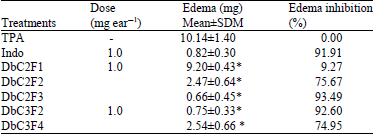
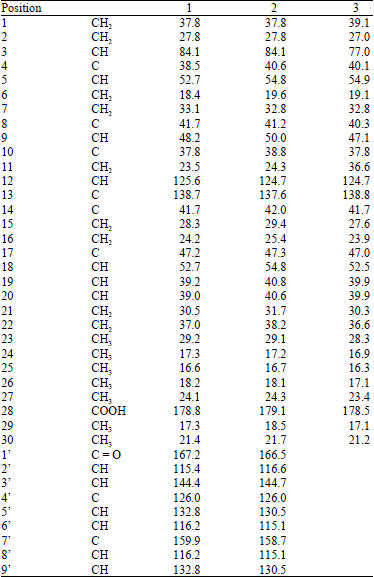
Research Article
Anti-inflammatory Extracts and Coumaroyl Ursolic Acid Derivatives from Distictis buccinatoria
Biomedical Research Center South, Mexican Social Security Institute, Argentina No. 1, Col. Centro, Xochitepec, Morelos, 62790, Mexico
Maria Gabriela Rojas-Bibriesca
Biomedical Research Center South, Mexican Social Security Institute, Argentina No. 1, Col. Centro, Xochitepec, Morelos, 62790, Mexico
Zuleica Nunez
Faculty of Medicine, University of the State of Morelos, Iztaccihuatl Street esq. Le�eros s/n, Col. Los Volcanes Cuernavaca, Mor. CP 62350, Mexico
Blanca Eda Dominguez-Mendoza
NMR Laboratory, Chemical Research Center, University of the State of Morelos, Av. Universidad 1001 Colonia Chamilpa CP 62210, Cuernavaca Morelos, Mexico
Margarita Aviles-Folres
Morelos INAH Center, Matamoros 14 Acapantzingo Cuernavaca Morelos, CP 62440, Mexico
Macrina Fuentes-Mata
Morelos INAH Center, Matamoros 14 Acapantzingo Cuernavaca Morelos, CP 62440, Mexico
Jaime Tortoriello
Biomedical Research Center South, Mexican Social Security Institute, Argentina No. 1, Col. Centro, Xochitepec, Morelos, 62790, Mexico
Alejandro Zamilpa
Biomedical Research Center South, Mexican Social Security Institute, Argentina No. 1, Col. Centro, Xochitepec, Morelos, 62790, Mexico