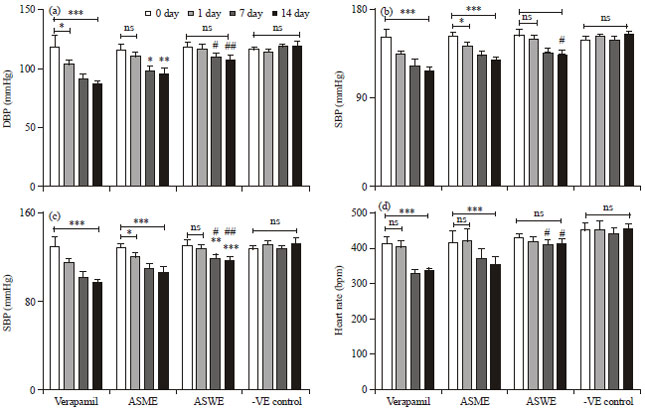
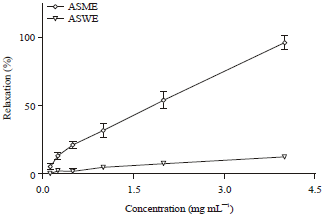
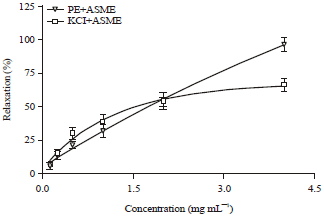
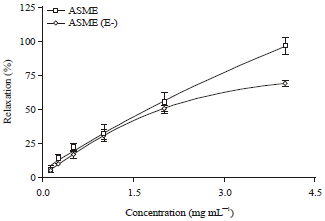
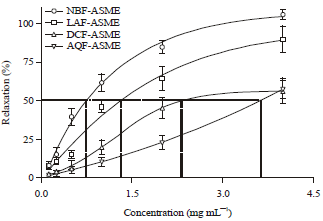
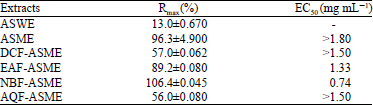
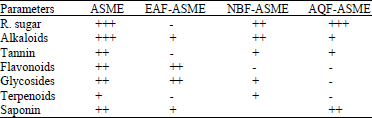
Research Article
Antihypertensive and Vasorelaxant Effect of Alstonia scholaris Stem Bark Extracts and Fractions
School of Pharmaceutical Sciences, University Sains Malaysia, 11800, Pulau Penang, Malaysia
Mohammad Zaini Asmawi
School of Pharmaceutical Sciences, University Sains Malaysia, 11800, Pulau Penang, Malaysia
Usman Salisu Nasiba
School of Pharmaceutical Sciences, University Sains Malaysia, 11800, Pulau Penang, Malaysia
Roziahanim Mahmud
School of Pharmaceutical Sciences, University Sains Malaysia, 11800, Pulau Penang, Malaysia
Kabiru Abubakar
Department of Pharmacology and Toxicology, Faculty of Pharmaceutical Sciences, Usmanu Danfodiyo University, Sokoto, Nigeria