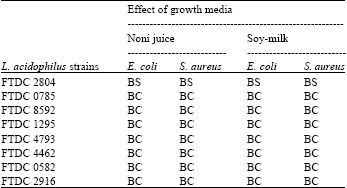
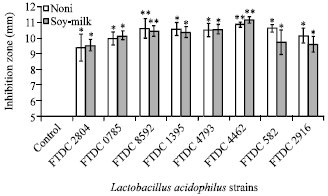
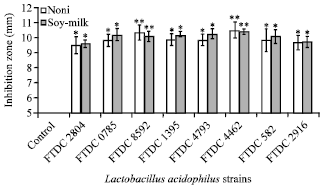
Research Article
Characteristics and Antibacterial Activity of Metabolites from Lactobacillus acidophilus Strains Produced from Novel Culture Media
School of Pharmaceutical Sciences, Universiti Sains Malaysia
Min-Tze Liong
School of Industrial Technology, Universiti Sains Malaysia
K.K Peh
School of Pharmaceutical Sciences, Universiti Sains Malaysia