity. Among them, generation of ROS, imbalance in redox state, lipid peroxidation and induction of inflammation and apoptosis. All these mechanisms lead to cell death following CP-administration. Diosmin alleviated CP-induced hepatotoxicity through attenuation of oxidative stress and inflammation and repression of apoptosis. These findings represent a novel protective mechanism of diosmin against CP-induced hepatotoxicity.
The author wishes to thank Aljouf University for providing facilities and support.